Table 3. Purification of AlaGln
UV 210 nm; retention times, 1: 15.0 min, isomer (D-alanyl-
L-glutamine): 18.4 min, 4: 26.3 min, 5: 6.0 min, 6: 4.0
min, 7: 34.0 min.
HPLC area (%)b
1
4
5
6
7
isomer
LC-MS analyses: HPLC conditions: column, Lichrosorb-
NH2 (GL Science); eluent, 0.05 mol/L AcONH4 and MeCN
(35:65), detection UV 210 nm, retention times, 4: 44.7 min,
5: 3.3 min, 7: 51.0 min. MS conditions: ion mode, ESI
positive; scan range, m/z 30-1000; source temp. 120 °C.
Found (m/z) L-alanyl-L-glutamic acid (4) C8H14N2O5: 219
(M + H)+, (2S,5S)-2-(2-carbamoylethyl)-5-methylpiperazine-
3,6-dione (5) C8H13N3O3: 200 (M + H)+, (2S,4S)-3-aza-
2,4-dimethyl-1,5-pentanedioyl-di-L-glutamine (7) C16H27-
N5O8: 418 (M + H)+.
before purification 97.73 0.12 0.02 0.06 0.12 1.28
resin eluenta
recrystall.
98.53 N.D. 0.01 0.05 N.D. 1.32
99.80 N.D. N.D. N.D. N.D. 0.19
a Collected fractionsts of resin treatment, see Experimental Section. b N.D.:
Not Detected (less than 0.01 %).
able biological contamination such as endotoxins. The use
of ethanol was also adopted from a wider allowance of the
residual solvent than methanol.
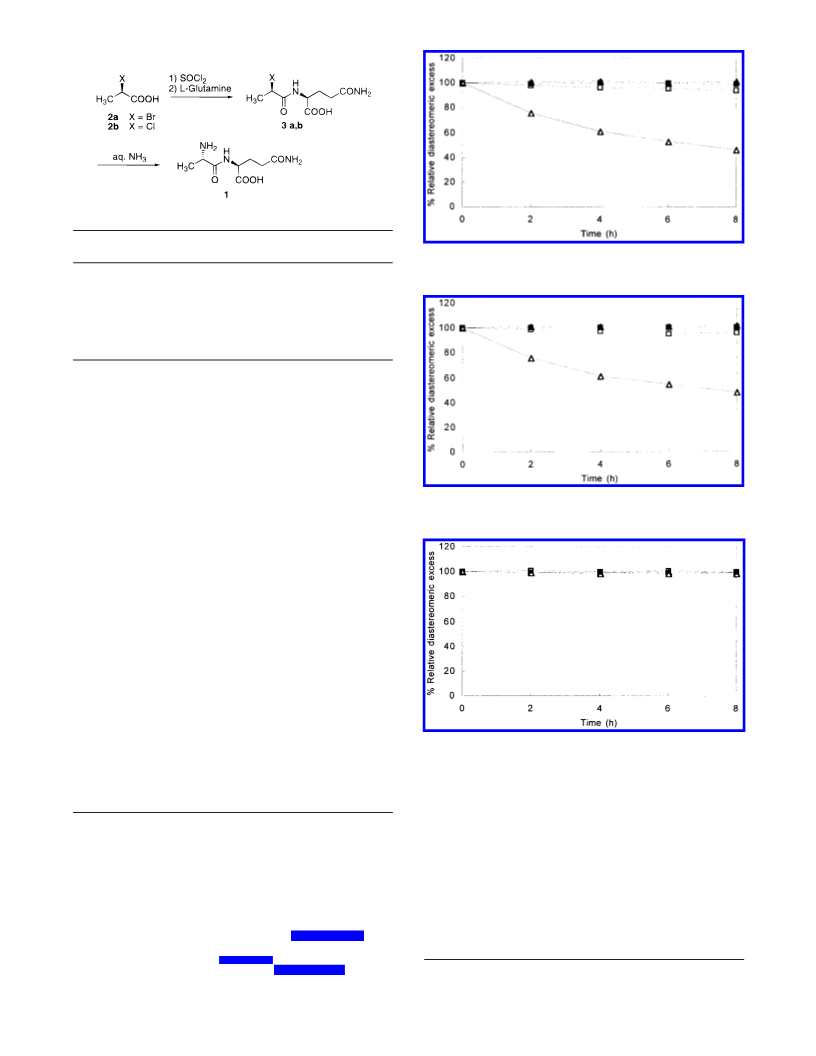
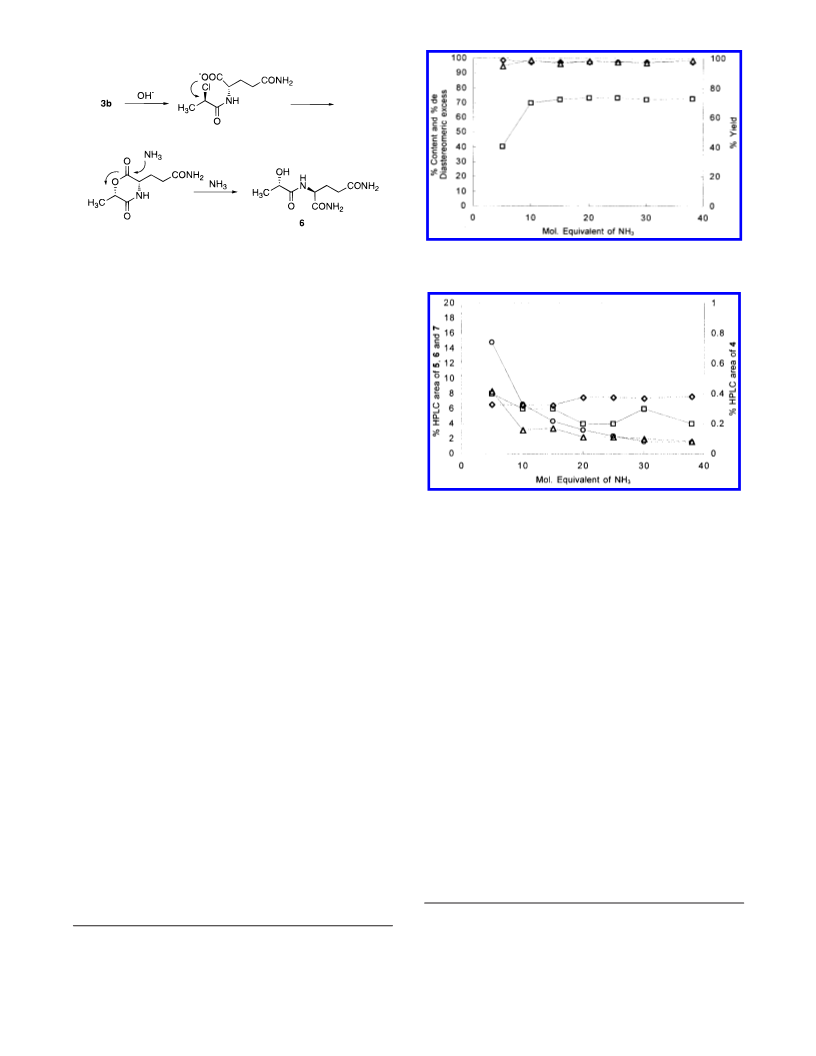
Synthesis of D-2-Chloropropionyl-L-glutamine (3b). To
D-2-chloropropionic acid (600 g, 5.53 mol; 98.8% ee), was
added SOCl2 (724 g, 6.08 mol) at 65 °C (Caution: the
scrubber should be completely prepared for remoVing
poisonous gas!) and heated further for 1 h at 85 °C. The
reaction mixture was cooled to room temperature to give
745 g of the oily product. The mixture (722 g) was diluted
with toluene (400 mL) to make a toluene solution of D-2-
chloropropionyl chloride (5.36 mol). A mixture of L-
glutamine (784 g, 5.36 mol), H2O (300 mL), and toluene
(150 mL) was cooled to 0-5 °C and stirred, and then to the
cooled solution was added 5 mol/L aqueous NaOH (1000
mL). To the solution, were added concurrently a toluene
solution of D-2-chloropropionyl chloride described above and
5 mol/L aqueous NaOH below 10 °C while maintaining the
pH of the reaction solution at pH 10. The reaction mixture
was stirred further 1 h at 10 °C. To the mixture, was added
concentrated HCl (30 mL) at room temperature to adjust the
pH to 6. Then, toluene was removed by extraction. To the
water layer, was added concentrated HCl (410 mL) at room
temperature to adjust the pH to 2. The precipitated crystals
were filtered and dried in vacuo to afford 3b: 1038 g (4.38
mol; diastereomeric excess 99.7% de; yield 81.7% from D-2-
chloropropionic acid); mp 153 °C dec (recrystallized twice
from H2O); [R]20D -9.9 ° (c ) 5, H2O); 1H NMR (DMSO-
d6) δ ) 1.54 (3H, d, J ) 6.6 Hz, CH3), 1.70-2.10 (2H, m,
CH2CH2CONH2), 2.14 (2H, t, J ) 7.1 Hz, CH2CH2CONH2),
4.13-4.23 (1H, m, CHNH-), 4.59 (1H, q, J ) 6.7 Hz,
CHCl), 6.82 (1H, s, CONH), 7.37 (1H, s, CONH), 8.60 (1H,
d, J ) 7.7 Hz, -NH-); IR (KBr) υ ) 1738, 1662 cm-1;
HRESIMS calcd for C8H1335ClN2O4 m/z 235.0486 (M - H)+,
found 235.0494.
Summary
Process research and development of AlaGln have been
performed for large-scale manufacture. From the viewpoint
of the stability and the cost, the method via D-2-chloropro-
pionyl-L-glutamine (3b) was adopted. In the following
ammonolysis reaction, the structures of the by-products were
inferred mainly from mass spectrometry, and a method
removing these compounds by passing through the anion
resin was developed. From the studies described here, a
manufacturing method of AlaGln used for a component of
parenteral nutrition has been accomplished.
Experimental Section
1
General. H NMR spectra were recorded at 300 MHz
on a Bruker AC-300 spectrometer, and signals are given in
ppm using TMS as an internal standard. IR spectra were
recorded on a Shimadzu FTIR-4300 spectrophotometer.
Optical rotation was measured with Jasco P-1020 polarim-
eter. HRMS were recorded on a Micromass LCT or a JEOL
LMS SX-102 mass spectrometer. LC-MS spectra were
recorded on Micromass Qauatro mass spectrometer. D-2-
Bromopropionic acid (2a; (R)-2-bromopropionic acid) was
purchased from Osaka Synthetic Chemical Laboratories Inc.
and D-2-chloropropionic acid (2b; (R)-2-chloropropionic
acid) was purchased from Nippon Fine Chemical Co., Ltd.
For HPLC analysis of the diastereomeric excess,9 L-alanyl-
D-glutamine and D-alanyl-L-glutamine were purchased from
Bachem AG, and D-alanyl-D-glutamine was synthesized.8
Other reagents and solvents are of commercial quality.
HPLC analyses: Purity and optical purity of 3a: column,
YMC-Pack ODS-AQ313 (YMC), eluent: 0.01 mol/L KH2-
PO4; detection, UV 210 nm; retention times, 3a: 31.0 min,
isomer (L-2-bromopropionyl-L-glutamine): 37.5 min. Purity
of 3b: column, Shim-Pack CLC-ODS (Shimadzu); eluent,
aqueous solution of 0.01 mol/L KH2PO4 and 0.01 mol/L
sodium octanesulfonic acid adjusted to pH 2.5 by H3PO4:
MeOH (100:1); detection, UV 210 nm; retention time, 3b:
6.3 min. Optical purity of 3b: column, YMC-Pack ODS-
AQ313; eluent 0.01 mol/L KH2PO4; retention times, 3b: 25.1
min, isomer (L-2-chloropropionyl-L-glutamine): 27.9 min.
Purity and diastereomeric excess of AlaGln(1):18 column,
TSK gel ODS-120 T (Tosoh); eluent, aqueous solution of
0.01 mol/L KH2PO4 and 0.01 mol/L sodium octanesulfonic
acid adjusted to pH 2.5 by H3PO4:MeOH (100:1); detection,
Synthesis of L-Alanyl-L-glutamine (1). D-2-Chloropro-
pionyl-L-glutamine (3b) (60.0 g, 0.24 mol; 96.9% de) and
28% aqueous NH3 (600 mL) were put into a 1 L glass
autoclave with mechanical stirring at room temperature. The
resulting solution was allowed to stand at 60 °C for 8 h.
The reaction mixture was cooled to room temperature and
concentrated to ∼150 mL in vacuo, and then MeOH was
added dropwise to the residue at room temperature. The
(18) The optical purity of AlaGln(1) was calculated as diastereomeric excess %
(% de) of 1 to D-alanyl-L-glutamine from the HPLC analysis described in
the Experimental Section. Other isomers were confirmed not to be contained
in the final product by the following HLPC conditions: column, Crown-
Pack CR (+) and/or (-) (Daicel); eluent, 0.1% aqueous HClO4 (pH 2);
detection, UV 210 nm.
Vol. 4, No. 3, 2000 / Organic Process Research & Development
•
151

 Sano, Takahiro
Sano, Takahiro
 Sugaya, Toru
Sugaya, Toru
 Inoue, Kunimi
Inoue, Kunimi
 Mizutaki, Sho-Ichi
Mizutaki, Sho-Ichi
 Ono, Yasuyuki
Ono, Yasuyuki
 Kasai, Masaji
Kasai, Masaji