S. Seok et al. / Ultrasonics Sonochemistry 28 (2016) 178–184
183
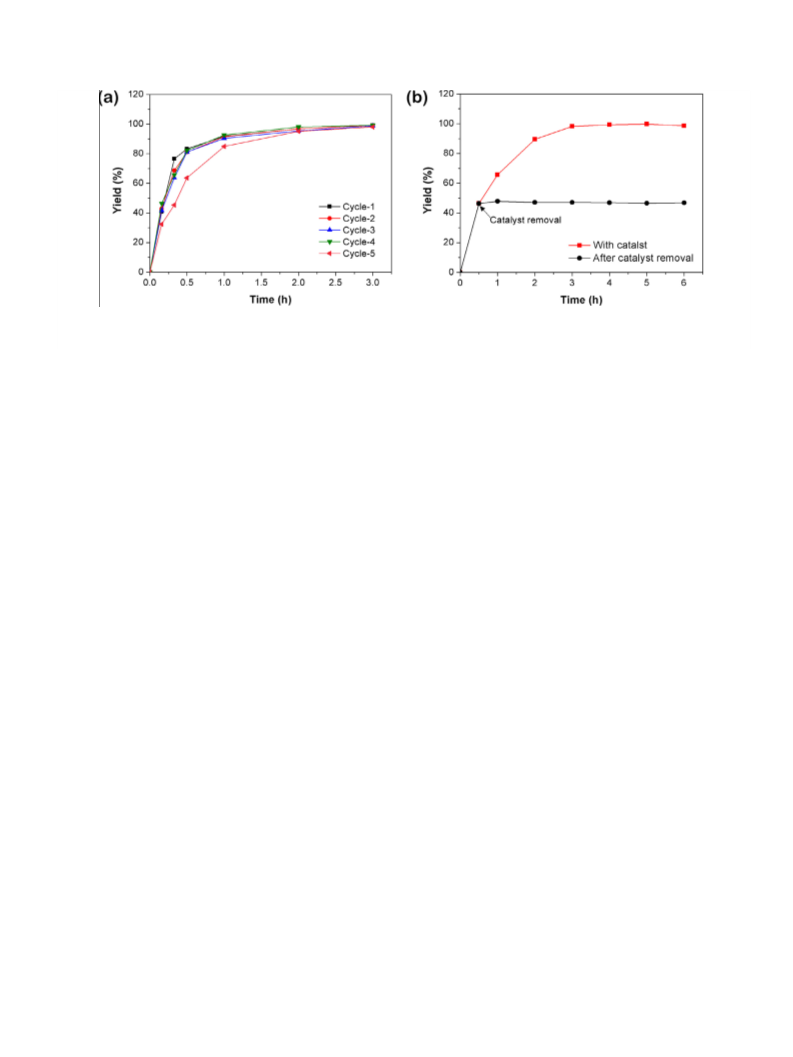
Fig. 5. Cinnamylalcohol oxidation yield of (a) PdO@SNP for five cycles and (b) with catalyst (red) or without catalyst after about 50% yields (black). Reaction condition:
cinnamylalcohol (0.2 mmol), PdO@SNP (40 mg, Pd = 3.3 mol% of substrate), toluene (2 mL), 90 °C, O (1 atm). (For interpretation of the references to color in this figure legend,
2
the reader is referred to the web version of this article.)
3.2. Selective alcohol oxidation reaction
the PdO@SNP catalyst is essential to carry out the oxidation reac-
tion (Table 1: entry 14).
PdO@SNP prepared by sonochemical method was applied for
The scope of the PdO@SNP for primary and secondary benzylic
and aliphatic alcohols was explored under the pre-defined reaction
conditions. Table 2 shows the catalytic activity and selectivity for
the aerobic oxidation reactions of variety of alcohols to aldehydes
and ketones. The benzyl alcohol tolerated the PdO@SNP comfort-
ably to give benzaldehyde with high yield and selectivity
(Table 2: entry 2). A small amount of naphthalene and benzene
emerged in the reaction due to the decarbonylation process. This
phenomenon occurs frequently for Pd based catalysts [27].
Benzylalcohol bearing electron donating and electron with draw-
ing groups was converted into aldehydes with very high selectivity
(Table 2: entries 3 and 4). Secondary benzyl alcohols (Table 2:
entries 5, 12 and 13) were also selectively catalyzed by the
PdO@SNP. The electron with drawing group strongly affected the
reaction rate and reaction time was longer for 4-chlorobenzyl alco-
hol and 4-chlorobenzhydrol (Table 2: entries 4 and 13). The
2-thiopehenmethanol containing the heteroatom and aliphatic
2-octanol afforded the products in good yield and selectivity
(Table 2: entries 5–7). Although the conversion for geraniol was
a little low, the cinnamyl alcohol and trans-crotonyl alcohol pro-
duced aldehydes in high selectivity without double bond iso-
merism (Table 2: entries 8–10). PdO@SNP comfortably catalyzed
the selective aerobic oxidation of alcohols. The oxidation reaction
of 2-naphthylmethanol was carried out as model reaction as
shown in Scheme 1. The Initial reaction was carried out at room
temperature in the toluene under atmospheric pressure of dioxy-
gen without any refluxing agent as shown in Table 1.
In this reaction a small amount of 2-naphthylmethanol con-
verted to 2-naphthaldehyde with the naphthalene (Table 1: entry
). The reaction rate and selectivity of the 2-naphthaldehyde
increased as we increased the temperature from 25 °C to 50 °C
and then to 90 °C (Table 1: entry 2, 3). A nearly complete conver-
sion of 2-naphthylmethanol was obtained when the reaction was
carried out over the period of 10 h at 90 °C with PdO@SNP
1
(
40 mg, Pd content = 3.3 mol% of substrate) catalyst under an oxy-
gen atmosphere (Table 1: entry 3). The conversion and selectivity
almost equalized when a similar reaction was carried out in a fairly
large amount of the catalyst (100 mg, Pd content = 8.3 mol% of sub-
strate), (Table 1: entry 5). However reaction did not proceed well in
a small amount of the catalyst (20 mg, Pd content = 1.6 mol% of
substrate) under the similar reaction condition (Table 1: entry 4).
Therefore, the further experiments were carried out with
PdO@SNP catalyst (40 mg, Pd content = 3.3 mol% of substrate). At
the time this reaction was carried out in different solvents; conver-
sion and selectivity were not as good as using of toluene (Table 1:
entries 6–9). The reaction proceeded quickly and a complete con-
version was obtained with biphasic solvent of water and toluene,
but the selectivity for 2-naphthaldehyde proved to be quite low
the
sterically
hindered
cyclic
2-adamantanol
into
2-adamantanone without giving any by product (Table 2: entry
11).
The catalyst is totally heterogeneous and can be separated from
the reaction mixture by centrifugation process. Upon being iso-
lated, the catalyst could endure five turns for cinnamylalcohol oxi-
dation, while the catalytic reactivity was not changed (Fig. 5a).
Catalyst is robust enough that no leached product was found in
the mixture by ICP-AES analysis, when catalyst was removed after
about 50% conversion of cinnamylalcohol. The reaction ceased
completely, once the catalyst removed from the mixture, preclud-
ing the further progress of the reaction (Fig. 5b).
(
i.e. ꢀ72%, Table 1: entry 10). To show the synergetic effect of
PdO with SNP support, we performed three control reactions using
Pd(NO O, SNP and PdO catalysts. The reaction hardly pro-
ÁxH
ceeded with precursor Pd(NO O or support SNP particles
ÁxH
Table 1: entries 11 and 12). However, an appreciable amount of
3
)
2
2
)
3 2
2
(
2
2
-naphthaldehyde was obtained upon the catalytic reaction of
-naphthylmthanol with PdO under the similar reaction conditions
(
Table 1: entry 13). These results ensured that the PdO is the active
site in this catalytic reaction. The conversion and selectivity of
-naphthylmethanol oxidation was obviously higher for
4
. Conclusions
2
PdO@SNP than PdO, which explained the significance that SNP
served as a significant support material and thus exhibited the
capacity to that provided PdO with a large dispersion area.
Hence, SNP enhanced the availability of active sites of the PdO
NPs for catalytic reaction. Oxidation of 2-naphthylmethanol did
not take place in the absence of the catalyst, which shows that
We successfully employed a sonochemical method for the syn-
thesis of PdO-doped SNP nanocomposite. The ultrasonic irradiation
method provided advantages of fast reaction time and mild reac-
tion condition without any surface modification. By adopting silica
as support material, we prevented PdO NP aggregation and gener-
ated well-dispersed PdO NP on surface of SNP. Fabricated

 Seok, Seunghwan
Seok, Seunghwan
 Hussain, Muhammad Asif
Hussain, Muhammad Asif
 Park, Kyun Joo
Park, Kyun Joo
 Kim, Jung Won
Kim, Jung Won
 Kim, Do Hyun
Kim, Do Hyun