Products Categories
| CAS No.: | 320-51-4 |
|---|---|
| Name: | 4-Chloro-alpha,alpha,alpha-trifluoro-m-toluidine |
| Article Data: | 35 |
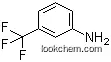
| Molecular Structure: | |
|
|
|
| Formula: | C7H5ClF3N |
| Molecular Weight: | 195.572 |
| Synonyms: | m-Toluidine,4-chloro-a,a,a-trifluoro-(6CI,7CI,8CI);(4-Chloro-3-trifluoromethylphenyl)amine;2-Chloro-5-aminobenzotrifluoride;3-(Trifluoromethyl)-4-chloroaniline;3-Amino-6-chlorobenzotrifluoride;4-Chloro-3-(trifluoromethyl)aniline;4-Chloro-3-(trifluoromethyl)benzenamine;4-Chloro-a,a,a-trifluoro-m-toluidine;NSC 61405; |
| EINECS: | 206-277-6 |
| Density: | 1.425 g/cm3 |
| Melting Point: | 34-38 °C(lit.) |
| Boiling Point: | 242.8 °C at 760 mmHg |
| Flash Point: | >110 °C |
| Appearance: | White to pinkish crystalline powder |
| Hazard Symbols: |
 Xi, Xi,  T T
|
| Risk Codes: | 36/37/38-23/24/25 |
| Safety: | 22-24/25-45-36-26 |
| Transport Information: | UN 2811 |
| PSA: | 26.02000 |
| LogP: | 3.52220 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; TPGS-750-M In tetrahydrofuran; water at 20℃; for 1h; Catalytic behavior; Reagent/catalyst; | 98% |
| With sodium tetrahydroborate In tetrahydrofuran; water at 20℃; for 2h; Inert atmosphere; Green chemistry; chemoselective reaction; | 94% |
| With sodium tetrahydroborate; iron; water at 20℃; for 2h; | 94% |

| Conditions | Yield |
|---|---|
| With copper dichloride; 1-hexyl-3-methylimidazol-1-ium chloride at 40℃; for 8h; regioselective reaction; | 94% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; potassium phosphate; copper(l) iodide; N1,N2-bis(5-methyl-[1,1'-biphenyl]-2-yl)oxalamide In water; dimethyl sulfoxide at 110℃; for 24h; Inert atmosphere; | 84% |
| Multi-step reaction with 2 steps 1: hydrazine hydrate; pyridine / ethyl acetate; dichloromethane / 4 h / 130 °C / Autoclave 2: Raney nickel / methanol / 1 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: 5-bromo-2-chlorobenzotrifluoride With magnesium In tetrahydrofuran Inert atmosphere; Stage #2: With C10H17NO In tetrahydrofuran; toluene at -45℃; for 2h; Inert atmosphere; Stage #3: With ammonium chloride In tetrahydrofuran; water; toluene Inert atmosphere; | 61% |
- 99586-65-9
4-chloropicolinamide

- 123-30-8
4-amino-phenol

A
- 284462-80-2
4-(4-aminophenoxy)-2-pyridinecarboxamide

B
- 320-51-4
4-chloro-3-trifluoromethyl-aniline

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; sodium chloride In methanol; hexane; ethyl acetate; N,N-dimethyl-formamide | A 58% B n/a |
| With potassium tert-butylate; sodium chloride In methanol; hexane; ethyl acetate; N,N-dimethyl-formamide | A 58% B n/a |
| With potassium tert-butylate; sodium chloride In methanol; hexane; ethyl acetate; N,N-dimethyl-formamide | A 58% B n/a |

- 1198619-79-2
1-(4-bromophenyl)-3-(4-chloro-3-(trifluoromethyl)phenyl)-5-thioxoimidazolidine-2,4-dione

- 100-46-9
benzylamine

A
- 1198619-80-5
3-benzyl-5-(benzylimino)-1-(4-bromophenyl)imidazolidine-2,4-dione

B
- 320-51-4
4-chloro-3-trifluoromethyl-aniline

| Conditions | Yield |
|---|---|
| for 1h; Reflux; | A 51% B n/a |


| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 83℃; for 24h; Irradiation; further reagents, further conditions; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 60 percent / HNO3; H2SO4 / 0.5 h / 50 - 60 °C 2: 65 percent / NaSH*9H2O; NaHCO3 / methanol; H2O / 1 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: HNO3, H2SO4 2: H2 / PtO2 View Scheme | |
| Multi-step reaction with 2 steps 1: nitric acid; sulfuric acid / 1 h / Heating 2: palladium 10% on activated carbon; hydrogen / methanol / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: acetic anhydride; nitric acid / 3 h / 10 - 12 °C 2: iron(III) chloride hexahydrate; hydrazine hydrate / ethanol / 3 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: sulfuric acid; nitric acid / water / 18 h / 0 - 20 °C / Inert atmosphere; Schlenk technique 2: iron; ammonium chloride; hydrogenchloride / ethanol; water / 2 h / 80 °C / Inert atmosphere; Schlenk technique View Scheme |
- 445-01-2
5-bromo-2-chlorobenzotrifluoride

A
- 1445972-32-6
C14H7Cl2F6N

B
- 320-51-4
4-chloro-3-trifluoromethyl-aniline

| Conditions | Yield |
|---|---|
| With dicyclohexyl(2',4',6'-triisopropyl-5-methoxy-3,4,6-trimethyl-[1,1'-biphenyl]-2-yl)phosphine; C50H70NO4PPdS; C50H70NO4PPdS; dicyclohexyl(2',4',6'-triisopropyl-4-methoxy-3,5,6-trimethyl-[1,1'-biphenyl]-2-yl)phosphine; ammonia; sodium t-butanolate In 1,4-dioxane at 100℃; for 24h; Inert atmosphere; Overall yield = 72 %; Overall yield = 140.9 mg; |
- 755037-03-7
regorafenib

A
- 757251-39-1
4-(4-amino-3-fluorophenoxy)pyridine-2-carboxylic acid methyl amide

B
- 320-51-4
4-chloro-3-trifluoromethyl-aniline

| Conditions | Yield |
|---|---|
| In water; acetonitrile at 80℃; for 72h; |
- 107-96-0Propanoicacid, 3-mercapto-
- 7048-04-6L-Cysteine hydrochloride monohydrate
- 192725-17-0Lopinavir
- 75-98-9Pivalic acid
- 5173-46-6Estra-4,9-diene-3,17-dione
- 2386-87-03,4-Epoxycyclohexylmethyl 3,4-epoxycyclohexanecarboxylate
- 557-04-0Octadecanoicacid, magnesium salt (2:1)
- 5451-40-12,6-Dichloropurine
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin

What can I do for you?
Get Best Price
Specification
The 5-Amino-2-chlorobenzotrifluoride with CAS registry number of 320-51-4 is also known as 3-(Trifluoromethyl)-4-chloroaniline. The IUPAC name is 4-Chloro-3-(trifluoromethyl)aniline. It belongs to product categories of Amines and Anilines; Aromatic Halides (substituted); Aniline Series. Its EINECS registry number is 206-277-6. In addition, the formula is C7H5ClF3N and the molecular weight is 195.57. Besides, this chemical is a white to pinkish crystalline powder.
Physical properties about 5-Amino-2-chlorobenzotrifluoride are: (1)XLogP3: 3.5; (2)H-Bond Donor: 1; (3)H-Bond Acceptor: 4; (4)Exact Mass: 195.006261; (5)MonoIsotopic Mass: 195.006261; (6)Topological Polar Surface Area: 26; (7)Heavy Atom Count: 12; (8)Complexity: 159; (9)Covalently-Bonded Unit Count: 1.
Uses of 5-Amino-2-chlorobenzotrifluoride: it is used as medicine and pesticide intermediate. What's more, it is used to produce 1,3-bis(4-chloro-3-(trifluoromethyl)phenyl)triazene. The reaction occurs with reagent SHNC and solvent H2O at ambient temperature for 25 hours. The yield is about 99%.

When you are using this chemical, please be cautious about it. As a chemical, it is irritating to eyes, respiratory system and skin. What's more, it is toxic by inhalation, in contact with skin and if swallowed. During using it, wear suitable protective clothing. Avoid contact with skin and eyes and do not breathe dust. If contact with eyes accidently, rinse immediately with plenty of water and seek medical advice. In case of accident or if you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: C1=CC(=C(C=C1N)C(F)(F)F)Cl
2. InChI: InChI=1S/C7H5ClF3N/c8-6-2-1-4(12)3-5(6)7(9,10)11/h1-3H,12H2
3. InChIKey: ASPDJZINBYYZRU-UHFFFAOYSA-N