120-51-4Relevant articles and documents
HOMOMOLECULAR ESTERIFICATION OF ALDEHYDES CATALYZED BY HYDRIDOTETRAKIS(TRIPHENYLPHOSPHINE)RHODIUM(I)
Massoui, M.,Beaupere, D.,Nadjo, L.,Uzan, R.
, p. 345 - 354 (1983)
Homomolecular esterification of benzaldehyde has been carried out under mild conditions using RhH(PPh3)4 as catalyst.A kinetic study of the reaction has revealed a law rate of the form r=k2.The rate-limiting step in the mechanism is the complexation of the second aldehyde molecule with the catalyst.As expected from this result, the lactonisation of ortho-phthalaldehyde is instantaneous at 19 deg C.In this homomolecular esterification RhH(PPh3)3 is the catalytic species and slowly transfers its hydrogen ligand to benzaldehyde to form benzyl alcohol in concentration less than half of that of the catalyst.The complex Rh(PPh3)3 which is formed also catalyses the homomolecular esterification, but 1.6 times less readily than RhH(PPH3s)3.
Bisphosphine phenol and phenolate complexes of Mn(i): Manganese(i) catalyzed Tishchenko reaction
Kadassery, Karthika J.,MacMillan, Samantha N.,Lacy, David C.
, p. 12652 - 12655 (2018)
We synthesized new organomanganese complexes using the phenolic "pincer" type ligand H-POP. The coordination chemistry of H-POP with Mn(i) was explored, revealing a wide range of binding motifs. Finally, we found that complex 1 catalyzes the formation of benzyl benzoate from benzaldehyde in a Tishchenko reaction.
Modification of mesoporous silicate SBA-15 with tris[bis(trimethylsilyl) amido]samarium and its utility in Tishchenko reaction
Chen, Yinghua,Zhu, Zhenyu,Zhang, Jie,Shen, Jianzhong,Zhou, Xigeng
, p. 3783 - 3789 (2005)
Treatment of mesoporous silicate SBA-15 with Sm[N(SiMe3) 2]3 led to the formation of a novel organolanthanide/ inorganic hybrid material [SBA-15]Sm[N(SiMe3)2] x via abstraction of N(SiMe3)2 by terminal silanol groups and subsequent surface silylation. The hybrid material was characterized by elemental analyses, IR spectroscopy, X-ray diffraction, and nitrogen sorption, indicating a successful tailoring inside the silicate SBA-15 and the maintenance of the well-ordered mesostructure. This hybrid material is a promising heterogeneous catalyst for the Tishchenko reaction, where it is superior to the homogeneous correspondent in deactivation behavior, reusability and relative tolerance to oxygen, particularly in the control of selectivity of mixed Tishchenko reaction due to the steric hindrance and the diffusion control derived from the surface confinement.
Superbase-promoted acylation of hindered alcohols
D'Sa, Bosco A.,Verkade, John G.
, p. 2963 - 2966 (1996)
The commercially available nonionic superbase P(MeNCH2CH2)3N (1a) is very useful for the acylation of unreactive hindered alcohols as well as acid-sensitive alcohols. The reactions proceed in high yields using an acid anhydride, and 1a can be regenerated in a single step. The relative rates for benzoylation of (±)-menthol in C6D6 using conventional acylation reagents and strong nonionic bases are compared. In general, acetylation with 1a is accelerated in the polar solvent CH3CN whereas benzoylation is faster in the nonpolar solvent C6H6. The benzoylation intermediate RC(O)P(MeNCH2CH2)3N+ was found to be in equilibrium with 1a, with lower temperatures favoring the intermediate. The relative stabilities of several known acylating intermediates are compared.
Lanthanoid-Catalyzed Aldehyde Dimerization and Its Application to Polyester Synthesis
Onozawa, Shun-ya,Sakakura, Toshiyasu,Tanaka, Masato
, p. 531 - 534 (1994)
A wide variety of aldehydes are efficiently dimerized to esters by a catalytic amount of (C5Me5)2LnCH(SiMe3)2 (Ln=Nd, La).The reaction is applicable to the synthesis of polyesters from dialdehydes.A stoichiometric reaction of the lanthanum complex with benzaldehyde indicates the intermediacy of alkoxo complexes in the catalysis.
Ruthenium complexes featuring cooperative phosphine-pyridine-iminophosphorane (PNN) ligands: Synthesis, reactivity and catalytic activity
Cheisson, Thibault,Mazaud, Louis,Auffrant, Audrey
, p. 14521 - 14530 (2018)
The coordination to ruthenium(ii) centres of two phosphine-pyridine-iminophosphorane ligands LR (PPh2CH2(C6H3N)CH2NPR3, R = Ph or Cy) differing by the nature of the substituent of the PN phosphorus was explored. Coordination to [RuCl2(PPh3)3] afforded the complexes [RuLRCl2(PPh3)] that were successfully deprotonated at the acidic phosphinomethyl position. With LCy, coordination led to a mixture of two isomers. The complexes [RuLRHCl(PPh3)] were similarly obtained from [RuHCl(PPh3)3]. The stability of these complexes depends on the ligand substitution pattern; with LPh a CH activation process took place, while [RuLCyHCl(PPh3)] was thermally stable. Deprotonation of this latter complex was achieved and gave a catalytically competent species for the acceptorless dehydrogenative coupling of alcohols.
Benzylation of carboxylic acids by oxidation-reduction condensation using quinones and benzyloxydiphenylphosphine
Mukaiyama, Teruaki,Shintou, Taichi,Kikuchi, Wataru
, p. 1126 - 1127 (2002)
Various carboxylic benzyl esters were obtained in high yields by way of oxidation-reduction condensation in which benzyloxydiphenylphosphine (BDPP), having been prepared easily from chlorodiphenylphosphine and benzyl alcohol, was treated with various carboxylic acids in the presence of 2,6-dimethyl-1,4-benzoquinone(DMBQ) under mild conditions.
Silver nanoparticles stabilized by a metal-organic framework (MIL-101(Cr)) as an efficient catalyst for imine production from the dehydrogenative coupling of alcohols and amines
Gumus, Ilkay,Karata?, Yas?r,Gülcan, Mehmet
, p. 4990 - 4999 (2020)
In this paper, we present silver nanoparticles supported on a metal-organic framework (Ag@MIL-101) as a catalyst for the one-pot tandem synthesis of imines from alcohols and amines. The Ag@MIL-101 catalyst was fabricated using the liquid phase impregnation technique as an easy and effective method. The morphology and chemical structure of the prepared catalyst were systemically evaluated by inductively coupled plasma optical emission spectroscopy (ICP-OES), powder X-ray diffraction (P-XRD), X-ray photoelectron spectroscopy (XPS) and transmission electron microscopy (TEM). The highly crystalline feature of the Ag metal nanoparticles on bifunctional MIL-101(Cr) was revealed by TEM and HRTEM analyses. The Ag@MIL-101 catalyst was examined as a catalyst for the synthesis of different imine derivatives. The catalytic performance of the Ag@MIL-101 catalyst was monitored by GC-MS and 1H-NMR analyses for the dehydrogenative coupling of alcohols and amines. Furthermore, the effect of various factors such as the amount of catalyst, base, temperature and solvent was investigated to optimize the conditions for the best performance of the Ag@MIL-101 catalyst. Catalytic activity test results showed that the Ag@MIL-101 catalyst has a good catalytic activity with TOF values in the range of 12.7-14.6 h-1 in the synthesis of various imines.
PH-sensitive gold nanoparticle catalysts for the aerobic oxidation of alcohols
Yuan, Yuan,Yan, Ning,Dyson, Paul J.
, p. 11069 - 11074 (2011)
Gold nanoparticles (NPs) stabilized by carboxylate modified polyvinylpyrrolidone have been prepared and fully characterized. The gold NPs efficiently catalyze the aerobic oxidation of benzyl alcohol in water at ambient temperature and are easily separated from the reaction mixture by lowering the pH of the solution, causing the NPs to precipitate. The mechanism of the precipitation process has been studied. Due to the efficiency of this process, the NPs may be reused as catalysts by readjusting their pH.
Tributylphosphine-catalyzed acylation of alcohol by active ester directed toward effective end-capping of pseudorotaxane consisting of ammonium group and crown ether
Kihara, Nobuhiro,Nakakoji, Naohisa,Takata, Toshikazu
, p. 924 - 925 (2002)
According to the results of a model study using benzyl alcohol, pseudorotaxane with terminal hydroxy group on the axle was acylated with S-2-pyridyl 3,5-dimethylthiobenzoate in the presence of tributylphosphine to produce rotaxane in 85% yield. Optically
2(3H)-BENZOXAZOLETHIONE.
Ueda,Kawaharasaki,Imai
, p. 85 - 89 (1984)
A new condensing agent, N,N prime -carbonyldi left bracket 2(3H)-benzoxazolethione right bracket , was readily prepared by the reaction of 2-benzoxazolethiol and trichloromethyl chloroformate in benzene. The condensing agent is shown to be useful for the preparation of amides, esters, and dipeptides under mild conditions. A successful polyamide synthesis by the polycondensation of isophthalic acid with diamines using the condensing agent is also described.
CeO2 as a versatile and reusable catalyst for transesterification of esters with alcohols under solvent-free conditions
Tamura, Masazumi,Hakim Siddiki,Shimizu, Ken-Ichi
, p. 1641 - 1646 (2013)
CeO2 acted as an efficient and reusable heterogeneous catalyst for transesterification of esters with alcohols under the solvent-free conditions at 160 °C. Among the 11 kinds of metal oxides, CeO2 is the most suitable catalyst in terms of catalytic activity, leaching-resistance and reusability. This catalytic system tolerates various esters and alcohols, and valuable esters such as heteroaromatic esters and benzyl benzoates are produced, demonstrating a practical utility of the system. On the basis of kinetic analysis and in situ IR studies of adsorbed species, a reaction mechanism is proposed, in which proton abstraction of alcohol by a Lewis base site of CeO2 to yield alkoxide species is the rate-limiting step.
Synthesis and coordination chemistry of tridentate (PNN) amine enamido phosphine ligands with ruthenium
Wambach,Lenczyk,Patrick,Fryzuk
, p. 5583 - 5589 (2016)
Tridentate amine-imine-phosphine ligands, R2PC5H7NC2H4NEt2 [RPNN(H)], where R = Pri or But are synthesized using a straightforward protocol of condensation, deprotonation, and addition of a chlorodialkylphosphine. Multinuclear NMR spectroscopy shows the ligands exist exclusively in the enamine tautomeric form in solution. Treating these ligands with RuHCl(PPri3)2(CO) forms the desired coordination compounds, RuHCl[RPNN(H)](CO), where the imine tautomeric form of the ligands coordinates to ruthenium. Deuterium labelling experiments show Ru-H/N-D scrambling occurs during ligand coordination. Treating the RuHCl[RPNN(H)](CO) precursors with potassium tert-butoxide allows for the synthesis of two new ruthenium enamido-phosphine complexes, RuH[RPNN](CO), which were fully characterized. The structure of one of the derivatives was confirmed by X-ray crystallography (R = Pri). The reactivity of the enamido-phosphine complexes with H2 and benzyl alcohol is also reported. For the enamido phosphine complex where R = Pri, the reaction with H2 is reversible and forms (RuH(CO)[PriPNN(H)])2(μ-H)2, a hydride-bridged dimer that results from cooperative activation of H2. The reactivity of both amine-enamido-phosphine ruthenium compounds with benzyl alcohol establishes that the complexes are catalyst precursors for acceptorless dehydrogenation (AD), although the turnover frequencies measured using both catalyst precursors are modest.
Synthesis and reactivity of ruthenium complexes bearing arsenic-containing arsenic-nitrogen-arsenic-type pincer ligand
Tanabe, Yoshiaki,Kuriyama, Shogo,Arashiba, Kazuya,Nakajima, Kazunari,Nishibayashi, Yoshiaki
, p. 5295 - 5300 (2014)
Dinuclear and mononuclear ruthenium complexes bearing an arsenic-containing arsenic-nitrogen-arsenic (ANA)-type pincer ligand are designed, prepared, and characterized by X-ray analyses. Both ruthenium complexes work as effective catalysts toward dehydrogenative transformations of alcohols.
Imidazolium ionic liquid-supported sulfonic acids: Efficient and recyclable catalysts for esterification of benzoic acid
Cai, Yue Qin,Yu, Guo Qiang,Liu, Chuan Duo,Xu, Yuan Yuan,Wang, Wei
, p. 1 - 4 (2012)
Several imidazolium ionic liquid (IL)-supported sulfonic acids with different anions, [C3SO3Hmim]HSO4, [C 3SO3Hmim]BF4, [C3SO 3Hmim]PF6, and [C3SO3Hmim]CF 3SO3, were synthesized and applied as catalysts for esterification reaction of benzoic acid. The experimental results indicate that imidazolium IL-supported sulfonic acid containing anion of HSO4 - shows the best catalytic activity. Only when less [C 3SO3Hmim]HSO4 (0.3 equiv.) applied, was the product obtained with high yield of 97%. Furthermore, the produced esters could be separated by decantation, and the catalyst could be reused after the removal of water.
Synthesis of aromatic esters catalyzed by palladium on charcoal: An efficient heterogeneous catalyst for alkoxycarbonylation of aryl iodides
Ramesh, Chinnasamy,Nakamura, Ryo,Kubota, Yoshihiro,Miwa, Minoru,Sugi, Yoshihiro
, p. 501 - 504 (2003)
Palladium on charcoal was found to catalyze the carbonylation of aryl iodides with various aliphatic alcohols as well as less reactive phenols to give the corresponding esters in high yield at 140°C and in the presence of carbon monoxide (1.0 MPa). The carbonylation-polycondensation of 2,7-diiodo-9,10-dihydrophenanthrene with bisphenol-A afforded high molecular weight polyarylate, poly [oxy-1,4-phenylene (1-methylethylidene)-1,4-phenyleneoxycarbonyl (9,10-dihydro-2,7-phenanthrenediyl) carbonyl].
Efficient acceptorless dehydrogenation of secondary alcohols to ketones mediated by a PNN-Ru(II) catalyst
Wang, Zheng,Pan, Bing,Liu, Qingbin,Yue, Erlin,Solan, Gregory A.,Ma, Yanping,Sun, Wen-Hua
, p. 1654 - 1661 (2017)
Four types of ruthenium(II) complexes, [fac-PNN]RuH(PPh3)(CO) (A), [fac-PNHN]RuH(η1-BH4)(CO) (B), [fac-PNHN]RuCl2(PPh3) (C) and [fac-PNHN]RuH(η1-BH4)(PPh3) (D) (where PNHN and PNN are N-(2-(diphenylphosphino)ethyl)-5,6,7,8-tetrahydroquinoline-8-amine and its deprotonated derivative), have been synthesized and assessed as catalysts for the acceptorless dehydrogenation of secondary alcohols to afford ketones. It was found that C, in combination with t-BuOK, proved the most effective and versatile catalyst allowing aromatic-, aliphatic- and cycloalkyl-containing alcohols to be efficiently converted to their corresponding ketones with particularly high values of TON achievable. Furthermore, the mechanism for this PNN-Ru mediated process been proposed on the basis of a number of intermediates that have been characterized by EI-MS and NMR spectroscopy. These catalysts show great potential for applications in atom-economic synthesis as well as in the development of organic hydride-based hydrogen storage systems.
Bicyclic guanidinate compounds of magnesium and their activity as pre-catalysts in the Tishchenko reaction
Day, Benjamin M.,Mansfield, Natalie E.,Coles, Martyn P.,Hitchcock, Peter B.
, p. 4995 - 4997 (2011)
A synthetic route to magnesium guanidinate compounds that avoids ligand redistribution is reported; selected derivatives are active pre-catalysts in the dimerization of aldehydes.
Copper Tetrasulfophthalocyanine Intercalated Hydrotalcite as an Efficient Bifunctional Catalyst for the Baeyer–Villiger Oxidation
Zhou, Weiyou,Chen, Yong,Qian, Junfeng,Sun, Fu’an,He, Mingyang,Chen, Qun
, p. 2157 - 2164 (2016)
Abstract: A heterogeneous bifunctional hybrid catalyst originated from copper tetrasulfophthalocyanine (CuPcTs) and hydrotalcite for Baeyer–Villiger (B-V) oxidation has been prepared and characterized. XRD, FTIR, DR UV-Vis and SEM characterization indicate that CuPcTs molecule has been successfully intercalated into the layer of ZnAl hydrotalcite. And the synthesized hybrid exhibited excellent catalytic activity in the B-V oxidation for various ketones under mild conditions. Its bifunctional role in the reaction through O2/benzaldehyde has been discussed and verified by controlled experiments. The study indicates that the designed catalyst not only catalyzes the oxidation of benzaldehyde to perbenzoic acid, but also accelerates the transformation of ketone to lactone or ester. Graphical Abstract: [Figure not available: see fulltext.]
-
Koenig,P.E. et al.
, p. 4777 (1961)
-
Synthesis of Ruthenium Complexes Bearing PCP-Type Pincer Ligands and Their Application to Direct Synthesis of Imines from Amines and Benzyl Alcohol
Eizawa, Aya,Nishimura, Shunsuke,Arashiba, Kazuya,Nakajima, Kazunari,Nishibayashi, Yoshiaki
, p. 3086 - 3092 (2018)
Ruthenium complexes bearing N-heterocyclic carbene- and phosphine-based PCP-type pincer ligands are synthesized and characterized by X-ray crystallography. The ruthenium-PCP complexes have catalytic activity toward direct synthesis of imines from reactions of amines and benzyl alcohol. The lifetime of the ruthenium complex bearing the PCP pincer ligand is longer than that of the ruthenium complex bearing a pyridine-based PNP-type pincer ligand.
Zr(IV)-Fe(III), -Ga(III), and -Sn(IV) binary metal complexes as synergistic and reusable esterification catalysts
Sato, Atsushi,Nakamura, Yuka,Maki, Toshikatsu,Ishihara, Kazuaki,Yamamoto, Hisashi
, p. 1337 - 1340 (2005)
Synergism in catalytic activity with the combined use of Hf(O-i-Pr) 4 or Zr(O-i-Pr)4 and Fe(O-i-Pr)3 for direct esterification and a very simple method for their extraction with ionic liquids for their recovery and reuse are described. This Zr(IV)-Fe(III) complex is an environmentally benign catalyst due to its high turnover number, extremely mild Lewis acidity, low toxicity, reusability, and commercial availability at low cost.
The Direct Alkylation of π-Rich, Acid-Sensitive Heterocyclic Compounds via Essentially Free Carbocations
Darbeau, Ron W.,White, Emil H.
, p. 8091 - 8094 (1997)
Alkylation of π-rich heteroaromatics such as pyrroles and furans by the standard Friedel-Crafts approach is impractical because the acid catalysts employed (Bronsted or Lewis) induce polyalkylation, ring opening, and polymerization. The present study describes the facile benzylation of π-excessive heteroaromatics using the nitrosoamide approach, which generates nitrogen-separated carbocation-counterion ion-pairs as the alkylating agent with no catalyst being required. N-Nitrosoamides are favorable sources of carbocations because of the following variables: mildness of the conditions required to generate cations, high reactivity of the unsolvated carbocations formed, solubility of the precursors in a wide range of solvents, homogeneity of the reactions, wide range of decomposition temperatures possible, straightforward chemistry, and excellent product balance. The majority of the cations that are generated in pyrrole (80%) are intercepted by the solvent, and only 20% are intercepted by the counterion; this result provides support for the intermediacy of nitrogen-separated ion-pairs in deamination. A nucleophilicity scale is presented for reactions of selected nucleophiles with essentially free carbocations.
Catalytic bond forming reactions promoted by amidinate, guanidinate and phosphaguanidinate compounds of magnesium
Schwamm, Ryan J.,Coles, Martyn P.,Day, Benjamin M.,Mansfield, Natalie E.,Knowelden, William,Hitchcock, Peter B.
, p. 14302 - 14314 (2014)
The synthesis and catalytic properties of a series of magnesium compounds consisting of monoanionic, N,N'-chelating ligands (N∩N = amidinates, guanidinates, phosphaguanidinates) is reported. The compounds were synthesized by (i) insertion of a carbodiimide into an existing Mg-C or Mg-N bond, or (ii) protonolysis of an organomagnesium compound by a neutral pre-ligand. Structural analyses of mono- or bis-(chelate) compounds with general formula Mg(N∩N)X(L)nand Mg(N∩N)2(L)n(X = halide, aryloxide, amide; L = Et2O, THF; n = 0, 1 or 2) have been performed and the influence that the ligand substituent patterns have on the solid-state structures has been probed. Selected examples of the compounds were tested as (pre)catalysts for the polymerization of lactide, the dimerization of aldehydes and the hydroacetylenation of carbodiimides.
Pentacarbonylmethylmanganese(i) as a synthon for Mn(i) pincer catalysts
Kadassery, Karthika J.,Lacy, David C.
, p. 4467 - 4470 (2019)
Mn(i) complexes that enable metal-ligand cooperative substrate activation catalyze a range of transformations. Use of MeMn(CO)5 as a synthon in place of typical Mn(CO)5Br was explored and found to be quite versatile, generating catalytically active species in situ by activation of O-H, N-H, and even C-H bonds.
One-step solvent-free aerobic oxidation of aliphatic alcohols to esters using a tandem Sc-Ru?MOF catalyst
Feng, Tingkai,Li, Conger,Li, Tao,Zhang, Songwei
supporting information, p. 1474 - 1480 (2022/03/08)
Esters are an important class of chemicals in industry. Traditionally, ester production is a multi-step process involving the use of corrosive acids or acid derivatives (e.g. acid chloride, anhydride, etc.). Therefore, the development of a green synthetic protocol is highly desirable. This work reports the development of a metal-organic framework (MOF) supported tandem catalyst that can achieve direct alcohol to ester conversion (DAEC) using oxygen as the sole oxidizing agent under strictly solvent-free conditions. By incorporating Ru nanoparticles (NPs) along with a homogeneous Lewis acid catalyst, scandium triflate, into the nanocavities of a Zr MOF, MOF-808, the compound catalyst, Sc-Ru?MOF-808, can achieve aliphatic alcohol conversion up to 92% with ester selectivity up to 91%. A mechanistic study reveals a unique “via acetal” pathway in which the alcohol is first oxidized on Ru NPs and rapidly converted to an acetal on Sc(iii) sites. Then, the acetal slowly decomposes to release an aldehyde in a controlled manner for subsequent oxidation and esterification to the ester product. To the best of our knowledge, this is the first example of DAEC of aliphatic alcohols under solvent-free conditions with high conversion and ester selectivity.
Sodium organoaluminate containing bidentate pyrrolyl ligand: Synthesis, structure, and catalytic activity for the Tishchenko reaction
Liu, Yu,Guo, Zhiqiang,Wang, Yakong
, (2021/05/26)
An novel sodium organoaluminate containing bidentate pyrrolyl ligand [C4H3NH(2-CH2NHtBu)] was efficiently synthesized and characterized by X-ray crystallography. The molecular structure shows it is a monodimensional infinite chain structures with linear arrangements. Its basic repeat unit comprises the Al atom bonded to two deprotonated pyrrole rings and Na atom coordinated to of nitrogen atoms of –NtBu fragment, which undergoes further to coordinates a pyrrolyl ring of an adjacent molecule in a ?2-fasion. Furthermore, this sodium organoaluminate exhibited high catalytic activities for Tishchenko reaction.
ZWITTERIONIC CATALYSTS FOR (TRANS)ESTERIFICATION: APPLICATION IN FLUOROINDOLE-DERIVATIVES AND BIODIESEL SYNTHESIS
-
Paragraph 0015; 0028, (2021/01/29)
An amide/iminium zwitterion catalyst has a catalyst pocket size that promotes transesterification and dehydrative esterification. The amide/iminium zwitterions are easily prepared by reacting aziridines with aminopyridines. The reaction can be applied a wide variety of esterification processes including the large-scale synthesis of biodiesel. The amide/iminium zwitterions allow the avoidance of strongly basic or acidic condition and avoidance of metal contamination in the products. Reactions are carried out at ambient or only modestly elevated temperatures. The amide/iminium zwitterion catalyst is easily recycled and reactions proceed in high to quantitative yields.
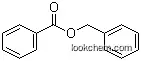
 Xn:Harmful;
Xn:Harmful;



