Products Categories
| CAS No.: | 136470-78-5 |
|---|---|
| Name: | Abacavir |
| Article Data: | 23 |
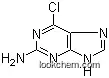
| Molecular Structure: | |
|
|
|
| Formula: | C14H18N6O |
| Molecular Weight: | 286.337 |
| Synonyms: | {(1S,4R)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]cyclopent-2-en-1-yl}methanol;Ziagen;2-Cyclopentene-1-methanol, 4-(2-amino-6-(cyclopropylamino)-9H-purin-9-yl)-, (1S-cis)-;4-Amino-5-chloro-2-ethoxy benzoic acid;Trizivir;2-Cyclopentene-1-methanol, 4-(2-amino-6-(cyclopropylamino)-9H-purin-9-yl)-, (1S,4R)-;Abacavir (Ziagen); |
| EINECS: | 620-487-9 |
| Density: | 1.7 g/cm3 |
| Melting Point: | 165° |
| Boiling Point: | 636 °C at 760 mmHg |
| Flash Point: | 338.4 °C |
| Appearance: | white powder |
| Hazard Symbols: |
 T, T, N N
|
| Risk Codes: | R21; R23/25; R43; R50/53 |
| PSA: | 101.88000 |
| LogP: | 1.74650 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 136522-33-3
(1S,4R)-4-[2-amino-6-chloro-9H-purin-9-yl]-2-cyclopentene-1-methanol

- 765-30-0
Cyclopropylamine

- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| In ethanol at 70℃; | 98% |
| In ethanol Heating; | 72% |
| Stage #1: (1S,4R)-4-[2-amino-6-chloro-9H-purin-9-yl]-2-cyclopentene-1-methanol; Cyclopropylamine In butan-1-ol at 40 - 70℃; for 5h; Stage #2: With sodium hydrogencarbonate In butan-1-ol at 20 - 25℃; for 1h; Product distribution / selectivity; | |
| In butan-1-ol at 45 - 90℃; for 2h; Product distribution / selectivity; | |
| With sodium hydrogencarbonate In ethanol; chloroform | 0.43 g (80%) |
- 765-30-0
Cyclopropylamine

- 172015-79-1
(1S,cis)-4-(2-amino-6-chloro-9-H-purin-9-yl)-2-cyclopentene-1-methanol hydrochloride

- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| In methanol at 70℃; for 12h; Substitution; | 90% |
| Stage #1: (1S,cis)-4-(2-amino-6-chloro-9-H-purin-9-yl)-2-cyclopentene-1-methanol hydrochloride With triethylamine In water Stage #2: Cyclopropylamine In water at 70 - 75℃; Concentration; Solvent; Temperature; | 90% |
| In isopropyl alcohol at 90 - 95℃; for 12h; sealed reactor; | |
| In ethanol at 25 - 78℃; for 6h; | |
| With sodium carbonate In ethanol at 70 - 80℃; | 25 g |
- 136470-88-7
(-)-N-{6-(cyclopropylamino)-9-[(1R,4S)-4-(hydroxymethyl)cyclopent-2-enyl]-9H-purin-2-yl}isobutyramide

- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| Stage #1: (-)-N-{6-(cyclopropylamino)-9-[(1R,4S)-4-(hydroxymethyl)cyclopent-2-enyl]-9H-purin-2-yl}isobutyramide With isopropyl alcohol; sodium hydroxide for 1h; Reflux; Stage #2: With hydrogenchloride In water; isopropyl alcohol at 20 - 25℃; pH=7.0 - 7.5; Product distribution / selectivity; | 90% |
| With sodium hydroxide; water; isopropyl alcohol for 1h; Heating / reflux; | 77% |
| With sodium hydroxide; water; isopropyl alcohol for 1h; Product distribution / selectivity; Heating / reflux; | 43% |
- 7440-44-0
pyrographite

- 765-30-0
Cyclopropylamine

- 172015-79-1
(1S,cis)-4-(2-amino-6-chloro-9-H-purin-9-yl)-2-cyclopentene-1-methanol hydrochloride

- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| With sodium hydroxide In acetone | 90% |

- 1067882-78-3
6-cyclopropylamine-9-((1R,4S)-4-((triisopropylsilyloxy)methyl)cyclopent-2-enyl)-9H-purin-2-amine

- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; water In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; | 89% |

- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; ethyl acetate at 25 - 45℃; pH=6 - 6.1; | 78% |
- 1360538-03-9
((1S,4R)-4-(6-(cyclopropylamino)-2-((4-methoxybenzyl)amino)-9H-purin-9-yl)cyclopent-2-en-1-yl)methanol

- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In chloroform at 50℃; for 72h; Inert atmosphere; | 73% |

- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 60℃; for 60h; Schlenk technique; | 70% |
- 1108600-46-9
((1S,4R)-4-(2-chloro-6-(cyclopropylamino)-9H-purin-9-yl)cyclopent-2-en-1-yl)methanol

- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; copper(l) iodide In ethanol at 150℃; for 40h; Schlenk technique; Sealed tube; | 63% |
| Stage #1: ((1S,4R)-4-(2-chloro-6-(cyclopropylamino)-9H-purin-9-yl)cyclopent-2-en-1-yl)methanol With hydrazine In methanol; water at 50℃; Stage #2: With acetic acid; sodium nitrite In water at 0℃; for 1h; Stage #3: With tin(ll) chloride In ethanol for 2h; Product distribution / selectivity; Heating / reflux; | |
| Stage #1: ((1S,4R)-4-(2-chloro-6-(cyclopropylamino)-9H-purin-9-yl)cyclopent-2-en-1-yl)methanol With hydrazine In methanol at 50℃; Stage #2: With acetic acid; sodium nitrite In water at 0℃; for 1h; Stage #3: With ammonia; triphenylphosphine In 1,4-dioxane; water for 5h; Product distribution / selectivity; Heating / reflux; | |
| Multi-step reaction with 2 steps 1: dimethyl sulfoxide / 150 °C 2: trifluoroacetic acid / chloroform / 72 h / 50 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: hydrazine hydrate / methanol / 50 °C 2: acetic acid; sodium nitrite / water / 1 h / Cooling with ice 3: stannous chloride dihydrate / ethanol / 2 h / Reflux View Scheme |
- 765-30-0
Cyclopropylamine

- 171887-04-0
(1R,4S)-1-[(2-amino-6-chloro-5-formamido-4-pyrimidinyl)amino]-4-(hydroxymethyl)-2-cyclopentene

A
- 141271-12-7
(1S, 4R)-(4-(2,5-diamino-6-chloro-4-pyrimidinyl)amino)-2-cyclopenten-1-methanol

B
- 136470-78-5
abacavir

| Conditions | Yield |
|---|---|
| In butan-1-ol at 130℃; for 21h; Substitution; hydrolysis; | A 16% B 60% |

Specification
1. Introduction of Abacavir
Abacavir is one kind of white powder. Its IUPAC name is called [(1S,4R)-4-[2-amino-6-(cyclopropylamino)purin-9-yl]cyclopent-2-en-1-yl]methanol. It belongs to classification codes which are Anti-HIV Agents, Anti-Infective Agents, Anti-Retroviral Agents, Antiviral Agents, Enzyme Inhibitors, Nucleic Acid Synthesis Inhibitors, Reverse Transcriptase Inhibitors. Besides, its Product Categories is Bases & Related Reagents;Inhibitors;Intermediates & Fine Chemicals;Nucleotides;Pharmaceuticals. It should be store at refrigerator. Abacavir can be soluble in water. Its Classification Codes is Anti-HIV Agents; Anti-Infective Agents; Anti-Retroviral Agents; Antiviral Agents; Enzyme Inhibitors; Nucleic Acid Synthesis Inhibitors; Reverse Transcriptase Inhibitors.
2. Properties of Abacavir
Physical properties about Abacavir are:
(1) ACD/LogP: -0.78 ; (2) # of Rule of 5 Violations: 0 ; (3) ACD/LogD (pH 7.4): 0.66 ; (4) ACD/BCF (pH 5.5): 1 ; (5) ACD/BCF (pH 7.4): 1.8 ; (6) ACD/KOC (pH 5.5): 4.47 ; (7) ACD/KOC (pH 7.4): 50.77 ; (8) #H bond acceptors: 7 ; (9) #H bond donors: 4 ; (10) #Freely Rotating Bonds: 4 ; (11) Polar Surface Area: 59.31 Å2 ; (12) Index of Refraction: 1.864 ; (13) Molar Refractivity: 75.8 cm3 ; (14) Molar Volume: 167.6 cm3 ; (15) Surface Tension: 80.1 dyne/cm ; (16) Density: 1.7 g/cm3 ; (17) Flash Point: 338.4 °C ; (18) Enthalpy of Vaporization: 98.77 kJ/mol ; (19) Boiling Point: 636 °C at 760 mmHg ; (20) Vapour Pressure: 4.77E-17 mmHg at 25°C.
3. Structure Descriptors of Abacavir
(1) SMILES:n3c1c(ncn1[C@H]2/C=C\[C@@H](CO)C2)c(nc3N)NC4CC4;
(2) InChI:InChI=1/C14H18N6O/c15-14-18-12(17-9-2-3-9)11-13(19-14)20(7-16-11)10-4-1-8(5-10)6-21/h1,4,7-10,21H,2-3,5-6H2,(H3,15,17,18,19)/t8-,10+/m1/s1;
(3) InChIKey:MCGSCOLBFJQGHM-SCZZXKLOBP
4. Uses of Abacavir
Abacavir (ABC) is a powerful nucleoside analog reverse transcriptase inhibitor (NRTI) used to treat HIV and AIDS. This compound has been used as a drug with the trade name of Ziagen. It is a kind of antiretroviral drugs approved by the Food and Drug Administration (FDA). It has tablets and oral solution. This drug is used to treat HIV and AIDS in combination with other antiretroviral agents. It can be used for research and manufacture Epzicom tablet.
5. Production of Abacavir
030-8 1.0g (0.0053mol), in the reaction flask was added cesium carbonate 1.75 g (0.0054 mol) and dry DMSO 50ml, stirred under N2 protection, the temperature was raised to 60 °C and stirred at this temperature for 2 h the mixture wascooled to room temperature, then add tetrakis (triphenylphosphine) combined palladium (TTP) [0.85 (0.00074mol)] and compound 030-5 [0.79g (0.0034 mol), DMSO (10 ml) solution was stirred and heated to 65 °C held 65 °C and stirred reaction 2.25h. The you can get the mixture containing compounds 030-9.
To the mixture was added methanol 100ml and K2CO3 is 2.10g, the mixture reaction was stirred for 45min at 40 °C, a solid precipitate which was filtered through a Celite layer and the filtrate was evaporated to a small volume under vacuum at 90 °C, and the remaining gum pounding mill was extracted with dichloromethane (100ml * 2) to give a brown solid residue was purified by silica gel (Merck 9385) column chromatography [eluent: dichloromethane / methanol (volume ratio 9:1)] to give a yellow foam was 030 0.26 g, yield 26.8.