V. F. Moreno, G. C. dos Santos, G. M. G. da Costa, M. H. A. Gomes, and L. C. d. Silva-Filho
Vol 000
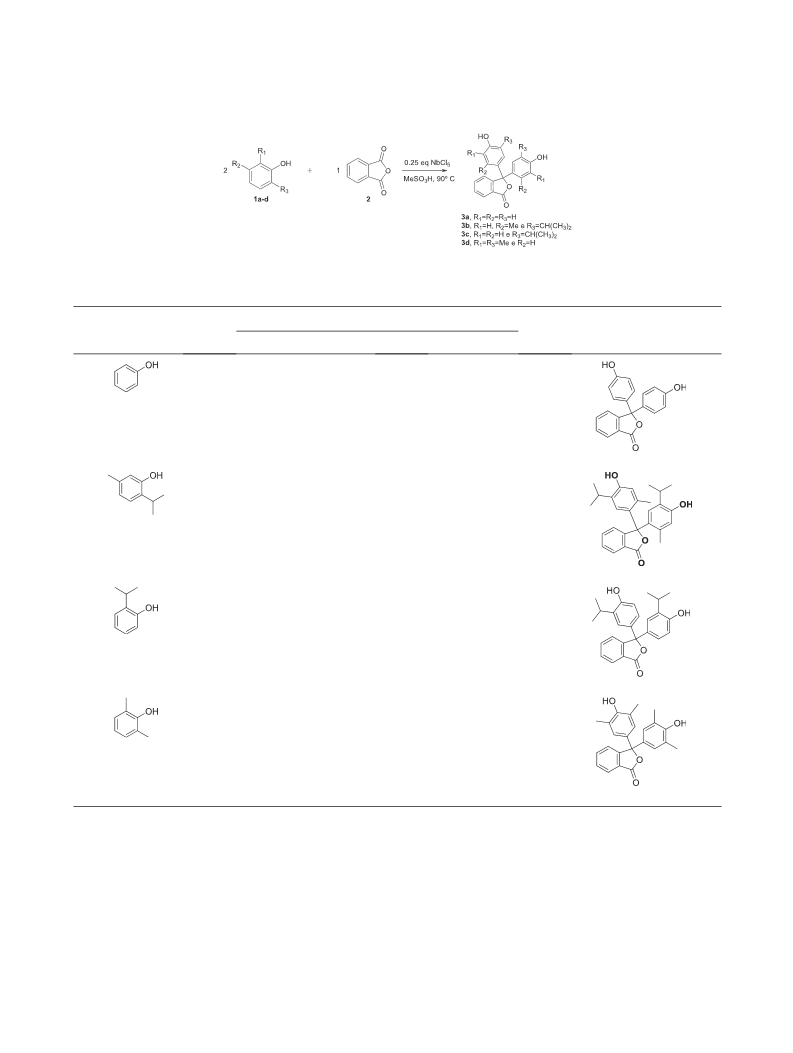
comes from compound 3a that presents a difference of
1 kJ/mol. Compounds 3b and 3d show a difference
of approximately 37 kJ/mol. Compound 3c has an energy
difference of approximately 42 kJ/mol.
In Figure 5a, we can see the experimental and theoretical
spectrum of the phthalein derivatives. As shown here, the
solution. The presence of alkyl groups stabilizes the
5
negative charge of the anionic form of the dye,
increasing the intensity of absorption in basic media.
The presence of these alkyl groups in some cases can
shift the absorption lengths of these derivatives
bathochromically. Theoretical calculations using B3LYP
functional helped us to show the differences in energy
of the three different forms of each compound. In this
study, we observed the greater stability of the lactonic
and zwitterionic forms in relation to the quinoidal. This
behavior was also seen in the MOs.
lactonic and zwitterionic forms exhibit
a similar
absorption spectrum with two bands, the first, intense, at
approximately 250 nm and the second with lower
intensity close to 320 nm. In the quinoidal form, we also
have two absorption bands, the first near 340 nm and the
second around 430 nm. The shape of the experimental
curve, as well as the shift in the spectra, shows that
in the conditions measured in ethanol solution, we
have the lactonic and zwitterionic forms. However, in
the experimental spectrum of compounds 3b and 3d, we
see a band of low intensity above 400 nm. This band
suggests that these compounds have a small amount in
quinoidal form. This is proven by the more intense color
of these compounds in ethanol. Another factor that
justifies the presence of the quinoidal form is the small
energy barrier of 3b and 3d that exists between the forms
Acknowledgments. The authors would like to thank Fundação de
Amparo à Pesquisa do Estado de São Paulo (FAPESP) (procs.
2
7
012/24199-8, 2015/00615-0, 2016/01599-1, and 2018/14506-
), Conselho Nacional de Desenvolvimento Científico
e
Tecnológico (CNPq) (proc. 302769/2018-8) and Pró-Reitoria de
Pesquisa (PROPe-UNESP) for their financial support. We
would also like to thank Companhia Brasileira de Mineralogia e
5
Mineração (CBMM) for the NbCl samples.
REFERENCES AND NOTES
(37 kJ/mol). As already mentioned earlier, this is due to
the fact that these compounds have relatively bulky
substituents, which may, by steric hindrance, favor the
quinoidal form.
[1] (a) Henrique Arpini, B.; de Andrade Bartolomeu, A.;
Andrade, K. Z.; Carlos da Silva-Filho, L.; Lacerda, V. Curr Org Synth
2015, 12(5), 570. (b) Dos Santos, G. C.; de Andrade Bartolomeu, A.;
Ximenes, V. F.; da Silva-Filho, L. C. J Fluoresc 2017, 27, 271. (c)
Martins, L. M.; de Faria Vieira, S.; Baldacim, G. B.; Bregadiolli, B.
A.; Caraschi, J. C.; Batagin-Neto, A.; da Silva-Filho, L. C. Dyes Pigm
In the molecular orbitals (MOs), we observed the same
behavior for all derivatives (Fig. S20). Here, we showed
only the MOs for compound 3a (Figure 5b). A push–pull
effect can be seen with well-localized MOs, showing a
charge transfer character in the molecule (CT) [25]. In
general, HOMO is located in the part of the phenol
ring. LUMO in the lactonic and zwitterionic forms is
located in the lactonic ring and in the carboxylate ion,
respectively. Here, again, we observe the similarity in
the lactonic and zwitterionic forms, where MOs are
located in the same parts of the molecular structure.
For the quinoidal form, we did not observe the push–
pull effect on the molecule. HOMO and LUMO are
located in the part of the molecule originating from the
phenol.
2018, 148, 81.
[2] Andrade, Z.; Carlos, K. Curr Org Synth 2004, 1(4), 333.
[3] (a) Gore, P. H. Chem Rev 1955, 55, 229. (b) Calloway, N. O.
Chem Rev 1935, 17, 327. (c) Bandini, M.; Melloni, A.; Umani-Ronchi, A.
Angew Chem Int Ed 2004, 43(5), 550.
[4] (a) da Silva Barbosa, J.; da Silva, G. V. J.; Constantino, M. G.
Tetrahedron Lett 2015, 56, 4649. (b) da Silva, B. H. S. T.; Bregadiolli, B.
A.; Graeff, C. F. D. O.; da Silva-Filho, L. C. ChemPlusChem 2017, 82(2),
261.
[
[
5] Sabnis, R. W. Tetrahedron Lett 2009, 50, 6261.
6] Sabnis, R. W. Handbook of Acid-Base Indicators; CRC Press:
Boca Raton, Fla, 2008.
[
[
7] Sabnis, R. W. Color Technol 2018, 134, 187.
8] Jha, A.; Garade, A. C.; Mirajkar, S. P.; Rode, C. V. Ind Eng
Chem Res 2012, 51, 3916.
9] Sabnis, R. W. Color Technol 2018, 134, 347.
[10] Kukreti, V.; Chamoli, R. P. Dyes Pigm 1996, 32, 15.
11] (a) Bhuchar, V. M.; Kukreja, V. P.; Das, S. R. Anal Chem
971, 43(13), 1847. (b) Takezawa, H.; Akiba, S.; Murase, T.; Fujita, M.
[
[
1
J Am Chem Soc 2015, 137, 7043.
CONCLUSION
[12] Arbeloa, F. L.; Costela, A.; Arbeloa, I. L. J Photochem
Photobiol A Chem 1990, 55, 97.
[
13] Lakowicz, J. R. Principles of frequency-domain fluorescence
It is concluded that NbCl is a good catalyst for the
5
spectroscopy and applications to cell membranes; In Fluorescence studies
synthesis of phthalein derivatives, because the reactions
occur in low reaction times and with high yields, using
low concentrations of NbCl5 (25 mol %). These
derivatives have good applications for use as acid–base
indicators, obtaining good color variation at different
pH values. The derivatives obtained absorptions within
the range of the visible light spectrum and with high
intensities of absorption when dissolved in alkaline
on biological membranes; Springer: Boston, MA, 1988, pp 89–126.
[14] Sabnis, R. W. Phthalein Dyes; In Kirk-Othmer Encyclopedia
of Chemical Technology; Wiley-Interscience: Hoboken, N.J, 2000,
pp 1–21.
[15] Mukhopadhyay, A.; Mishra, A. K.; Jana, K.; Moorthy, J. N. J
Photochem Photobiol A Chem 2017, 347, 199.
[
16] Benassi, E.; Carlotti, B.; Fortuna, C. G.; Barone, V.; Elisei, F.;
Spalletti, A. Chem A Eur J 2015, 119, 323.
17] Yang, Z.; Qin, W.; Lam, J. W.; Chen, S.; Sung, H. H.; Wil-
liams, I. D.; Tang, B. Z. Chem Sci 2013, 4, 3725.
[
Journal of Heterocyclic Chemistry
DOI 10.1002/jhet

 Moreno, Vitor Fernandes
Moreno, Vitor Fernandes
 dos Santos, Giovanny Carvalho
dos Santos, Giovanny Carvalho
 da Costa, Gyordanna Mayara Gaspar
da Costa, Gyordanna Mayara Gaspar
 Gomes, Marcelo Henrique Ayala
Gomes, Marcelo Henrique Ayala
 Silva-Filho, Luiz Carlos da
Silva-Filho, Luiz Carlos da