Products Categories
| CAS No.: | 7440-44-0 |
|---|---|
| Name: | Carbon |
| Article Data: | 1437 |
| Molecular Structure: | |
|
|
|
| Formula: | C |
| Molecular Weight: | 12.011 |
| Synonyms: | 207A (carbon);BAC-MP;BAC-MQ 50801;BACM;BACM (carbon);BAU(carbon);BAU 1;BAU 1 (carbon);BAU-MF;BAX 1100;BAX 1500;BAX 950;BC 120;BC 51 (carbon);BC 53;BG 20-1;BG 6080;BGWJX 108;BP 20;BP 20(carbon);BP 25;BP 25 (carbon);BPL (adsorbent);BPL 80X270;BPL-F 3;BSP10AS;BU 200;BU 201;BU 202;BU 203;BX-Super;BX-Ultra;Barnebey Sutcliffe4707;Baytube 150P;Baytubes C 150HP;Baytubes C 150P;Baytubes CNT-WFA 147;Baytubes DP-HP;Bellfine;Bellfine AB;Bellfine AP;Bellfine BG;Bellfine MG;Bintyotan;Biolite (carbon film);BuckyPearls;Buckytubes;Buttuber;C 150HP;C150P;C 2000S;CA 10;CA 10 (carbon);CAR 95;CB 10-1000;CB 3-1000;CB30-1000;CC 1230;CC 521G;CC 602;CCE 01PA;CECA SA 1763;CF 17;CF 32-1;CF 8;CF 8 (carbon);CFF 30;CFOAM 17;CFOAM 25;CG 10;CG 1040;CG 48B;CG 5;CG 80X325;CGF 1-3/100;CHR 98-55;CHSH 2;CL-K;CLAP 8333;CLF II;CM 95;CMB200;Charcoal active;207E3;20SPD;2C98;340F;3GX;4GCX;4GM;601D;601D(carbon);7144EL;7GX;AC 01;AC 01 (adsorbent);AC 100;AC 100(adsorbent);AC 40 (adsorbent);ACP-W;AG 2 (catalyst support);AG2-4;AG 3 (adsorbent);AG 5;AG 5 (adsorbent);AG 95;AG 95 (carbon);AG-M;AG-M (carbon);AG-OV 1;AGN 1 (carbon);AGN 2 (carbon);AGN3;AGS 3;AGS 4;AGS 4 (adsorbent);AK;AK (adsorbent);AMI 200H;AP 4-60;APB10C;APF;APF film;AR 2;AR 2 (carbon);AR 3;AR 3 (carbon);AR-A;AR-A(carbon);AR-B;ARD;ARD (carbon);ART 2;ART 2 (carbon);ART 2-302;AS-O;AS-O(adsorbent);AT 15;AT 15 (carbon);AT 2C;AT 3CM;ATE;ATI-MWNT 001;AU;AU(adsorbent);AU 1;AU 10;AU 10Mg;AU 11;AU 14;AU 15;AU 3;AU 42;AU 47;AU63;AU 65;AU 66;AU 69;AU 7;AU 8;AUVM;AX 21;AX 21 (carbon);Acticarbon25K;Acticarbon ENO;Acticarbon TK;Actitex CS 1501;Actitex SC 1501;ActitexWWP 3;Activated carbon;Active carbon beads;Advanced Patterning Film;AldrichG 60;Alfa Aesar 41774;Amoco PX 21;Anthrasorb;AquaGuard;AquaSorb 202;BAC;BAC-G 70R;BAC-LP; |
| EINECS: | 231-153-3 |
| Density: | ~1.7 g/mL at 25 °C(lit.) |
| Melting Point: | 3550 °C(lit.) |
| Boiling Point: | 500-600 °C(lit.) |
| Flash Point: | >230 °F |
| Solubility: | Insoluble in water. |
| Appearance: | grey solid |
| Hazard Symbols: | R36/37/38:; |
| Risk Codes: | Xi:Irritant; ">  Xi:Irritant; Xi:Irritant; |
| Safety: | 22-24/25 |
| Transport Information: | UN 1361/1362 |
| PSA: | 0.00000 |
| LogP: | 0.00000 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 90221-55-92-bromo-5-methylbenzaldehyde
- 885590-99-82,3-DIFLUORO-4-IODOBENZALDEHYDE
- 97730-31-9(S)-4'-(2-Methylbutyl)Biphenyl-4-Carbonitrile

| Conditions | Yield |
|---|---|
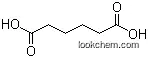
| With nitrogen In neat (no solvent) heating at 600°C for 2.5 h; color change from grey to black;; | A 100% B 100% |
| With nitrogen In neat (no solvent) heating at 600°C for 30 min; color change from grey to black;; | A 99.1% B 99.1% |
| With nitrogen In neat (no solvent) heating at 400°C for 5 h; color change from grey to black;; | A 51.8% B 51.8% |
| With nitrogen In neat (no solvent) heating at 400°C for 30 min; color change from grey to black;; | A 32.8% B 32.8% |
| In neat (no solvent) decompn.;; |

| Conditions | Yield |
|---|---|
| thermal decomposition of Mg2{Fe(CN)6} at 800 °C;; | A n/a B n/a C 99.67% D n/a E n/a |
| thermal decomposition of Mg2{Fe(CN)6} at 700 °C;; | A n/a B 0.3% C 91.85% D n/a E n/a |
| thermal decomposition of Mg2{Fe(CN)6} at 600 °C;; | A n/a B 1.03% C 62.54% D n/a E n/a |


- 7440-44-0
pyrographite

| Conditions | Yield |
|---|---|
| at 600℃; for 6.5h; Inert atmosphere; | 66% |
- 563-63-3
silver(I) acetate

A
- 124-38-9
carbon dioxide

B
- 7440-22-4
silver

C
- 7440-44-0
pyrographite

D
- 64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| In neat (no solvent) thermic decompn.;; | A 4.09-8.42 B 64.61% C 1.21% D 26-27 |
| In neat (no solvent) thermic decompn. on heating in an open tube or a covered porcelain-vessel;; |

- 34557-54-5
methane

- 7440-44-0
pyrographite

| Conditions | Yield |
|---|---|
| nickel In neat (no solvent, gas phase) byproducts: H2, C2-hydrocarbons; other Radiation; natural gas converted in flow-through reactor, catalyst heated (300-650 °C) with microwave generator; gas chromy.; | 62.9% |
| iron In neat (no solvent, gas phase) byproducts: H2, C2-hydrocarbons; other Radiation; natural gas converted in flow-through reactor, catalyst heated (300-650 °C) with microwave generator; gas chromy.; | 46.7% |
| molybdenum In neat (no solvent, gas phase) byproducts: H2, C2-hydrocarbons; other Radiation; natural gas converted in flow-through reactor, catalyst heated (300-650 °C) with microwave generator; gas chromy.; | 35.9% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) start of reaction at 550.degree C.; max. formation of Mg2C3 at 680.degree C. and MgC2 at 660.degree C.;; | A 57% B 0.15% C n/a |
| In neat (no solvent) start of reaction at 550.degree C.; max. formation of Mg2C3 at 680.degree C. and MgC2 at 660.degree C.;; | A 57% B 0.15% C n/a |


| Conditions | Yield |
|---|---|
| With thiophene; tungsten In gaseous matrix pyrolysis (750°C), fibre growth; | 55.4% |
| With thiophene; titanium In gaseous matrix pyrolysis (775°C), fibre growth; | 54% |
| With thiophene; manganese In gaseous matrix pyrolysis (700°C), fibre growth; | 52.4% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) decomposition at 390°C, formation of acetone, Na2CO3 and traces of C between 410 and 450°C while distilling;; | A n/a B <1 C 53% |
| In neat (no solvent) decomposition at 390°C, formation of acetone, Na2CO3 and traces of C between 410 and 450°C while distilling;; | A n/a B <1 C 53% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) at 1000°C;; | A n/a B n/a C 46% |

- 1592-23-0Calcium stearate
- 151767-02-1Montelukast sodium
- 123-86-4Butyl acetate
- 25068-38-6Phenol, 4,4'-(1-methylethylidene)bis-, polymer with (chloromethyl)oxirane
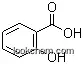
- 65-85-0Benzoic acid
- 25316-40-9Doxorubicin hydrochloride
- 7173-51-5Didecyl dimethyl ammonium chloride
- 1344-09-8Sodium silicate
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Chemistry
Molecular Structure of Carbon (CAS NO.7440-44-0):
.gif)
Empirical Formula: C
Molecular Weight: 12.0107
H bond acceptors: 0
H bond donors: 0
Freely Rotating Bonds: 0
Polar Surface Area: 0 Å2
Enthalpy of Vaporization: 11.28 kJ/mol
Vapour Pressure: 205000 mmHg at 25 °C
InChI: InChI=1/C
Smiles: C
Classification Code: Mutation data; Tumor data
Density (near r.t.): amorphous: 1.8–2.1 g/cm3
Density (near r.t.): graphite: 2.267 g/cm3
Density (near r.t.): diamond: 3.515 g/cm3
Sublimation point: 3915 K, 3642 °C, 6588 °F
Triple point: 4600 K (4327°C), 10800 kPa
EINECS: 231-153-3
Product Categories: Industrial / Fine Chemicals; Inorganics; Others; Organic Chemicals; Pharmacopoeia (USP); Pharmacopoeia A-ZPharmacopoeia (USP); Pharmacopoeial Organics; Activated CarbonAir Monitoring; Bulk Adsorbents; General Purpose Adsorbents (silica gel&activated charcoal); Packed GC; Packings, Uncoated
History
Carbon (CAS NO.7440-44-0) was discovered in prehistory and was known in the forms of soot and charcoal to the earliest human civilizations. Diamonds were known probably as early as 2500 BCE in China, In 1722, René A. F. de Réaumur demonstrated that iron was transformed into steel through the absorption of some substance, now known to be carbon. In 1786, the French scientists Claude Louis Berthollet, Gaspard Monge and C. A. Vandermonde then showed that this substance was carbon. A new allotrope of carbon, fullerene, that was discovered in 1985 includes nanostructured forms such as buckyballs and nanotubes.Their discoverers (Curl, Kroto, and Smalley) received the Nobel Prize in Chemistry in 1996.
Uses
Carbon (CAS NO.7440-44-0) black is used as the black pigment in printing ink, artist's oil paint and water colours, carbon paper, automotive finishes, India ink and laser printer toner. Carbon black is also used as a filler in rubber products such as tyres and in plastic compounds. It is used in chemical reduction at high temperatures. Carbides of silicon, tungsten, boron and titanium, are among the hardest known materials, and are used as abrasives in cutting and grinding tools. Carbon compounds make up most of the materials used in clothing, such as natural and synthetic textiles and leather, and almost all of the interior surfaces in the built environment other than glass, stone and metal.
Production
Commercially viable natural deposits of graphite occur in many parts of the world, but the most important sources economically are in China, India, Brazil, and North Korea.
According to the USGS, U.S. (synthetic) graphite electrode production in 2006 was 132,000 tons valued at $495 million and in 2005 was 146,000 tons valued at $391 million, and high-modulus graphite (carbon) fiber production in 2006 was 8,160 tons valued at $172 million and in 2005 was 7,020 tons valued at $134 million.
Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| dog | LD | intraperitoneal | > 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 34, Pg. 416, 1992. | |
| dog | LD | oral | > 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 34, Pg. 416, 1992. | |
| dog | LD | subcutaneous | > 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 34, Pg. 416, 1992. | |
| mouse | LD | intraperitoneal | > 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 34, Pg. 416, 1992. | |
| mouse | LD | oral | > 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 34, Pg. 416, 1992. | |
| mouse | LD | subcutaneous | > 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 34, Pg. 416, 1992. | |
| mouse | LD50 | intravenous | 440mg/kg (440mg/kg) | Toxicology and Applied Pharmacology. Vol. 24, Pg. 497, 1973. | |
| rat | LD | intraperitoneal | > 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 34, Pg. 416, 1992. | |
| rat | LD | oral | > 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 34, Pg. 416, 1992. | |
| rat | LD | subcutaneous | > 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 34, Pg. 416, 1992. |
Consensus Reports
Carbon is reported in EPA TSCA Inventory.
Safety Profile
Safety Description: S22;S24/25
S22:Do not breathe dust.
S24/25:Avoid contact with skin and eyes.
Transport Information: UN 1361/1362
Standards and Recommendations
OSHA PEL: (Natural graphite) TWA 2.5 mg/m3; (Synthetic graphite) TWA Total Dust: 10 mg/m3; Respirable Fraction: 5 mg/m3
ACGIH TLV: TWA 2 mg/m3 (respirable dust)
DFG MAK: 1.5 mg/m3
DOT Classification: 4.2; Label: Spontaneously Combustible
Specification
Carbon , with CAS number of 7440-44-0, can be called AX 21 (carbon) ; Acticarbon25K ; Acticarbon ENO ; Acticarbon TK ; Actitex CS 1501 ; Actitex SC 1501 ; ActitexWWP 3 ; Activated carbon ; Active carbon beads ; Advanced Patterning Film ; Alfa Aesar 41774 ; BAC-MQ 50801 ; Barnebey Sutcliffe4707 ; Baytubes DP-HP ; Charcoal active . Carbon (CAS NO.7440-44-0) is the chemical element with symbol C and atomic number 6. As a member of group 14 on the periodic table, it is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.All forms of carbon are highly stable, requiring high temperature to react even with oxygen. Carbon may burn vigorously and brightly in the presence of air at high temperatures, as in the Windscale fire, which was caused by sudden release of stored Wigner energy in the graphite core. Large accumulations of coal, which have remained inert for hundreds of millions of years in the absence of oxygen, may spontaneously combust when exposed to air, for example in coal mine waste tips.