Products Categories
| CAS No.: | 87-89-8 |
|---|---|
| Name: | Inositol |
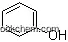
| Molecular Structure: | |
|
|
|
| Formula: | C6H12O6 |
| Molecular Weight: | 180.158 |
| Synonyms: | INOSITE; (1R,2r,3S,4R,5s,6S)-cyclohexane-1,2,3,4,5,6-hexaol; nositol; myo-Inositol; cis-1,2,3,5-trans-4,6-Cyclohexanehexol; meso-inositol; Inosital; bios I; INS; Nucite; 2-(PROPYLAMINO)-M-PROPIONOTOLUIDIDE HYDROCHLORIDE; Dambose; Mesol; Mesovit; 查看更多英文别名 收起 |
| EINECS: | 201-781-2 |
| Density: | 2.039 g/cm3 |
| Melting Point: | 222-227 °C(lit.) |
| Boiling Point: | 291.326 °C at 760 mmHg |
| Flash Point: | 143.387 °C |
| Solubility: | 4 g/100 mL (25 °C) in water |
| Appearance: | white powder |
| Safety: | 22-24/25 |
| PSA: | 121.38000 |
| LogP: | -3.83460 |
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 90221-55-92-bromo-5-methylbenzaldehyde
- 885590-99-82,3-DIFLUORO-4-IODOBENZALDEHYDE
- 97730-31-9(S)-4'-(2-Methylbutyl)Biphenyl-4-Carbonitrile
- 926293-55-26-Bromo-2-methylpyridine-3-carboxaldehyde
- 911112-05-55-Iodo-3-(trifluoromethyl)-2-pyridinamine
- 886364-50-71-(5-Bromo-2-pyridinyl)-2,2,2-trifluoroethanone

- 51548-88-0, 96039-15-5, 98974-89-1, 128110-25-8
(+/-)-1,2:4,5-di-O-isopropylidene-myo-inositol


- 87-89-8
D-myo-inositol

| Conditions | Yield |
|---|---|
| palladium dihydroxide In methanol for 88h; Heating; | 97% |


- 87-89-8
D-myo-inositol

| Conditions | Yield |
|---|---|
| palladium dihydroxide In methanol for 72h; Heating; | 96% |
| With Pd(OH)2/C In methanol for 72h; Reflux; |


- 87-89-8
D-myo-inositol

| Conditions | Yield |
|---|---|
| palladium dihydroxide In methanol for 22h; Heating; | 95% |


- 87-89-8
D-myo-inositol

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 70℃; for 3h; Deacetylation; | 94% |

A

- 87-89-8
D-myo-inositol

| Conditions | Yield |
|---|---|
| With water; trifluoroacetic acid at 65 - 70℃; | A n/a B 83% |

A

- 87-89-8
D-myo-inositol

B

- 1259936-03-2
4,6-O-methylene-myo-inositol

| Conditions | Yield |
|---|---|
| With methanol; toluene-4-sulfonic acid In chloroform at 40℃; for 48h; | A n/a B 74% |

- 15421-51-9
myo-inositol 1-phosphate


- 87-89-8
D-myo-inositol

| Conditions | Yield |
|---|---|
| With alkaline phosphatase type III from Escherichia coli In various solvent(s) at 25℃; for 16h; pH=8.0; Substitution; | 63.6% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride | |
| With sodium methylate In methanol for 2h; |

| Conditions | Yield |
|---|---|
| With water; hydrogen; palladium at 50 - 55℃; | |
| With ethanol; nickel at 125℃; under 95616 Torr; Hydrogenation; | |
| With water; palladium at 20℃; Hydrogenation.sowie bei 55grad; |

| Conditions | Yield |
|---|---|
| With hydrogen iodide | |
| With hydrogen iodide Heating; | 0.08 g |
- 87-90-1Trichloroisocyanuric acid
- 87901-60-84,4'-BIS(TRIFLUOROMETHYL)DIPHENYLMETHANE
- 879088-41-24-chloro-3-(pyridin-2-yl)aniline
- 87913-26-6N-(2-ADAMANTYL)-N-(4-BROMOPHENYL)AMINE
- 879-18-51-Naphthoyl chloride
- 879496-95-4Acai berry extract
- 879765-63-63-(2,4-dimethoxyphenyl)-1H-pyrazole-5-carboxylic acid
- 88026-65-7SYNFEROL AH EXTRA
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Specification
Inositol(CAS NO.87-89-8) is also named as 1,2,3,5-trans-4,6-Cyclohexanehexol, cis-; AI3-16111; CCRIS 6745; Dambose; Inosital; Inositene; Inositina; Inositol (VAN). It is white powder. Inositol or cyclohexane-1,2,3,4,5,6-hexol is a chemical compound with formula C6H12O6 or (-CHOH-)6, a sixfold alcohol (polyol) of cyclohexane. It is almost tasteless, with a small amount of sweetness. It is soluble in water, slightly soluble in ethanol, acetic acid, ethylene glycol and glycerin, insoluble in ether, acetone and chloroform.
Physical properties about Inositol are: (1)ACD/LogP: -3.861; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): -3.86; (4)ACD/LogD (pH 7.4): -3.86; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 1.00; (8)ACD/KOC (pH 7.4): 1.00; (9)#H bond acceptors: 6; (10)#H bond donors: 6; (11)#Freely Rotating Bonds: 6; (12)Index of Refraction: 1.784; (13)Molar Refractivity: 37.213 cm3; (14)Molar Volume: 88.375 cm3; (15)Polarizability: 14.752 10-24cm3; (16)Surface Tension: 101.601997375488 dyne/cm; (17)Density: 2.039 g/cm3; (18)Flash Point: 143.387 °C; (19)Enthalpy of Vaporization: 61.583 kJ/mol; (20)Boiling Point: 291.326 °C at 760 mmHg
Preparation of Inositol: Inositol is produced from glucose-6-phosphate (G-6-P) in two steps. First, G-6-P is isomerised by ISYNA1 to inositol 1-phosphate; then it is dephosphorylated by IMPase 1 to give free Inositol . In humans most inositol is synthesized in the kidneys, in typical amounts of a few grams per day.
Uses of Inositol: Inositol has been used as an adulterant (or cutting agent) in many illegal drugs, such as cocaine, methamphetamine, and sometimes heroin.[citation needed] It is presumed that this use is connected with one or more of the substance's properties of solubility, powdery texture, or reduced sweetness (50%) as compared with more common sugars. Inositol is also used as a stand-in for cocaine on television.
You can still convert the following datas into molecular structure:
(1)InChI=1S/C6H12O6/c7-1-2(8)4(10)6(12)5(11)3(1)9/h1-12H/t1-,2-,3-,4+,5-,6-; S27:Take off immediately all contaminated clothing.
(2)InChIKey=CDAISMWEOUEBRE-GPIVLXJGSA-N;
(3)Smiles[C@@H]1([C@@H]([C@H]([C@@H]([C@H]([C@H]1O)O)O)O)O)O
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 10gm/kg (10000mg/kg) | Toksikologicheskii Vestnik. Vol. (2), Pg. 25, 1994. | |
| rat | LD50 | intraperitoneal | > 3gm/kg (3000mg/kg) | Japanese Kokai Tokyo Koho Patents. Vol. #92-9328, | |
| rat | LD50 | intravenous | > 750mg/kg (750mg/kg) | Japanese Kokai Tokyo Koho Patents. Vol. #92-9328, |
-
Premium Related Products