Zeng et al.
JOCArticle
due to the mild reaction conditions and high functional
group tolerance of the starting materials.6-8 However, the
palladium-catalyzed reductive homocoupling requires ex-
cess external reducing reagents as the hydrogen donor and/
or electron source to regenerate the reductive Pd0 active
species from the oxidative Pd2þ species to complete the
catalytic redox cycle.9 The active Pd0 species is usually
needed to be in situ regenerated because it is prone to
agglomerate to form black sediment and lose the catalytic
activity. Various auxiliary reducing reagents including
hydroquinone,7a formate salt,10 amines,11 zinc,12 indium,13
and triphenylarsine14 have been used for the palladium-
catalyzed reductive homocoupling reactions. The major
drawback of these methods is the necessity for the subse-
quent separation of the excess reducing reagents as well as
their oxidized products. Recently, we found that the palla-
dium-catalyzed reductive homocoupling reactions of various
aromatic halides could be carried out in dimethyl sulfoxide
(DMSO) solution to form the corresponding biaryl products
without the need for any external reductants.15 Although the
solvent DMSO molecules were shown to be involved in the
in situ regeneration of the reductive Pd0 species, we failed to
characterize the oxidation product(s), primarily due to the
complicated oxidation of DMSO molecules. This prompted
us to search for other potential solvents for the palladium-
catalyzed reductive homocoupling of aromatic halides,
which could also be used as the reducing reagents.
halides and oxidation of alcohols could be concomitantly
catalyzed by palladium catalyst without the need for any
external reductants and oxidants. Herein, we would like to
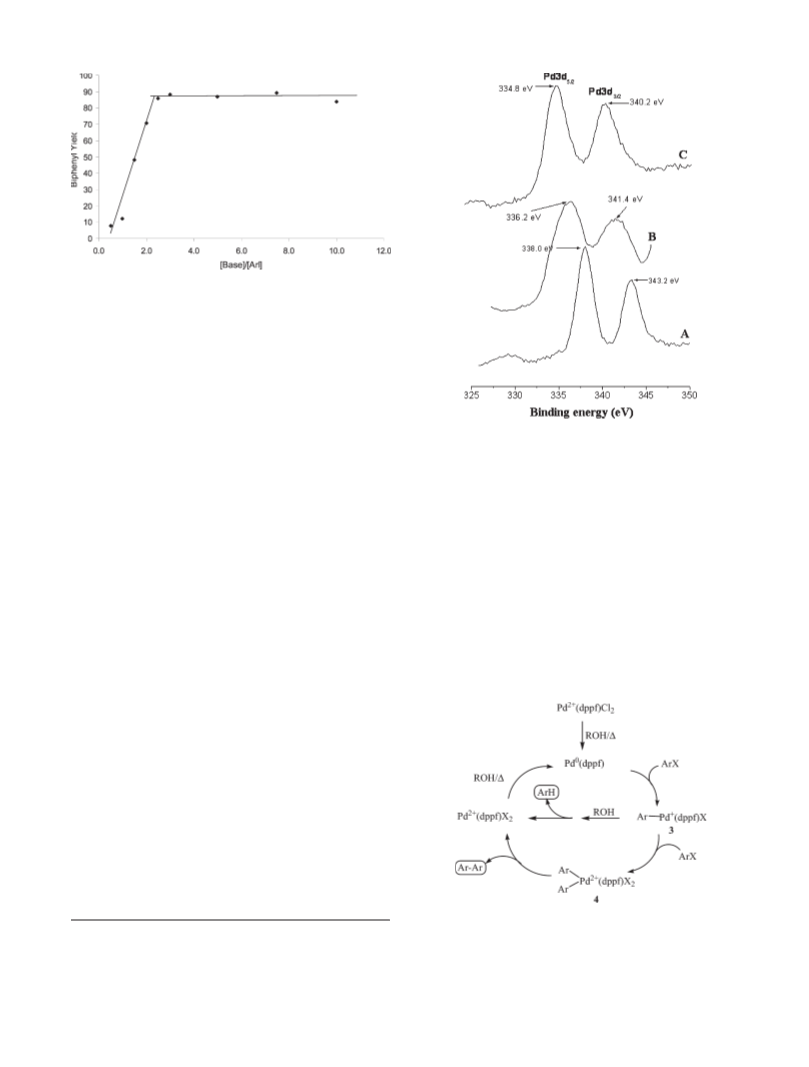
report our initial results of the Pd(dppf)Cl2-catalyzed reduc-
tive homocoupling of aromatic halides in alcohol solutions.
Results and Discussion
Thermodynamic Feasibility for Redox Reactions of Aro-
matic Halides with Alcohols. A series of theoretical calcula-
tions have been performed to examine whether it is
thermodynamically feasible for the redox reactions of aro-
matic halides with alcohols to form biaryls and ketones/
aldehydes. The calculated Gibbs free energy differences (ΔG)
for the redox reactions of aromatic halides with 3-pentanol
are summarized in Scheme 1. The base effects have also been
examined on these redox reactions, and the related results are
summarized in Scheme 2.
SCHEME 1
On the other hand, the Pd2þ-mediated oxidation of alco-
hols has been well documented in the literature.8,16 The key
step for alcohol oxidation is the in situ regeneration of the
oxidative Pd2þ species from the reductive Pd0 species using
various oxidants. Aromatic halides were reported as the
oxidant for alcohol oxidation, but the reduction products
were not characterized.17 Thus, we hypothesized that the
reductive Pd0 species generated from the Pd2þ-mediated
alcohol oxidation could be employed to catalyze the Ull-
mann-type reductive homocoupling of aromatic halides. In
other words, the reductive Pd0 species could be regenerated
in situ from the alcohol oxidation by the oxidative Pd2þ
species. As a result, the reductive homocoupling of aromatic
Examination of Scheme 1 shows that the calculated Gibbs
free energy differences (ΔG1) are all largely negative, suggest-
ing that the redox reactions of these aromatic halides with 3-
pentanol are thermodynamically favorable. Similar conclu-
sions are also reached for the redox reactions of these
aromatic halides with other alcohols such as methanol,
ethanol, and 2-propanol. Moreover, the calculation results
indicate that the Gibbs free energy differences (ΔG2) are
significantly augmented in the presence of base such as
cesium fluoride (CsF) and triethylamine (Et3N). The base
neutralization of the acidic byproduct(s) (HX, X = Cl, Br, or
I) is expected to shift the redox equilibrium toward the right,
i.e., completion. In summary, the redox reactions of aro-
matic halides with alcohols are thermodynamically allowed,
especially in the presence of base.
Pd(dppf)Cl2-Catalyzed Reductive Homocoupling of Iodo-
benzene in Various Alcoholic Solutions. Palladium catalyst is
the catalyst of choice to examine the thermodynamically
allowed redox reaction of aromatic halides with alcohols
because of its remarkable ability to catalyze a wide variety of
organic reactions.8 In the previous studies, we have demon-
strated that Pd(dppf)Cl2 [dppf = 1,10-bis(diphenylphosphino)-
ferrocene] is an effective homogeneous catalyst for the
reductive homocoupling of aromatic halides in DMSO
solution.15 Thus, Pd(dppf)Cl2 was the first choice to test
the model reductive homocoupling of iodobenzene in differ-
ent alcohol solutions. In addition, a similar protocol was
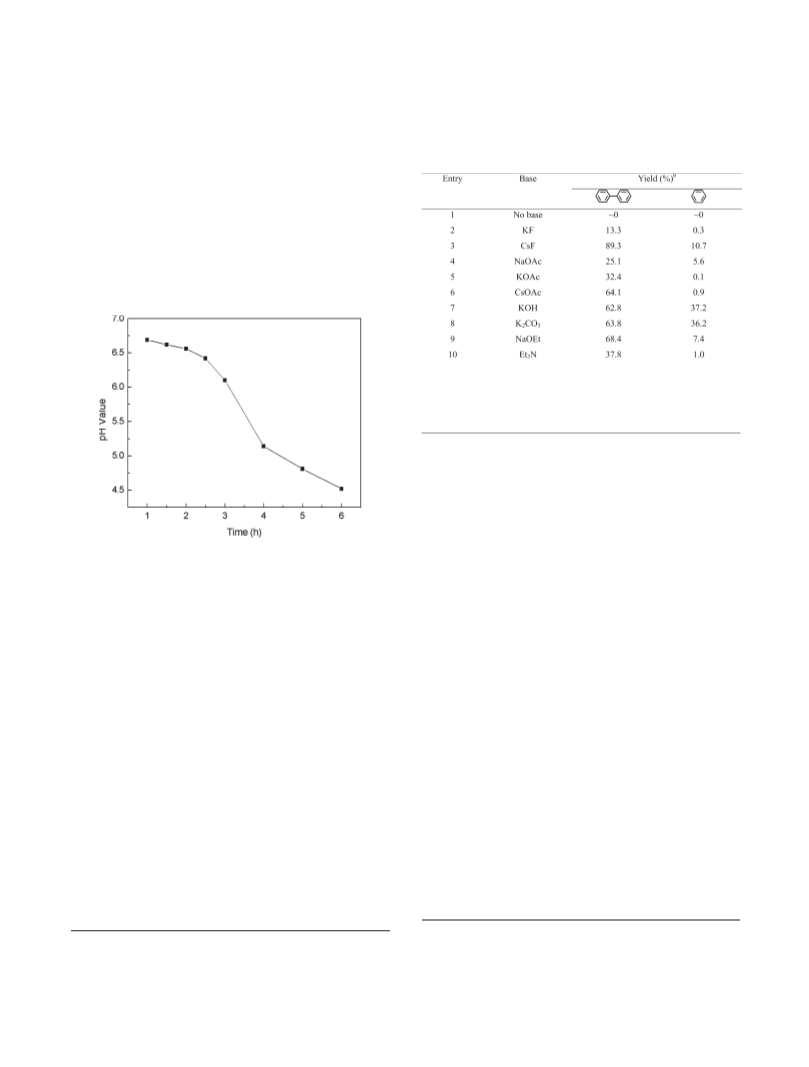
adapted, i.e., 1.0 mmol of iodobenzene, 0.03 mmol of
palladium catalyst, 7.5 mmol of cesium fluoride (CsF) in
5.0 mL of solvent alcohol under an atmosphere of argon. The
homocoupling biphenyl (1) and dehalogenated benzene (2)
are the only products observed for the palladium-catalyzed
reduction of iodobenzene in alcohol solutions as shown
(6) Hassan, J.; Sevignon, M.; Gozzi, C.; Schulz, E.; Lemaire, M. Chem.
Rev. 2002, 102, 1359.
(7) (a) Hennings, D. D.; Iwama, T.; Rawal, V. H. Org. Lett. 1999, 1, 1205.
(b) Kuroboshi, M.; Waki, Y.; Tanaka, H. J. Org. Chem. 2003, 68, 3938.
(8) Tsuji, J. Palladium Reagents and Catalysts, 2nd ed., Wiley, New York,
2004; Chapter 2 .
(9) (a) Jutand, A.; Mosleh, A. J. Org. Chem. 1997, 62, 261. (b) Jutand, A.;
Negri, S.; Mosleh, A. J. Chem. Soc. Chem. Commun. 1992, 1729.
(10) (a) Mukhopadhyay, S.; Rothenberg, G.; Gitis, D.; Wiener, H.;
Sasson, Y. J. Chem. Soc., Perkin Trans. 2 1999, 2481. (b) Mukhopadhyay,
S.; Rothenberg, G.; Qafisheh, N.; Sasson, Y. Tetrahedron Lett. 2001, 42,
6117.
(11) Brenda, M.; Knebelkamp, A.; Greiner, A.; Heitz, W. Synlett 1991,
809.
(12) Li, J.-H; Xie, Y.-X.; Yin, D.-L. J. Org. Chem. 2003, 68, 9867.
(13) Chang, Y. M.; Lee, S. H.; Cho, M. Y.; Yoo, B. W.; Rhee, H. J.; Lee,
S. H.; Yoon, C. M. Synth. Commun. 2005, 35, 1851.
(14) Kikukawa, K.; Yamane, T.; Tagaki, M.; Matsuda, T. J. Chem. Soc.,
Chem. Commun. 1972, 695.
(15) Qi, C.; Sun, X.; Lu, C.; Yang, J.; Du, Y.; Wu, H.; Zhang, X.-M.
J. Organomet. Chem. 2009, 694, 2912.
(16) (a) Peterson, K. P.; Larock, R. C. J. Org. Chem. 1998, 63, 3185.
(b) Stahl, S. S. Angew. Chem., Int. Ed. 2004, 43, 3400. (c) Steinhoff, B. A.;
Stahl, S. S. J. Am. Chem. Soc. 2006, 128, 4348. (d) Arends, I. W. C. E.; Sheldon,
R. A. Modern Oxidation Methods, Backvall, J.-E., Ed.; Wiley-VCH: Weinheim,
2004; Chapter 4 .
(17) Tamaru, Y.; Yamada, Y.; Inoue, K.; Yamamoto, Y.; Yoshida, Z.
J. Org. Chem. 1983, 48, 1286.
J. Org. Chem. Vol. 75, No. 8, 2010 2557

 Zeng, Minfeng
Zeng, Minfeng
 Du, Yijun
Du, Yijun
 Shao, Linjun
Shao, Linjun
 Qi, Chenze
Qi, Chenze
 Zhang, Xian-Man
Zhang, Xian-Man