Angewandte
Chemie
stable than the parent metal borohydrides,[1b] which further
facilitates their use for the synthesis of M3 [M2(BH4)z].[11,23b]
y
In summary, we have described a wet-chemistry method
for the synthesis of complex metal borohydrides at room
temperature. This method eliminates the “dead mass” prob-
lem that occurs when the mechanochemical synthesis is used
as an exclusive synthetic tool, or when ethereal solvents are
applied. The new method is quantitative, scalable, leads to
borohydride products of high purity, and eliminates the need
to use toxic B2H6 for the synthesis.[22] Use of low-boiling
solvents such as CH2Cl2 (which may circulate in a closed
system in an industrial setting) also renders the method
energy efficient.[24,25] The described method, which works
neatly for bimetallic borohydrides, complements the recently
developed approach for the synthesis of unsolvated mono-
metallic borohydrides with dimethyl sulfide as the solvent.[26]
Figure 2. a) Temperature-resolved mass spectrum of the gases that
evolved during the thermal decomposition of Li[Zn2(BH4)5] (only
W_LiC). The ions that result in the most significant maxima in ion
current have been specified. b) TGA and DSC profiles of contaminated
(D_Li2.5) and pure (W_LiC) Li[Zn2(BH4)5] samples.
Received: August 22, 2014
Revised: November 13, 2014
Published online: && &&, &&&&
Keywords: alkali metal halides · borohydrides ·
.
hydrogen storage · organic solvents · weakly coordinating anions
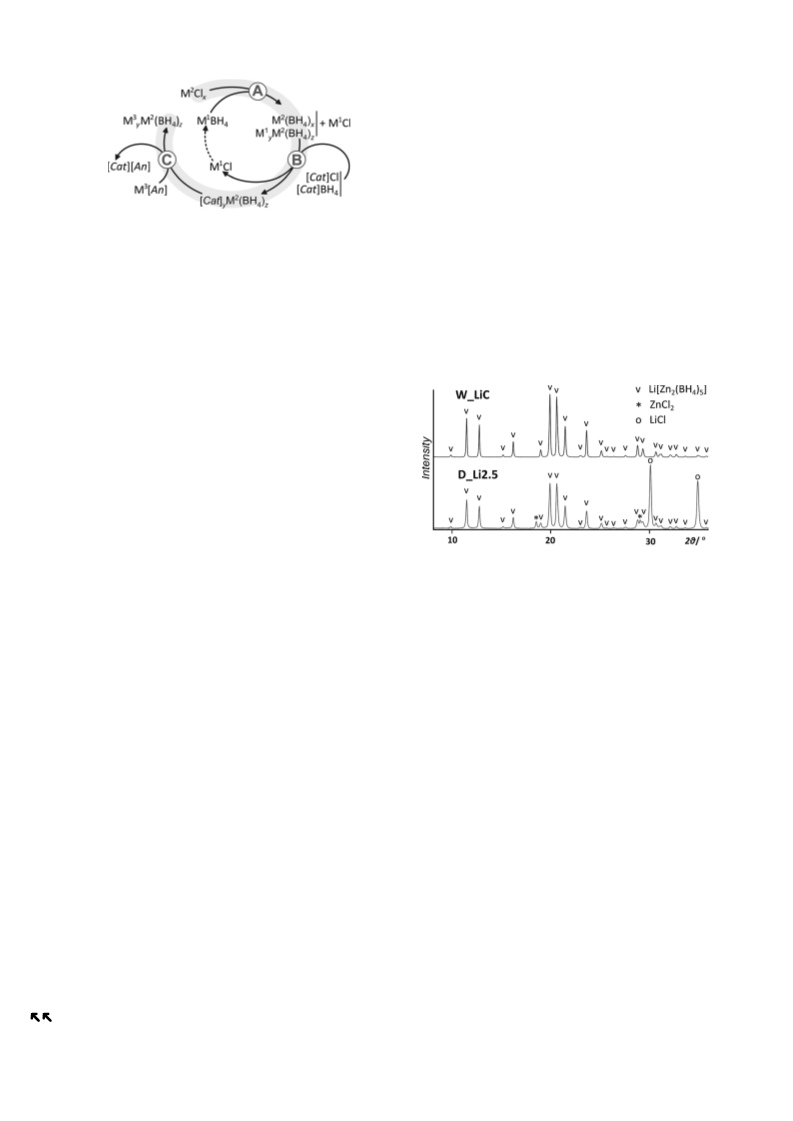
the basis of the crystallite size, which is larger for the sample
prepared by precipitation (an estimate from the Scherrer
equation gives a more than 50% larger average crystallite size
for the sample W_LiC as compared to D_Li2.5).
[2] A. Karkamkar, S. M. Kathmann, G. K. Schenter, D. J. Helde-
[3] a) L. H. Rude, T. K. Nielsen, D. B. Ravnsbæk, U. Bçsenberg,
M. B. Ley, B. Richter, L. M. Arnbjerg, M. Dornheim, Y.
Next, the technicalities and limitations of the new method
are discussed. The use of volatile and weakly coordinating
dichloromethane as the reaction medium enables an easy
desolvation. This is a critical issue, especially for the thermally
less stable borohydrides that could not be prepared in their
desolvated form using typical ethereal, organic sulfide, or
amine solvents. Simultaneously, the moderate polarity of
CH2Cl2 restricts the choice of precursors for the mixed-metal
borohydrides. While the borohydrides that contain large
organic cations, [Cat]+, are very soluble in CH2Cl2, the
solubility of many common precursors that contain alkaline
metal cations, M[An], is insufficient. The successful meta-
thesis in CH2Cl2 has been enabled only by the application of
the M[An] precursors that contain weakly coordinating
anions, such as [Al{OC(CF3)3}4]ꢁ or [B{3,5-(CF3)2C6H3}4]ꢁ.[20]
As a result of the large volume of the anion and significant
shielding of the negative charge by the bulky, fluorine-rich
substituents, the salts that contain these anions show a small
lattice enthalpy (even for the tiny and hard Li+ cation), which
enhances their solubility in the weakly solvating solvents and
renders them an excellent source of “naked” metal cations.[21]
Our preliminary results indicate that the method described
here may easily be transferred to other combinations of metal
(M2, M3) and organic ([Cat]) cations, because soluble complex
borohydrides, [Cat]y[M2(BH4)z], have been reported for
selected metals M2, for example, [Ph4P][Zn(BH4)3],
[Ph4P]2[Mg(BH4)4], [Et4N][Al(BH4)4].[23] These compounds
can be conveniently synthesized either in solvent-mediated
reactions, or in a one-pot mechanochemical process followed
by the extraction as for [Cat][Y(BH4)4].[18] These organic
mixed-cation borohydrides are typically more thermally
´
[4] E. A. Nickels, M. Owen Jones, W. I. F. David, S. R. Johnson,
[5] The properties of borohydrides can also be modified by doping
with a catalyst, synthesis of composite materials, or incorpora-
tion into nanoporous scaffolds (i.e. nanoconfinement). See for
example: H.-W. Li, Y. Yan, S. Orimo, A. Zꢀttel, C. M. Jensen,
[6] a) H. Hagemann, M. Longhini, J. W. Kaminski, T. A. Wesolow-
ˇ
´
ski, R. Cerny, N. Penin, M. H. Sørby, B. C. Hauback, G. Severa,
ˇ
´
Cerny, G. Severa, D. B. Ravnsbæk, Y. Filinchuk, V. D’Anna, H.
Hagemann, D. Haase, C. M. Jensen, T. R. Jensen, J. Phys. Chem.
ˇ
´
C 2010, 114, 1357 – 1364; c) R. Cerny, D. B. Ravnsbæk, G.
Severa, Y. Filinchuk, V. D’Anna, H. Hagemann, D. Haase, J.
Skibsted, C. M. Jensen, T. R. Jensen, J. Phys. Chem. C 2010, 114,
19540 – 19549.
ˇ
´
ˇ
[7] R. Cerny, P. Schouwink, Y. Sadikin, K. Stare, L. Smrcok, B.
Richter, T. R. Jensen, Inorg. Chem. 2013, 52, 9941 – 9947.
ˇ
´
[8] R. Cerny, N. Penin, H. Hagemann, Y. Filinchuk, J. Phys. Chem. C
2009, 113, 9003 – 9007.
[9] D. Ravnsbæk, Y. Filinchuk, Y. Cerenius, H. J. Jakobsen, F.
Besenbacher, J. Skibsted, T. R. Jensen, Angew. Chem. Int. Ed.
2009, 48, 6659 – 6663; Angew. Chem. 2009, 121, 6787 – 6791.
[10] a) N. N. Mal’tseva, N. B. Generalova, A. Y. Masanov, K. Y.
Zhizhin, N. T. Kuznetsov, Russ. J. Inorg. Chem. 2012, 57, 1631 –
1652, and the references therein; b) J. Huot, D. B. Ravnsbæk, J.
Angew. Chem. Int. Ed. 2014, 53, 1 – 5
ꢀ 2014 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
3
These are not the final page numbers!

 Jaro, Tomasz
Jaro, Tomasz
 Orlowski, Piotr A.
Orlowski, Piotr A.
 Wegner, Wojciech
Wegner, Wojciech
 Fijalkowski, Karol J.
Fijalkowski, Karol J.
 Leszczyski, Piotr J.
Leszczyski, Piotr J.
 Grochala, Wojciech
Grochala, Wojciech