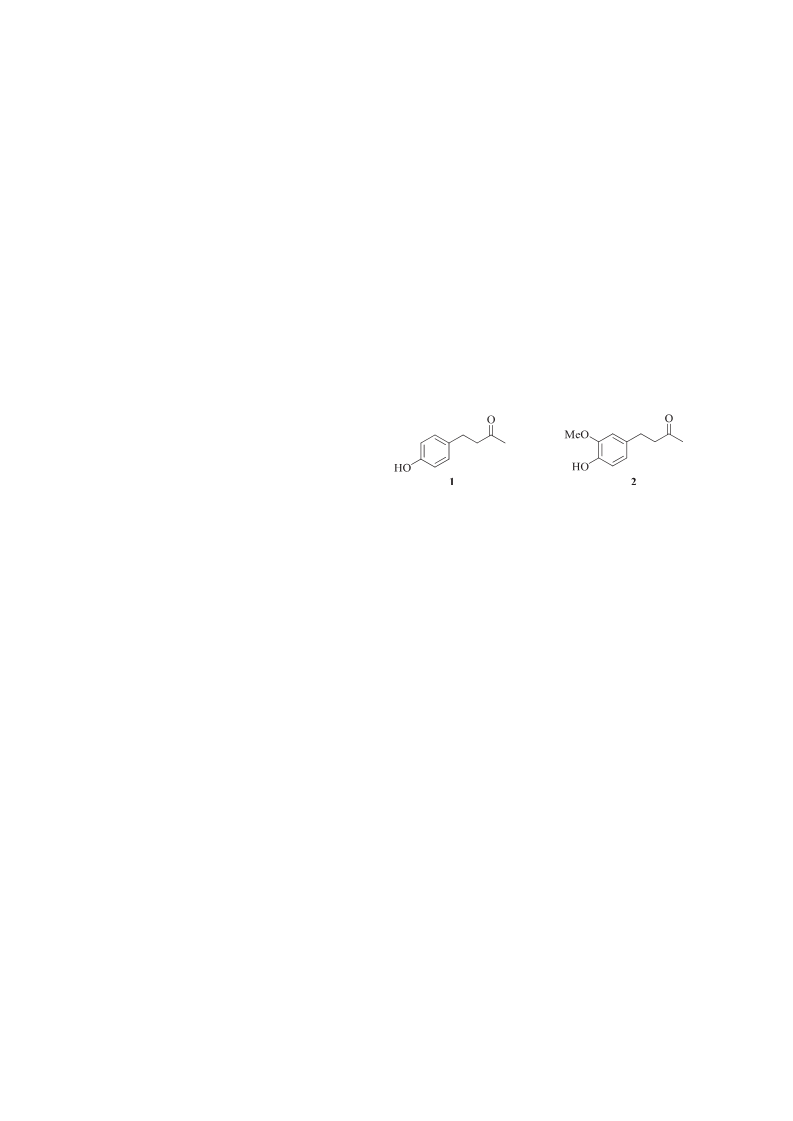M. Bandarenko – V. Kovalenko · Raspberry and Ginger Ketones
887
enone 4 (Scheme 2). The reaction mixture contained fluxing under argon atmosphere for 15 min. Then, the mix-
about 98% of ketone 2 and 2% of alcohol 6 based on ture was cooled to room temperature, and unsaturated ketone
3
(1.50 g, 9.25 mmol) was added. The stirred mixture was
GC. Due to a low melting point, we had some trouble
with the crystallization of zingerone 2 and preferred
column chromatography for its purification. However,
we also succeeded in crystallization of crude 2 us-
ing seed crystals of the pure product and a mixture of
methanol/water as solvent.
treated with hydrogen (∼1 atm) keeping the external temper-
◦
ature at 25 – 30 C. Once the green color of the methanolic
solution had disappeared (about 1 – 2 h), the hydrogenation
was stopped, and the mixture was filtered. The nickel boride
cake was washed with methanol, and the combined filtrates
were evaporated under reduced pressure. Then, the residue
was treated with saturated aqueous NH Cl (20 mL) and ethyl
4
Conclusion
acetate (20 mL). The organic phase was separated, and the
aqueous phase was extracted with ethyl acetate (10 mL). The
combined organic phases were washed with brine (10 mL),
Readily available products of the aldol condensa-
tion of 4-hydroxybenzaldehyde or vanillin with ace-
tone were hydrogenated in the presence of cheap nickel
boride to give raspberry and zinger ketones, respec-
tively, in good yield. The advantage of the reported
procedures is that a two-step sequence (preparation of
catalyst and hydrogenation) can be carried out in a one-
pot reaction and in a short time.
dried with Na SO , and evaporated under reduced pressure.
2
4
According to GC analysis, the unpurified reaction mixture
contained > 98% of product 1 (t = 21.3 min), 1% of by-
R
product 5 (t = 21.6 min) and traces of starting enone 3
R
(tR = 25.5 min). The residue was recrystallized from a mix-
ture of methanol/water (1 : 3) to give pure raspberry ke-
tone 1 in a yield of 1.14 g (75%) as colorless crystals;
◦
◦
m. p. 82 – 83 C (lit. [12]: m. p. 80 – 82 C). Alternatively,
the residue was recrystallized from the mixture of toluene-
Experimental Section
ethyl acetate (4 : 1), the yield of compound 1 being 1.08 g
◦
(
(
3
71%), m. p. 81 – 83 C. – IR (KBr): ν = 3373 (OH), 1691
Melting points were determined with a capillary appa-
ratus. IR spectra were recorded on a Vertex 70 spectrom-
eter. GC-MS analyses were performed using a Shimadzu
−1
1
C=O) cm . – H NMR (400 MHz, CDCl ): δ = 2.14 (s,
3
H, CH ), 2.71 – 2.76 (m, 2H, CH ), 2.80 – 2.85 (m, 2H,
3
2
CH ), 6.29 (br s, 1H, OH), 6.75 – 6.79 (m, 2H, aromatic
2
TM
GCMS-QP2010 instrument equipped with an Equity -5
CH), 7.00 – 7.04 (m, 2H, aromatic CH) ppm. The spectral
data were similar to those reported in the literature [12, 20]. –
capillary column (30 m, 0.25 mm ID, 0.25 µm film thick-
ness) in the electron impact ionization mode at 70 eV. The
carrier gas helium was applied. 1H NMR spectra were
recorded on a Bruker AC 400 instrument at 400 MHz, CHCl3
was used as internal standard (δ = 7.26 ppm). Elemen-
+
+
MS: m/z (%) = 164 (20) [M] , 149 (5) [M–CH ] , 131 (3)
3
+
+
[
M–CH –H O] , 121 (11) [M–CH CO] , 107 (100) [M–
3 2 3
+
CH COCH ] . – C H O (164.2): calcd. C 73.15, H 7.37;
3
2
10 12 2
found C 73.28, H 7.28.
TM
tal analyses were performed using a Thermo Scientific
Flash 2000 CHNS/O analyzer. Methanol was freshly dis-
0
0
4
-(4 -Hydroxy-3 -methoxyphenyl)butan-2-one (2)
tilled from magnesium methoxide. Petroleum ether of
b. p. 40 – 70 C was used. Silica gel 60 F 254 plates
were used for TLC analysis, and column chromatog-
raphy was performed on silica gel 70 – 230 mesh using
Compound 2 was prepared from enone 4 (1.78 g,
.26 mmol) in accordance to the procedure for raspberry
ketone 1. As shown by GC analysis, the unpurified reac-
◦
9
tion mixture contained 98% of product 2 (t = 23.6 min)
R
1
0 – 30% solutions of ethyl acetate in petroleum ether as
and 2% of by-product 6 (t = 24.1 min). The crude product
0
R
eluent. Starting compounds (E)-4-(4 -hydroxyphenyl)but-3-
en-2-one (3) and (E)-4-(4 -hydroxy-3-methoxyphenyl)but-
was purified by column chromatography to give zingerone
0
2
as a colorless liquid which was crystallized in a refrig-
3
-en-2-one (4) were prepared by NaOH-catalyzed con-
◦
erator. Yield 1.69 g (94%); m. p. 39 – 40 C (lit. [2]: m. p.
4
densation of acetone with 4-hydroxybenzaldehyde and 4-
hydroxy-3-methoxybenzaldehyde, respectively (see for ex-
ample ref. [12]).
◦
0 – 41 C). Alternatively, the crude product was recrystal-
lized from a mixture of methanol/water (1 : 3) using seed
crystals, the yield of compound 2 being 1.23 g (68%); m. p.
◦
−1
.
0
41 – 42 C. – IR (KBr): ν = 3400 (OH), 1705 (C=O) cm
4
-(4 -Hydroxyphenyl)butan-2-one (1)
1
–
H NMR (400 MHz, CDCl ): δ = 2.13 (s, 3H, CH ),
3 3
NaBH4 (0.46 g, 12.1 mmol) was added portionwise at 2.71 – 2.76 (m, 2H, CH ), 2.79 – 2.84 (m, 2H, CH ), 3.86 (s,
2
2
◦
0
C to a stirred solution of NiCl ·6H O (1.10 g, 4.63 mmol) 3H, CH O), 5.63 (br s, 1H, OH), 6.64 – 6.67 (m, 1H, aro-
2
2
3
in methanol (20 mL). Once the addition was complete, the matic CH), 6.68 – 6.69 (m, 1H, aromatic CH), 6.81 – 6.83 (m,
◦
suspension was stirred for 15 min at 0 C followed by re- 1H, aromatic CH) ppm. The spectral data were similar to
Brought to you by | New York University Bobst Library Technical Services
Authenticated
Download Date | 5/23/15 12:11 AM

 Bandarenko, Mikhail
Bandarenko, Mikhail
 Kovalenko, Vitaly
Kovalenko, Vitaly