Brief Articles
Journal of Medicinal Chemistry, 2006, Vol. 49, No. 21 6415
silica gel flash column chromatography (ethyl acetate/n-hexane )
1:2) to afford 11 as a pale yellow solid, yield 41%; mp 94.0-94.9
°C. 1H NMR (400 MHz, CDCl3) δ 3.64 (s, 6H), 3.74 (s, 3H), 3.79
(s, 3H,), 6.25 (d, J ) 2.4 Hz, 1H), 6.30 (dd, J ) 8.4, 2.4 Hz, 1H),
6.42 (d, J ) 12 Hz, 1H), 6.49 (d, J ) 14.8 Hz, 3H), 7.03 (d, J )
8.4 Hz). 13C NMR (100 MHz, CDCl3) δ 55.1, 55.7, 60.7, 100.6,
104.2, 105.7, 115.985, 125.6, 130.5, 130.8, 132.2, 137.2, 144.8,
152.6, 160.0. MS (EI) m/z 315 (M+, 90%), 300 (100%). HRMS
(EI) calcd for C18H21NO4 (M+), 315.1471; found, 315.1471. Anal.
(C18H21NO4) C, H, N.
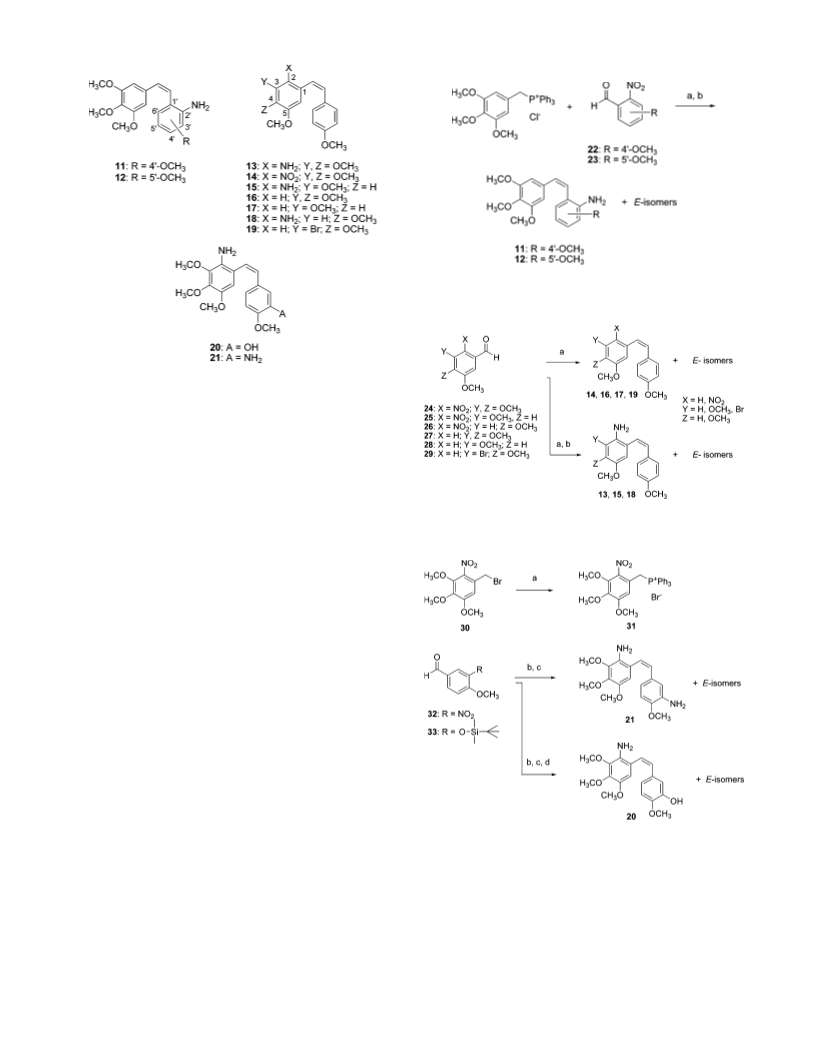
2-Amino-3,4,4′,5-tetramethoxy-(Z)-stilbene (13). The title com-
pound was obtained in 39% overall yield from (4-methoxybenzyl)-
triphenylphosphonium bromide and 2-nitro-3,4,5-trimethoxyben-
zaldehyde (24). 1H NMR (500 MHz, CD3OD) δ 3.52 (s, 3H), 3.73
(s, 3H), 3.82 (s, 3H), 3.83 (s, 3H), 6.35 (d, J ) 11.9 Hz, 1H), 6.45
(s, 1H), 6.55 (d, J ) 12.0 Hz, 1H), 6.74 (d, J ) 8.6 Hz, 2H), 7.15
(d, J ) 8.6 Hz, 2H). 13C NMR (125 MHz, CD3OD) δ 55.6, 56.9,
60.8, 61.3, 110.3, 114.5, 120.0, 125.0, 130.8, 131.2, 131.4, 133.6,
142.8, 143.4, 146.6, 160.4. MS (EI) m/z 315 (M+, 100%), 300
(58%). HRMS (EI) calcd for C18H21NO4 (M+), 315.1469; found,
315.1470. Anal. (C18H21NO4) C, H, N.
procedures for biological evaluations. This material is available free
References
(1) Jordan, A.; Hadfield, J. A.; Lawrence, N. J.; McGown, A. T. Tubulin
as a Target for Anticancer Drugs: Agents Which Interact with the
Mitotic Spindle. Med. Res. ReV. 1998, 18, 259-296.
(2) Hamel, E. Antimitotic Natural Products and Their Interactions with
Tubulin. Med. Res. ReV. 1996, 16, 207-231.
(3) Jordan, M. A.; Wilson, L. Microtubules as a Target for Anticancer
Drugs. Nat. ReV. Cancer 2004, 4, 253-265.
(4) Siemann, D. W.; Bibby, M. C.; Dark, G. G.; Dicker, A. P.; Eskens,
F. A. L. M.; Horsman, M. R.; Marme, D.; LoRusso, P. M.
Differentiation and Definition of Vascular-Targeted Therapies. Clin.
Cancer Res. 2005, 11, 416-420.
(5) Siemann, D. W. Vascular-Targeted Therapies in Oncology; John
Wiley & Sons: New York, 2006.
(6) Chaplin, D. J.; Horsman, M. R.; Siemann, D. W. Current Develop-
ment Status of Small-Molecule Vascular Disrupting Agents. Curr.
Opin. InVest. Drugs 2006, 7, 522-528.
(7) Beerepoot, L. V.; Radema, S. A.; Witteveen, E. O.; Thomas, T.;
Wheeler, C.; Kempin, S.; Voest, E. E. Phase I Clinical Evaluation
of Weekly Administration of the Novel Vascular-Targeting Agent,
ZD6126, in Patients With Solid Tumors. J. Clin. Oncol. 2006, 24,
1491-1498.
2-Amino-3,4′,5-trimethoxy-(Z)-stilbene (15). The title com-
pound was obtained in 40% overall yield from (4-methoxybenzyl)-
triphenylphosphonium bromide and 3,5-dimethoxy-2-nitrobenzal-
(8) Thorpe, P. E. Vascular Targeting Agents as Cancer Therapeutics.
Clin. Cancer Res. 2004, 10, 415-427.
1
dehyde (25); mp 83.2-86.3 °C. H NMR (500 MHz, CDCl3) δ
(9) Cirla, A.; Mann, J. Combretastatins: from Natural Products to Drug
Discovery. Nat. Prod. Rep. 2003, 20, 558-564.
3.63 (s, 3H), 3.75 (s, 3H), 3.83 (s, 3H), 6.31 (d, J ) 2.3 Hz, 1H),
6.39 (s, 1H), 6.41 (d, J ) 12.9 Hz, 1H), 6.58 (d, J ) 12.0 Hz, 1H),
6.73 (d, J ) 8.7 Hz, 2H), 7.19 (d, J ) 8.7 Hz, 2H). 13C NMR (125
MHz, CDCl3) δ 55.0, 55.5, 98.4, 103.9, 113.4, 123.5, 124.2, 127.2,
129.2, 130.0, 130.8, 148.4, 152.1, 158.8. MS (EI) m/z 285 (M+,
100%), 270 (29%). HRMS (EI) calcd for C17H19NO3 (M+),
285.1369; found, 285.1367. Anal. (C17H19NO3) C, H, N.
(10) Pettit, G. R.; Cragg, G. M.; Herald, D. L.; Schmidt, J. M.;
Lohavanijaya, P. Isolation and Structure of Combretastatin. Can. J.
Chem. 1982, 60, 1374-1376.
(11) Pettit, G. R.; Singh, S. B.; Niven, M. L.; Hamel, E.; Schmidt, J. M.
Isolation, Structure and Synthesis of Combretastatins A-1 and B-1,
Potent New Inhibitors of Microtubule Assembly, Derived from
Combretum caffrum. J. Nat. Prod. 1987, 50, 119-131.
(12) Morinaga, Y.; Nihei, Y.; Ohishi, K.; Suga, Y.; Akiyama, Y.; Tsuji,
T. Novel Combretastatin Analogues Effective against Murine Solid
Tumors: Design and Structure-Activity Relationships. J. Med.
Chem. 1998, 41, 3022-3032.
2,3′-Diamino-3,4,4′,5-tetramethoxy-(Z)-stilbene (21). The title
compound was obtained in 34% overall yield from 2-nitro-3,4,5-
(trimethoxybenzyl)triphenylphosphonium bromide (31) and 4-meth-
1
(13) Pettit, G. R.; Minardi, M. D.; Rosenberg, H. J.; Hamel, E.; Bibby,
M. C.; Martin, S. W.; Jung, M. K.; Pettit, R. K.; Cuthbertson, T. J.;
Chapuis, J.-C. Antineoplastic Agents. 509. Synthesis of Fluorcomb-
statin Phosphate and Related 3-Halostilbenes. J. Nat. Prod. 2005,
68, 1450-1458.
(14) Cushman, M.; Nagarathnam, D.; Gopal, D.; He, H. M.; Lin, C. M.;
Hamel, E. Synthesis and Evaluation of Analogues of (Z)-1-(4-
Methoxyphenyl)-2-(3,4,5-trimethoxyphenyl)ethane as Potential Cy-
totoxic and Antimitotic Agents. J. Med. Chem. 1992, 35, 2293-
2306.
(15) Pettit, G. R.; Grealish, M. P.; Jung, M. K.; Hamel, E.; Pettit, R. K.;
Chapuis, J. C.; Schmidt, J. M. Antineoplastic Agents. 465. Structural
Modification of Resveratrol: Sodium Resverastatin Phosphate. J.
Med. Chem. 2002, 45, 2534-2542.
(16) Tozer, G. M.; Kanthou, C.; Baguley, B. C. Disrupting Tumour Blood
Vessels. Nat. ReV. Cancer 2005, 5, 423-435.
(17) Pettit, G. R.; Temple, C., Jr.; Narayanan, V. L.; Varma, R.; Simpson,
M. J.; Boyd, M. R.; Rener, G. A.; Bansal, N. Antineoplastic Agents
322. Synthesis of Combretastatin A-4 prodrugs. Anticancer Drug Des.
1995, 10, 299-309.
(18) Pettit, G. R.; Lippert III, J. W. Antineoplastic Agents 429. Syntheses
of Combretastatin A-1 and Combretastatin B-1 prodrugs. Anticancer
Drug Des. 2000, 15, 203-216.
(19) Hori, K.; Saito, S.; Nihei, Y.; Suzuki, M.; Sato, Y. Antitumor Effects
Due to Irreversible Stoppage of Tumor Tissue Blood Flow: Evalu-
ation of A Novel Combretastatin A-4 Derivative, AC7700. Jpn. J.
Cancer Res. 1999, 90, 1026-1038.
(20) Chaplin, D. J.; Garner, C. M., III; Kane, R. R.; Pinney, K. G.;
Prezioso, J. A. Functionalized Stilbene Derivatives as Improved
Vascular Targeting Agents. WO 03035008 A2, 2003.
oxy-3-nitrobenzaldehyde (32); mp 97.3-98.1 °C. H NMR (500
MHz, acetone-d6) δ 3.54 (s, 3H), 3.77 (s, 3H), 3.79 (s, 3H), 3.80
(s, 3H), 4.01 (s, 2H), 4.25 (s, 2H), 6.26 (d, J ) 12.1 Hz), 6.39 (d,
J ) 12.1 Hz, 1H), 6.53 (s, 1H), 6.56 (dd, J ) 8.2, 1.7 Hz, 1H),
6.66 (d, J ) 8.2 Hz, 1H), 6.68 (d, J ) 1.8 Hz, 1H). 13C NMR (125
MHz, acetone-d6) δ 56.1, 57.1, 61.0, 61.4, 110.5, 111.2, 117.0,
120.4, 120.9, 124.9, 131.3, 132.1, 133.6, 137.3, 143.0, 143.5, 146.8,
148.8. MS (EI) m/z 330 (M+, 100%), 315 (27%). HRMS (EI) calcd
for C18H22N2O4 (M+), 330.1578; found, 330.1570. Anal. (C18H22N2O4)
C, H, N.
2-Amino-3′-hydroxy-3,4,4′,5- tetramethoxy-(Z)-stilbene (20).
The title compound was obtained in 30% overall yield from 2-nitro-
(3,4,5-trimethoxybenzyl)triphenylphosphonium bromide (31) and
3-(tert-butyldimethylsilyloxy)-4-methoxybenzaldehyde (33) accord-
ing to the above procedure and one extra procedure, which was 3
equiv of tetra-n-butylammonium fluoride/THF at room temperature,
stirring for 1 h. 1H NMR (500 MHz, CD3OD) δ 3.55 (s, 3H), 3.79
(s, 3H), 3.82 (s, 3H), 3.83 (s, 3H), 6.33 (d, J ) 12 Hz, 1H), 6.49
(s, 1H, H-6), 6.50 (d, J ) 11.9 Hz, 1H), 6.71 (dd, J ) 8.2, 1.9 Hz,
1H), 6.74 (d, J ) 1.8 Hz, 1H), 6.77 (d, J ) 8.2 Hz, 1H). 13C NMR
(125 MHz, CD3OD) δ 56.3, 57.0, 60.9, 61.3, 110.4, 112.3, 116.5,
120.0, 122.0, 125.1, 131.4, 131.5, 133.5, 142.9, 143.4, 146.7, 147.1,
148.5. MS (EI) m/z 331 (M+, 100%), 284 (25%). HRMS (EI) calcd
for C18H21NO5 (M+), 331.1422; found, 331.1421. Anal. (C18H21-
NO5) C, H, N.
(21) Monk, K. A.; Siles, R.; Hadimani, M. B.; Mugabe, B. E.; Ackely, J.
F.; Studerus, S. W.; Edvardsen, K.; Trawick, M. L.; Garner, C. M.;
Rhodes, M. R.; Pettit, G. R.; Pinney, K. G. Design, Synthesis, and
Biological Evaluation of Combretastatin Nitrogen-containing Deriva-
tives as Inhibitors of Tubulin Assembly and Vascular Disrupting
Agents. Bioorg. Med. Chem. 2006, 14, 3231-3244.
(22) Cushman, M.; Nagarathnam, D.; Gopal, D.; Chakraborti, A. K.; Lin,
C. M.; Hamel, E. Synthesis and Evaluation of Stilbene and Dihy-
drostilbene Derivatives as Potential Anticancer Agents That Inhibit
Tubulin Polymerization. J. Med. Chem. 1991, 34, 2579-2588.
Acknowledgment. This research was supported by the
National Science and Technology Program for Biotechnology
and Pharmaceuticals, Taiwan (Grant No. NSC 94-2323-B-038-
005), National Science Council of the Republic of China (Grant
No. NSC 95-2752-B-400-001-PAE), and National Health Re-
search Institutes, Taiwan (Grant No. 92A1CAPP06-1).
Supporting Information Available: Spectral data of com-
pounds 12, 14, 16-19, 22-25, 30, 31, 33 and experimental
JM060616K

 Chang, Jang-Yang
Chang, Jang-Yang
 Yang, Ming-Fang
Yang, Ming-Fang
 Chang, Chi-Yen
Chang, Chi-Yen
 Chen, Chi-Ming
Chen, Chi-Ming
 Kuo, Ching-Chuan
Kuo, Ching-Chuan
 Liou, Jing-Ping
Liou, Jing-Ping