3114
V. Singh et al.
LETTER
(15) Austin, K. A. B.; Banwell, M. G.; Harfoot, G. J.; Willis,
A. C. Tetrahedron Lett. 2006, 47, 7381.
(16) Enholm, E. J.; Jia, Z. J. J. Org. Chem. 1997, 62, 174.
(17) Data of Compound 14
(7) (a) Mehmoud, B.; West, F. G. Tetrahedron Lett. 2007, 48,
5051. (b) Srikrishna, A.; Gowri, V. Tetrahedron: Asymmetry
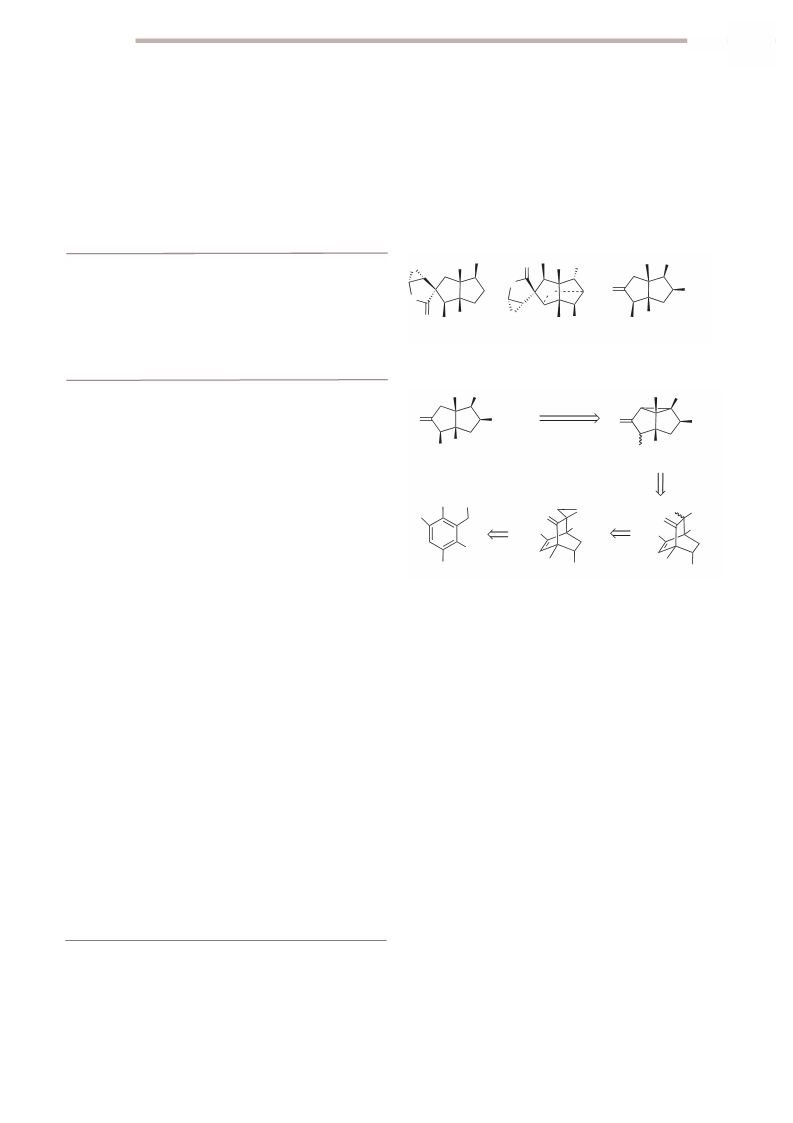
2007, 18, 1663. (c) Tzvetkov, N. T.; Neumann, B.;
Stammler, H.-G.; Mattay, J. Eur. J. Org. Chem. 2006, 351.
(d) Khan, F. A.; Rao, C. N. Tetrahedron Lett. 2006, 47,
7567. (e) Wu, Y. T.; Vidovic, D.; Magull, J.; De Meijere, A.
Eur. J. Org. Chem. 2005, 1625. (f) Srikrishna, A.; Dethe,
D. H. Org. Lett. 2003, 5, 2295. (g) Dhimane, A.; Aissa, C.;
Malacria, M. Angew. Chem. Int. Ed. 2002, 41, 3284.
(h) Shino, M.; Sato, Y.; Shisido, K. Tetrahedron Lett. 2002,
43, 5039. (i) Harrowven, D. C.; Lucas, M. C.; Howes, P. D.
Tetrahedron 2001, 57, 9157.
IR (neat): 1735 cm–1. 1H NMR (400 MHz, CDCl3): d = 3.68
(s, 3 H), 2.44–2.36 (m, 1 H), 2.32–2.15 (m, 2 H), 2.01 (d of
AB pattern, J = 19.42 Hz, 1 H), 1.92 (d of AB pattern,
J = 19.42 Hz, 1 H), 1.76–1.62 (m, 2 H), 1.09 (s, 3 H), 1.04–
0.98 (m, 9 H). 13C NMR (100 MHz, CDCl3): d = 219.0,
176.6, 53.2, 52.0, 50.7, 48.1, 45.6, 45.1, 36.4, 23.5, 21.4,
20.7, 14.2, 8.8. ESI-HRMS: m/z calcd for C14H23O3:
239.1647 [M + H]+; found: 239.1655 [M + H]+.
(8) (a) Hashimoto, H.; Abe, Y.; Mayuzumi, Y.; Karikomi, M.;
Seki, K.; Haga, K.; Uyehara, T. Tetrahedron Lett. 2002, 43,
265. (b) Solaja, B.; Huguet, J.; Karpf, M.; Dreiding, A. S.
Tetrahedron 1987, 43, 4875. (c) Huguet, J.; Karpf, M.;
Dreiding, A. S. Tetrahedron Lett. 1983, 24, 4177.
(9) (a) Singh, V. Acc. Chem. Res. 1999, 31, 324. (b) Singh, V.;
Sahu, P. K.; Singh, R. B.; Mobin, S. M. J. Org. Chem. 2007,
72, 10155. (c) Singh, V.; Chandra, G.; Mobin, S. M. Synlett
2008, 2267.
(18) Data for Hydrazone Derivative 15
IR (neat): 1731 cm–1. 1H NMR (400 MHz, CDCl3): d = 10.82
(br s, 1 H), 9.13 (d, J = 2.44 Hz, 1 H), 8.30 (dd, J1 = 8.85 Hz,
J2 = 1.83 Hz, 1 H), 7.97 (d, J = 9.47 Hz, 1 H), 3.69 (s, 3 H),
2.67–2.45 (m, 1 H), 2.41–2.31 (m, 1 H), 2.30–2.25 (m, 2 H),
2.10 (d, J = 18.32 Hz, 1 H), 1.74–1.62 (m, 2 H), 1.19 (d,
J = 6.72 Hz, 3 H), 1.10–1.09 (m, 6 H), 1.03 (s, 3 H). 13
C
NMR (100 MHz, CDCl3): d = 176.6, 167.7, 145.2, 137.8,
130.1, 129.0, 123.7, 116.5, 53.4, 53.4, 52.0, 48.3, 47.9, 45.7,
35.8, 35.4, 20.9, 20.8, 14.3, 11.1. ESI-HRMS: m/z calcd for
C20H27O6N4: 419.1931 [M + H]+; found: 419.1939 [M + H]+.
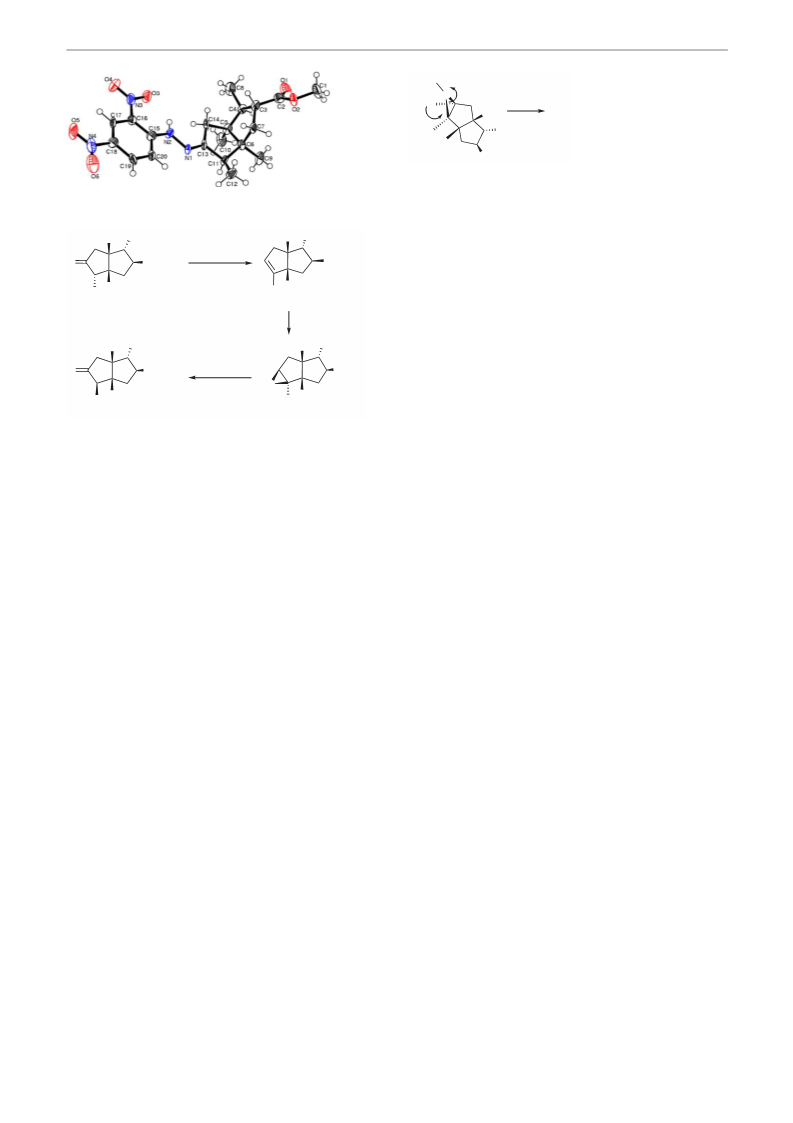
Crystal Data of Hydrazone Derivative 15
(10) Andreetti, G. D.; Böhmer, V.; Jordon, J. G.; Tabatabai, M.;
Ugozzoli, F.; Vogt, W.; Wolff, A. J. Org. Chem. 1993, 58,
4023.
(11) (a) Adler, E.; Brasen, S.; Miyake, H. Acta Chem. Scand.
1971, 25, 2055. (b) Becker, H.-D.; Bremholt, T.; Adler, E.
Tetrahedron Lett. 1972, 13, 4205. (c) Singh, V.; Porinchu,
M.; Vedantham, P.; Sahu, P. K. Org. Synth. 2005, 81, 171.
(12) All the compounds gave satisfactory spectral data.
Data of Compound 6
C20H26N4O6, M 418.45, space group, monoclinic, P 21/c,
a = 20.475 (4), b = 14.245 (2), c = 7.3712 (12) Å, a = 90,
b = 99.326 (19), g = 90.0, U = 2121.5 (6)A3, Z = 4,
Dc = 1.310 g/m3, T = 150 (2) K, F(000) = 888,
size = 0.21 × 0.18 × 0.15 mm. Reflections/collected/unique
10087/3694 [R(int) = 0.1809], final R indices [I >
2s(I)] = R1 = = 0.0732, wR2 = 0.1210 R indices (all data)
R1 = 0.2346, wR2 = 0.1622. The complete crystal data can be
obtained free of charge from The Cambridge
IR (neat): 1735 (br) cm–1. 1H NMR (400 MHz, CDCl3): d =
5.55 (br s, 1 H), 3.68 (s, 3 H), 3.06 (part of an AB system,
J = 5.86 Hz, 1 H), 2.92 (part of an AB system, J = 5.86 Hz,
1 H), 2.77 (dd, J1 = 10.26 Hz, J2 = 5.87 Hz, 1 H), 2.15 (dd,
J1 = 13.19 Hz, J2 = 10.26 Hz, 1 H), 1.87 (d, J = 1.46 Hz,
3 H), 1.74 (dd, J1 = 12.83 Hz, J2 = 5.87 Hz, 1 H), 1.26 (s, 3
H), 1.04 (s, 3 H). 13C NMR (100 MHz, CDCl3): d = 204.3,
173.8, 146.2, 124.9, 59.1, 51.9, 50.6, 49.6, 45.2, 40.9, 36.7,
17.9, 15.7, 14.4. ESI-HRMS: m/z calcd for C14H19O4:
251.1283 [M + H]+; found: 251.1277 [M + H]+.
data_request/cif quoting the CCDC number 694681.
(19) Data of Compound 18
IR (neat): 1736 (br) cm–1. 1H NMR (400 MHz, CDCl3): d =
3.70 (s, 3 H), 2.58–2.50 (dd with structure, J1 = 18 Hz, J2 = 9
Hz, 1 H), 2.41–2.34 (d with structure, J = 9 Hz, 1 H), 2.24–
2.16 (m, 2 H), 2.00–1.86 (m, 3 H), 1.03 (d, partly merged
with a s, J = 7.33 Hz, 3 H), 1.02 (s, 3 H), 1.00 (s, 3 H), 0.99
(d, partly merged with s, J = 6.72 Hz, 3 H). 13C NMR (100
MHz, CDCl3): d = 220.3, 176.3, 53.2, 51.9, 50.9, 50.6, 49.0,
47.3, 46.1, 44.2, 23.7, 18.9, 15.2, 11.9. ESI-HRMS: m/z
calcd for C14H23O3: 239.1647 [M + H]+; found: 239.1655 [M
+ H]+.
(13) (a) Demuth, M.; Hisken, W. Angew. Chem., Int. Ed. Engl.
1985, 24, 973. (b) Demuth, M.; Schaffner, K. Angew.
Chem., Int. Ed. Engl. 1982, 21, 820.
(14) (a) Zimmerman H. E., Armesto D.; Chem. Rev.; 1996, 96:
3065. (b) Armesto, D.; Ortiz, M. J.; Agarrabeitia, A. R. CRC
Handbook of Organic Photochemistry and Photobiology;
Horspool, W.; Lenci, F., Eds.; CRC: Boca Raton, 2004,
95.1. (c) Singh, V. In CRC Handbook of Organic
Photochemistry and Photobiology; Horspool, W.; Lenci, F.,
Eds.; CRC: Boca Raton, 2004, 78.1.
Synlett 2008, No. 20, 3111–3114 © Thieme Stuttgart · New York

 Singh, Vishwakarma
Singh, Vishwakarma
 Chandra, Girish
Chandra, Girish
 Mobin, Shaikh M.
Mobin, Shaikh M.