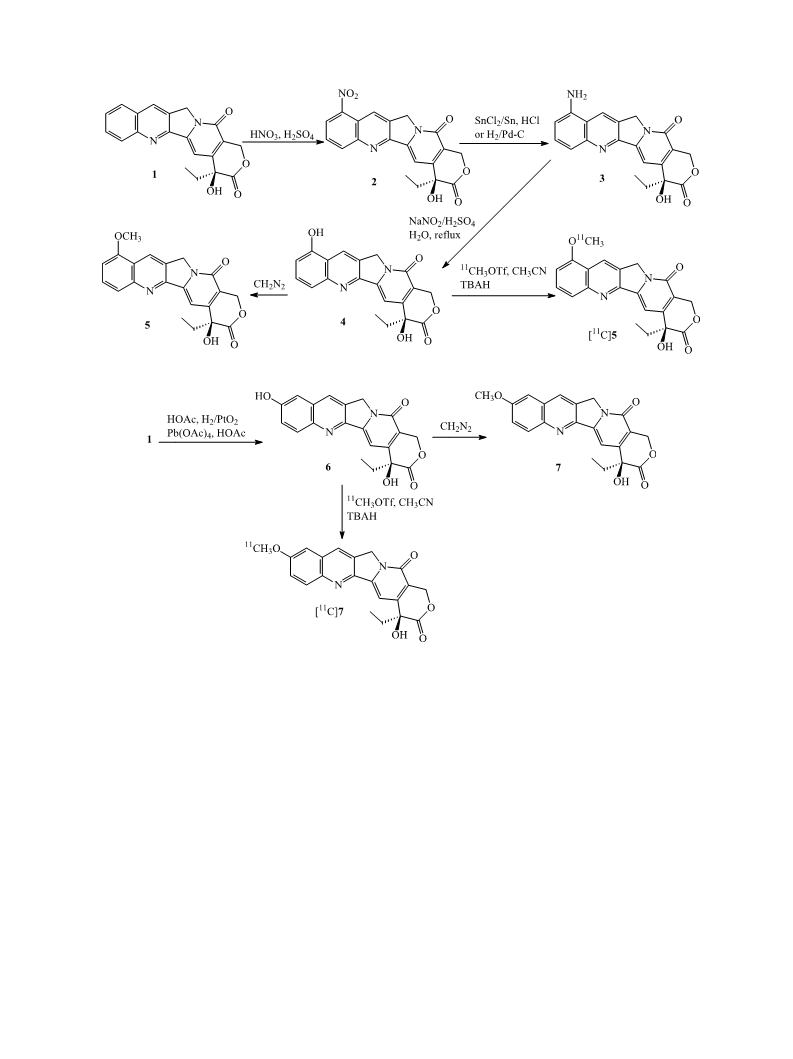
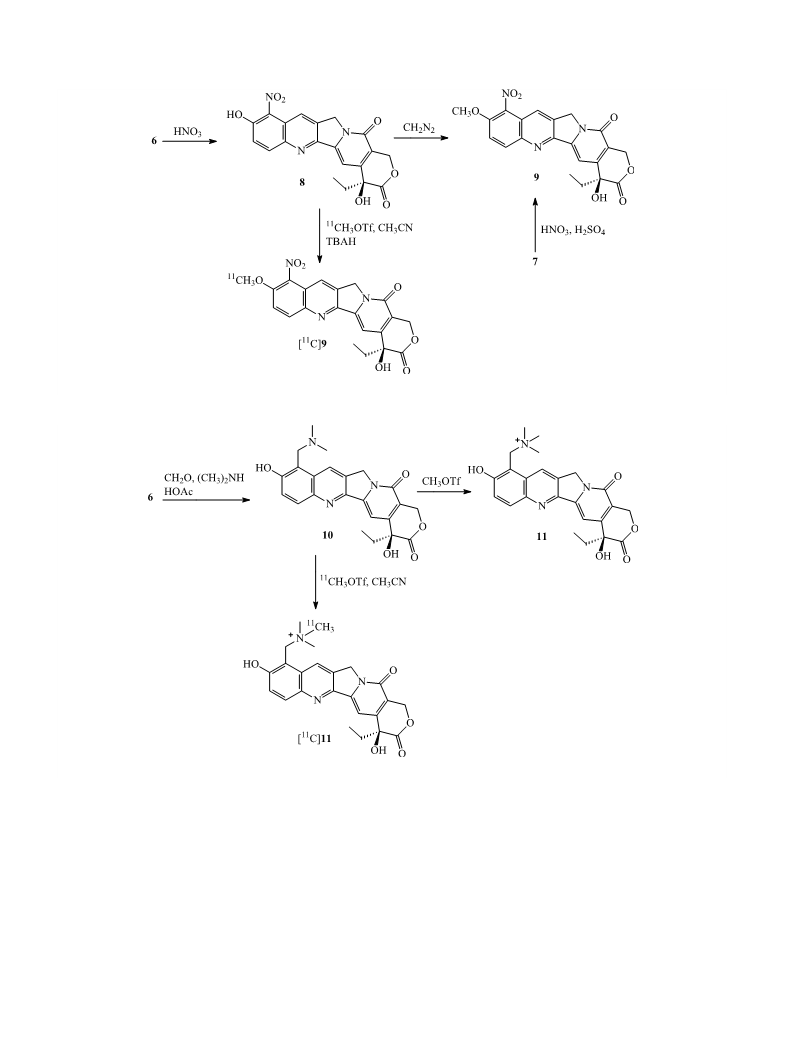
M. Gao et al. / Bioorg. Med. Chem. Lett. 15 (2005) 3865–3869
3869
1
column 1 · 1 cm was obtained from E. S. Industries,
Berlin, NJ, and part number 300121-C18-BD 10l. Semi-
prep SiO2 Sep-Pak type cartridge was obtained from
Waters Corporate Headquarters, Milford, MA, USA.
Sterile Millex-GS 0.22 lm vented filter unit was
obtained from Millipore Corporation, Bedford, MA,
yield 60%, R
(300 MHz, acetone-d
1.93–1.97 (m, 2H, MeCH ), 3.23 (s, 9H, (CH ) N ), 5.24
f
= 0.25 (1:9, MeOH/CH
2
Cl
2
). H NMR
6
): d 0.95 (t, J = 7.35 Hz, 3H, CH
3
),
+
2
3 3
(s, 2H), 5.46 (d, J = 13.24 Hz, 2H), 5.56 (s, 2H), 7.93 (s,
1H), 8.12 (d, J = 9.56 Hz, 1H), 8.60 (d, J = 9.56 Hz, 1H),
9.88 (s, 1H), 11.32 (s, 1H, 10-OH). (l) Tracers [ C]5,
[ C]7, and [ C]9: typical experimental procedure for the
radiosynthesis: the precursor (4, 6, or 8) (0.6–1.0 mg) was
1
1
1
1
11
USA. (b) Compound 2: a yellow solid, yield 24%, mp
= 0.81 (1:9, MeOH/CH
1
). H NMR
1
(
90–192 ꢁC, R
300 MHz, DMSO-d
f
2
Cl
2
6
): d 0.88 (t, J = 7.35 Hz, 3H, CH
3
),
3
dissolved in CH CN (300 lL). To this solution was added
1
1
8
1
1
.86 (dq, J = 4.41, 7.35 Hz, 2H, CH Me), 5.31 (s, 2H, H-
7), 5.43 (s, 2H, H-5), 6.56 (s, 1H, 20-OH), 7.36 (s, 1H),
.01 (t, J = 8.50 Hz, 1H), 8.51 (t, J = 8.50 Hz, 2H), 9.13 (s,
tetrabutylammonium hydroxide (TBAH) (2–3 lL, 1 M
solution in methanol). The mixture was transferred to a
small volume, three-neck reaction tube. CH OTf was
3
2
1
1
H). (c) Compound 3: a yellow solid, yield 48% (H
2
-
passed into the air-cooled reaction tube at À15 ꢁC to
À20 ꢁC, which was generated by a Venturi cooling device
powered with 100 psi compressed air, until radioactivity
reached a maximum (ꢀ3 min), then the reaction tube was
heated at 70–80 ꢁC for 3 min. The contents of the reaction
0%Pd/C), yield 81% (SnCl /Sn–HCl), mp 300 ꢁC (dec.),
2
1
R
f
= 0.50 (1:9, MeOH/CH
DMSO-d ): d 0.87 (t, J = 7.0 Hz, 3H, CH
m, 2H, CH Me), 5.25 (s, 2H, H-17), 5.41 (s, 2H, H-5),
.11 (s, 2H, NH ), 6.51 (s, 1H, 20-OH), 6.78 (d,
2
Cl
2
). H NMR (300 MHz,
6
3
), 1.83–1.87
(
6
2
tube were diluted with NaHCO (1 mL, 0.1 M). This
2
3
J = 7.40 Hz, 1H), 7.29 (t, J = 6.26 Hz, 2H), 7.50
t,J = 8.10 Hz, 1H), 8.83 (s, 1H). (d) Compound 4: a
solution was passed onto a C18 cartridge by gas pressure.
The cartridge was washed with H
(
2
O (2 · 3 mL), and the
yellow solid, yield 36%, mp 224–224 ꢁC, R = 0.58 (1:9,
aqueous washing was discarded. The product was eluted
from the column with EtOH (2 · 3 mL), and then passed
onto a rotatory evaporator. The solvent was removed by
evaporation under high vacuum. The labeled product
f
1
MeOH/CH Cl ). H NMR (300 MHz, DMSO-d ): d 0.87
2
2
6
(
t, J = 7.35 Hz, 3H, CH
3
), 1.85 (dq, J = 4.41, 7.35 Hz, 2H,
2
CH Me), 5.25 (s, 2H, H-17), 5.42 (s, 2H, H-5), 6.52 (s, 1H,
1
1
11
11
2
0-OH), 7.02 (d, J = 7.36 Hz, 1H), 7.03 (s, 1H), 7.57–7.66
m, 2H), 8.81 (s, 1H), 10.72 (s, 1H, 9-OH). (e) Compound
: a yellow solid, yield 94%, mp 223–225 ꢁC, R = 0.52
). H NMR (300 MHz, DMSO-d ):
d 0.86 (t, J = 7.35 Hz, 3H, CH ), 1.86 (dq, J = 5.15,
[ C]5, [ C]7, or [ C]9 was formulated with NaH PO
2 4
(
(50 mM), whose volume was dependent upon the use of
1
1
11
11
5
f
the labeled product [ C]5, [ C]7, or [ C]9 in tissue
biodistribution studies (ꢀ6 mL, 3 · 2 mL) or in micro-
PET imaging studies (1–3 mL) of cancer animal mod-
1
(
1:19, MeOH/CH
2
Cl
2
6
3
8
,11,12
7
.35 Hz, 2H, CH
2
Me), 4.03 (s, 3H, OCH
3
), 5.23 (s, 2H, H-
els,
sterile-filtered through a sterile vented Millex-GS
1
J = 6.61 Hz, 1H), 7.30 (s, 1H), 7.68–7.75 (m, 2H), 8.82 (s,
7), 5.42 (s, 2H, H-5), 6.52 (s, 1H, 20-OH), 7.15 (d,
0.22 lm cellulose acetate membrane and collected into a
sterile vial. Total radioactivity was assayed and total
volume was noted. The overall synthesis time was
ꢀ20 min. The decay corrected radiochemical yield, from
1
2
H). (f) Compound 6: a yellow solid, yield 51%, mp 265–
1
67 ꢁC,
R
f
= 0.50 (1:9, MeOH/CH
): d 0.87 (t, J = 7.35 Hz, 3H, CH
.85 (dq, J = 4.40, 7.35 Hz, 2H, CH Me), 5.20 (s, 2H, H-
2
Cl
2
).
H
NMR
1
1
(
300 MHz, DMSO-d
6
3
),
2
CO , was 30–50%, and the radiochemical purity was
1
1
>95% by analytical HPLC. Retention times in the
analytical HPLC system were: RT4 = 1.67 min,
2
7), 5.39 (s, 2H, H-5), 6.49 (s, 1H, 20-OH), 7.25 (t,
1
1
RT6 = 1.99 min, RT8 = 1.64 min; RT[ C]5 = 2.49 min,
J = 1 Hz, 2H), 7.41 (dd, J = 2.58, 9.18 Hz, 1H), 7.98 (d,
J = 8.83 Hz, 1H), 8.42 (s, 1H), 10.31 (s, 1H, 10-OH). (g)
1
RT[ C]7 = 2.91 min, RT[ C]9 = 2.33 min. The chemical
1
11
1
purities of the target tracers [ C]5, [ C]7, and [ C]9
1
11
11
Compound 7: a yellow solid, yield 88%, mp 254–255 ꢁC,
1
). H NMR (300 MHz,
11
were >93%. (m) Tracer [ C]11: the precursor 10 (0.6–
R
f
= 0.84 (1:9, MeOH/CH
2
Cl
2
DMSO-d
(
5
6
): d 0.87 (t, J = 7.35 Hz, 3H, CH
m, 2H, CH Me), 3.92 (s, 3H, OCH ), 5.21 (s, 2H, H-17),
.40 (s, 2H, H-5), 6.50 (s, 1H, 20-OH), 7.25 (s, 1H), 7.45
3
), 1.81–1.90
1 mg) was dissolved in acetonitrile (250 lL). The mixture
was transferred to a small volume, three-neck reaction
2
3
1
tube. CH OTf was passed into air-cooled reaction tube
1
3
(
1
2
(
1
1
7
d, J = 6.62 Hz, 2H), 8.01 (d, J = 10.3 Hz, 1H), 8.50 (s,
H). (h) Compound 8: a yellow solid, yield 45%, mp 205–
at À15 ꢁC to À20 ꢁC, which was generated by a Venturi
cooling device powered with 100 psi compressed air, until
radioactivity in solution reached a maximum (2–3 min),
then reaction tube was isolated and heated at 70–80 ꢁC for
1
08 ꢁC, R = 0.58 (1:9, MeOH/CH Cl ).
H NMR
f
2
2
300 MHz, DMSO-d ): d 0.86 (t, J = 7.35 Hz, 3H, CH ),
6
3
.84 (dq, J = 4.41, 7.35 Hz, 2H, CH
7), 5.41 (s, 2H, H-5), 6.51 (s, 1H, 20-OH), 7.28 (s, 1H),
2
Me), 5.24 (s, 2H, H-
2–3 min. The reaction tube was connected to the SiO
Pak. The product solution was passed onto the SiO
2
Sep-
Sep-
2
.63 (d, J = 9.56 Hz, 1H), 8.24 (d, J = 9.56 Hz, 1H), 8.41
Pak for SPE purification by gas pressure. The reaction
tube and Sep-Pak were washed with ethanol (5 mL), and
the washing solution was discarded to a waste bottle. The
(
s, 1H), 12.10 (s, 1H, 10-OH). (i) Compound 9: a yellow
solid, yield 90%, mp 218 ꢁC (dec.), R = 0.86 (1:9, MeOH/
). H NMR (300 MHz, DMSO-d ): d 0.87 (t,
f
1
CH
2
Cl
2
6
2
product was eluted from the Sep-Pak with 90:8:2 H O/
J = 7.35 Hz, CH ), 1.86 (dq, J = 4.41, 6.62 Hz, CH Me),
EtOH/HOAc (2–4 mL) and sterile-filtered through a
0.22 lm cellulose acetate membrane and collected in a
sterile vial. The pH was adjusted to 5.5–7.0 with 2 M
NaOH and 150 mM NaH PO mixed solution (1/20, 0.2–
3
2
4
6
1
.11 (s, 3H, OCH ), 5.24 (s, 2H, H-17), 5.41 (s, 2H, H-5),
.53 (s, 1H, 20-OH), 7.31 (s, 1H), 8.00 (d, J = 9.56 Hz,
H), 8.43 (t, J = 4.83 Hz, 2 H). (j) Compound 10: a
3
2
4
hygroscopic yellow solid, yield 65%, R = 0.27 (1:9,
f
0.4 mL). Total radioactivity was assayed and the total
volume (2.5–5.0 mL) was noted. The overall synthesis time
was 10–15 min. The decay corrected yields, from CO ,
2
1
MeOH/CH
2
Cl
2
). H NMR (300 MHz, DMSO-d
), 1.86 (q, J = 4.0 Hz, 2H, CH
.89 (s, 2H, CH CO ), 2.29 (s, 6H, (CH ) N), 3.99 (s, 2H,
6
): d 0.87
1
1
(
t, J = 7.0 Hz, 3H, CH
3
2
Me),
1
were 40–65%, and the radiochemical purity was >99% by
analytical HPLC. Retention times in the analytical HPLC
system were RT10 = 2.98 min and RT[ C]11 = 1.83 min.
3
2
3 2
2
ArCH N), 5.22 (s, 2H, H-17), 5.40 (s, 2H, H-5), 7.24 (s,
1
1
1
8
H), 7.39 (d, J = 8.83 Hz, 1H), 7.94 (d, J = 9.56 Hz, 1H),
.58 (s, 1H). (k) Compound 11: a hygroscopic yellow solid,
1
1
The chemical purity of the target tracer [ C]11 was >95%.

 Gao, Mingzhang
Gao, Mingzhang
 Miller, Kathy D.
Miller, Kathy D.
 Sledge, George W.
Sledge, George W.
 Zheng, Qi-Huang
Zheng, Qi-Huang