Refernces
10.1002/ejic.201000802
The study focuses on the synthesis and investigation of ruthenium complexes bearing N-H acidic pyrazole ligands and their application in catalytic hydrogenation reactions. The researchers treated chelate ligands containing pyrazole groups with various ruthenium precursors to form complexes with protic N-H groups near the catalytically active ruthenium center. These complexes were characterized by spectroscopic methods and DFT calculations, and their structure and reactivity were analyzed. The study aimed to understand the role of the acidic N-H groups in metal-ligand-bifunctional hydrogenation, where a hydrido ligand and a proton from a protic group are transferred simultaneously. The catalytic performance of these complexes was evaluated through the hydrogenation and transfer hydrogenation of acetophenone, and the results were connected to the ligand's electronic and structural properties. The research provides insights into the design of efficient catalysts for hydrogenation reactions by leveraging the acidic N-H groups in pyrazole ligands.
10.1016/j.tet.2009.06.041
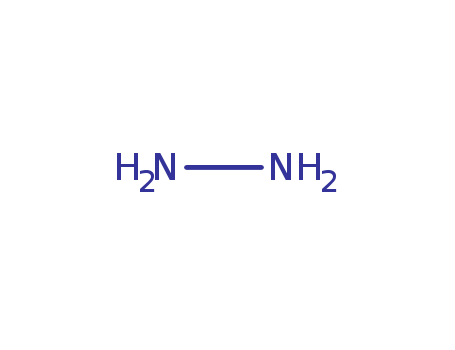
The research focuses on the chemical conversion of isothiazoles into pyrazoles using hydrazine as the primary reactant. The study explores the influence of various substituents at the C-3, C-4, and C-5 positions of the isothiazole ring on the ring transformation process and identifies limitations of this conversion. Experiments involve treating isothiazoles with anhydrous hydrazine, either neat or in combination with co-solvents like DMF or DMSO, and examining the products formed under different conditions, such as temperature and reaction time. The analyses used to characterize the products include techniques like thin-layer chromatography (TLC), melting point determination, UV-Vis spectroscopy, infrared (IR) spectroscopy, nuclear magnetic resonance (NMR) spectroscopy, and mass spectrometry (EI), which collectively provide a comprehensive evaluation of the reaction outcomes and the structural features of the synthesized pyrazole compounds.
10.1016/j.tetlet.2003.11.133
The study presents a concise synthesis method for a novel class of homochiral aromatic amino acid surrogates, featuring tetrahydroindazole or benzisoxazole systems. These surrogates were synthesized through the acylation of cyclic 1,3-diketone by the side-chain carboxyl functionality of specific amino acid precursors, followed by a regioselective condensation with hydrazine, N-benzylhydrazine, and hydroxylamine. The synthetic strategy is versatile, allowing for the creation of structurally diverse derivatives. These novel amino acids can be efficiently incorporated into proteins and have potential applications in imparting unique properties to biological peptides. The study also includes the synthesis of Na-Fmoc-protected derivatives, which are useful for solid-phase peptide assembly, and the exploration of the stereochemistry integrity of the homochiral starting material through chemical transformations. The synthesized amino acids offer opportunities as structural surrogates of tryptophan and as building blocks for designing molecular probes.
10.1021/jo102285z
The research focuses on the Ag(I)-catalyzed regioselective ring-opening of N-tosylaziridines and N-tosylazetidines with various S-, O-, and N-nucleophiles, as well as tethered dinucleophiles. The experiments utilized [Ag(COD)2]PF6 as a catalyst to facilitate the ring-opening reactions with nucleophiles such as alcohols, amines, thiols, and related 1,2-ethane dinucleophiles. Initial rate studies and DFT-based evaluations of stepwise energetics were conducted to understand the relationship between nucleophilic reactivity and binding affinity to cationic Ag(I). The study suggests an inverse relationship between the nucleophilic reactivity of a heteroatom donor and its binding affinity to cationic Ag(I). The analysis involved monitoring reactions using 1H NMR, and the products were purified and characterized using column chromatography and NMR spectroscopy. The research also explored the substrate scope and functional group selectivity, yielding a range of 1,2-amino ethers, diamines, amino thioethers, and 1,3-amino ethers in good to excellent yields.
10.1016/j.bmc.2007.10.070
The research focuses on the synthesis and antiplatelet activity of ethyl 4-(1-benzyl-1H-indazol-3-yl)benzoate (YD-3) derivatives. The main objective was to develop novel anti-PAR4 (protease-activated receptor 4) agents by synthesizing a series of YD-3 derivatives and evaluating their selective anti-PAR4 activity. The study involved a structure–activity relationship (SAR) analysis to identify key functional groups contributing to the anti-PAR4 activity. The experiments included the synthesis of various YD-3 derivatives through chemical reactions using reagents such as Et3N, CHCl3, HCl, NH2NH2, Pd/C, and EtOH, among others. The synthesized compounds were then tested for their ability to inhibit platelet aggregation, ATP release, and P-selectin expression, which are critical factors in arterial thrombosis. The analyses used included melting point determination, IR spectroscopy, NMR spectroscopy, MS spectrometry, UV spectrophotometry, and elemental analysis to characterize the compounds. The antiplatelet activity was assessed by measuring the inhibition of platelet aggregation induced by various agents such as U46619, collagen, thrombin, SFLLRN, and GYPGKF. Additionally, the release of ATP and expression of P-selectin were measured to evaluate the effect of the compounds on platelet granule secretion. The findings from these experiments were used to establish guidelines for the development of new antithrombotic drugs.
10.1002/ardp.19853180215
The research focuses on the synthesis and investigation of pyran derivatives, specifically benzodipyrandiones that are double fused with pyrimidine and pyrazole. The purpose of the study was to explore the chemical reactions and properties of these complex organic compounds. The researchers started with the Kostanecki method to synthesize benzodi-pyrandion from diacetylresorcin, which was then hydrolyzed to form a tetraketon. This intermediate reacted with CS2/KOH/DMS to yield 2,8-bisdimethylthiobenzodipyrandion, which was further transformed into various derivatives through reactions with piperidine, hydrazine, and benzamidin. The resulting compounds, including 3,7-diphenyl-benzo[1,2-b:5,4-b']di(pyrano[2,3-c]pyrazol)-4,6-dion, 4,8-diphenyl-benzo[1,2-b:5,4-b']di(pyrano[2,3-d]pyrimidin)-5,7-dion, and 3,7-Dibenzoyl-2,8-dipiperidino-benzo[1,2-b:5,4-b']dipyran-1,6-dion, were found to be poorly soluble in common solvents. The study concluded with the characterization of these compounds through melting point determination, yield calculation, and spectroscopic analysis, including infrared (IR) and proton nuclear magnetic resonance (1H-NMR) spectroscopy. The chemicals used in the process included diacetylresorcin, CS2, KOH, DMS, piperidine, hydrazine, benzamidin, and various solvents such as DMSO, aceton, and ethanol.
10.1248/cpb.37.2679
The research focused on the synthesis and pharmacological evaluation of pyridazino[4,5-b]carbazoles, a class of heterocyclic compounds with potential antineoplastic properties. The purpose of the study was to create and test these compounds for their cytotoxic activity against L1210 leukemia in mice. The synthesis involved a cyclization reaction of hydrazine with carbazole-2,3-methyl dicarboxylates to form 1,4-dioxo-1,2,3,4-tetrahydro-pyridazino[4,5-b]carbazoles. Further chemical manipulations, such as chlorodehydroxylation and nucleophilic substitution, led to the formation of 1,4-dichloropyridazino[4,5-b]carbazoles and 1,4-dialkoxy pyridazino[4,5-b]carbazoles. Despite efforts to improve solubility for pharmacological testing through chemical modifications, the tested compounds did not show significant cytotoxic activity. The chemicals used in the process included various carbazole derivatives, hydrazine, phosphorus oxychloride, and alkoxides like sodium methoxide and sodium ethoxide. The conclusions were that the synthesized pyridazino[4,5-b]carbazoles lacked significant antitumor activity in vivo, possibly due to insufficient solubility of the tested substances.