P. Pourrostam-Ravadanaq, et al.
AnalyticalBiochemistry597(2020)113693
charge and hydrophobicity [46–49]. The benefits and particular prop-
erties of histidine and imidazole as pseudo-affinity ligands have made
them unique. Imidazole can be found in the structures of important
biomolecules, including histidine, which has an important role in the
location of protein binding [50–52]. Furthermore, they possess some
specific properties such as good stability, mild hydrophobicity, a wide
range of pKa values, the asymmetric carbon atom and weak charge
transfer due to the presence of imidazole moiety [46,53–57].
Herein we report the design and preparation of new adsorbents
based on sepharose for purification of immunoglobulin G from bovine
milk in one step and with high purity and efficiency. The sepharose
beads activated by 1,2-ethanediol diglycidyl ether, 1,4-butanediol di-
glycidyl ether, and epichlorohydrin in different binding capacities and
L-histidine and imidazole ligands were covalently attached to them. The
performance of modified beads and effects of the binding capacity, li-
gand type and linker length on the purification of immunoglobulin G
from bovine milk were evaluated.
2.3.1. 1,2-Ethanediol diglycidyl ether (EDDGE)
1H NMR (400 MHz, CDCl3): δ 3.59–3.71 (m, 4H, OCH2CH2),
3.77–3.80 (dd, 2H, OCH2), 3.38–3.42 (dd, 2H, OCH2), 3.13–3.16 (m,
2H, CH2(O)CH), 2.58–2.60 (dd, 2H, CH(O)CH2), 2.76–2.78 (dd, 2H, CH
(O)CH2).
2.3.2. 1,4-Butanediol diglycidyl ether (BDDGE)
1H NMR (400 MHz, CDCl3): δ 1.62–1.63 (m, 4H, –CH2-), 3.45–3.50
(m, 4H, OCH2CH2), 2.55–2.56 (dd, 2H, CH(O)CH2), 2.73–2.75 (dd, 2H,
CH(O)CH2), 3.09–3.11 (m, 2H, CH2(O)CH), 3.30–3.35 (dd, 2H, OCH2),
3.65–3.68 (dd, 2H, OCH2).
2.4. Epoxy activation of sepharose
The sepharose CL-6B was activated with epichlorohydrin, 1,2-
ethanediol diglycidyl ether, and 1,4-butanediol diglycidyl ether by
procedures as previously described elsewhere with some modifications
2. Experimental
2.4.1. Activation of sepharose with epichlorohydrin (Sep-Epch)
In a round-bottom flask equipped with a magnetic stirrer, 1 g of
washed and suction dried sepharose CL-6B was added into the aqueous
solutions of 2 mL NaOH (0.5 M, 1 M, 2 M) containing 5 mg sodium
borohydride. Epichlorohydrin (0.075, 0.15, 0.25 mL) was added to the
mixture and shaken at 45 °C for 6 h. The activated gels were washed
with an excess of distilled water and dried under vacuum conditions.
Finally, epoxy activated sepharose gels with various amounts of ep-
oxide groups (containing around 80, 120, 160 μmol of epoxide groups/
mL gel) were obtained.
2.1. Materials
Sepharose CL-6B was obtained from Arg. Biotech. Co. (Tabriz, Iran).
L-histidine (98%, Sigma-Aldrich), imidazole (99%, Sigma-Aldrich), 1,2-
ethanediol (99%, Sigma-Aldrich), 1,4-butanediol (99%, Sigma-Aldrich),
sodium borohydride (98%, Sigma-Aldrich), sodium sulfate (99%,
Sigma-Aldrich), tetrabutylammonium chloride (97%, Sigma-Aldrich),
acetic acid (99%, Sigma-Aldrich), sodium acetate (99%, Sigma-Aldrich)
and sodium carbonate (99%, Dae-Jung) were used as received without
further purifications. Sodium hydroxide (97%) and epichlorohydrin
(99%) were obtained either from Merck (Darmstadt, Germany) and
used as received. All the commercial solvents such as CH2Cl2 were
distilled before use.
2.4.2. Activation of sepharose with EDDGE and BDDGE (Sep-EDDGE and
Sep-BDDGE)
In a round-bottom flask equipped with a magnetic stirrer, 1 g of
washed and suction dried sepharose CL-6B was added into the aqueous
solution of 1 mL NaOH 1 M containing 2 mg sodium borohydride. Then
1 mL EDDGE or BDDGE was added to the reaction mixture and in-
cubated overnight on a shaking at room temperature. The activated gels
were washed with an excess of distilled water and vacuum dried.
2.2. Instrumentation
The 1H NMR spectra of synthesized compounds were recorded using
a Bruker FT-400 MHz (Bruker Co., United States) spectrometer in
deuterated chloroform (CDCl3) as solvent at room temperature. The FT-
IR spectra of synthesized compounds were recorded in/on pressed KBr
plate on a Bruker-Tensor 270 (Bruker Co., United States) spectrometer.
The morphology, microstructures and particle size distribution of the
samples were analyzed by scanning electron microscopy (SEM) and
Energy-dispersive X-ray spectroscopy (EDX) using an MIRA3 FE-SEM
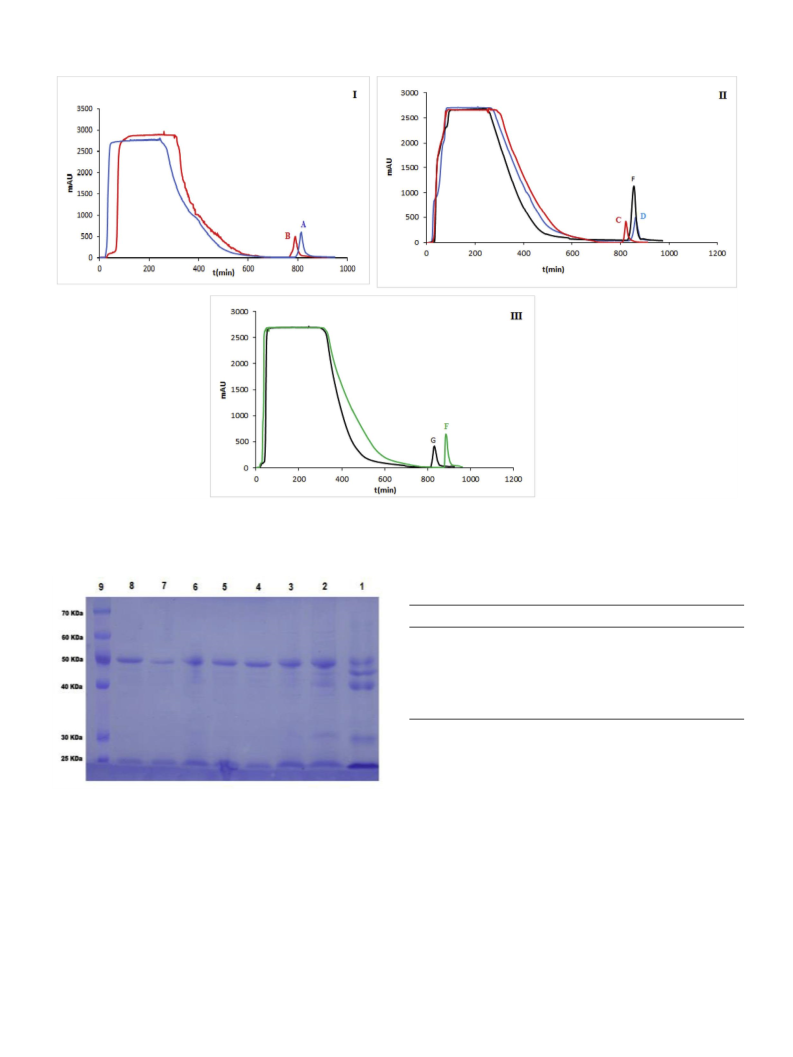
(TESCAN, Czech Republic) instrument. Fast protein liquid chromato-
graphy (GE healthcare Amersham AKTA FPLC UPC-900 P-920 INV-
907 M-925 system) was used for purification of target protein from
protein mixture. The electrophoresis analysis was carried out on an
SDS-PAGE (Sodium dodecyl sulfate-polyacrylamide gel electrophoresis)
page by a Mini-PROTEAN® 3 (Bio-Rad Co., United States) cell.
2.5. Determination of epoxy groups
The determination of epoxy groups on the activated sepharose gels
was carried out according to the method described by Sundberg and
Porath [60]. 150 mg of modified gel, was added into the 15 mL of 1.3 M
sodium thiosulfate solution and titrated with 0.1 M hydrochloric acid.
2.6. Immobilization of L-histidine and imidazole on epoxy activated
sepharose (Sep-Epch-His and Sep-Epch-Im, Sep-EDDGE-Im and Sep-
BDDGE-Im)
1 g of suction-dried epoxy activated sepharose with different epoxy
content (80, 120 and 160 μmolepoxide/mLgel), EDDGE or BDDGE acti-
vated Sepharose, was transferred to 25 mL shaking flask containing
200–500 mg L-histidine or imidazole in 2 mL sodium carbonate
(Na2CO3) 2 M and 5 mg sodium borohydride. The reaction mixture was
stirred at 50 °C for 18 h. In the end, the modified sepharose gel was
washed with plenty of distilled water and vacuum dried.
2.3. General method for synthesis of diglycidyl ether intermediates
Diglycidyl ether intermediates were prepared under phase-transfer
catalytic condition: To the vigorously stirred mixture of epi-
chlorohydrin (9.34 mL, 120 mmol), sodium hydroxide pellets (4.8 g,
120 mmol), water (0.5 mL, 28 mmol), and tetrabutylammonium
chloride (0.28 g, 1 mmol), cooled Glycol (20 mmol, 1,2-ethanediol or
1,4-butanediol) was added dropwise. After the completion of the ad-
dition, the mixture was stirred for 45 min at 40 °C. The solid products
that were formed alongside the reaction removed by filtration and
washed with CH2Cl2 (30 mL×3). The combined organic layers were
dried over anhydrous Na2SO4 and additional dichloromethane and
excess epichlorohydrin evaporated off by distillation to give a yellow
oil. The resulting oil was purified by distillation under reduced pressure
to give a pure and colorless product (89% yields).
2.7. Chromatographic separation of IgG from skim milk
For preparation of the skim milk, the fresh bovine milk was cen-
trifuged at 6000 rpm for 15 min at room temperature to remove the fat
in the form of the upper cream layer. The pH of milk was adjusted in 4.6
with HCl 1 N and centrifuged again at 6000 rpm for 15 min to pre-
cipitate the casein. Afterward, the clear skim milk was collected and the
pH raised to 7 by 1 N NaOH to protect immunoglobulins from
2

 Abbasi, Hassan
Abbasi, Hassan
 Pourrostam-Ravadanaq, Pariya
Pourrostam-Ravadanaq, Pariya
 Safa, Kazem D.
Safa, Kazem D.