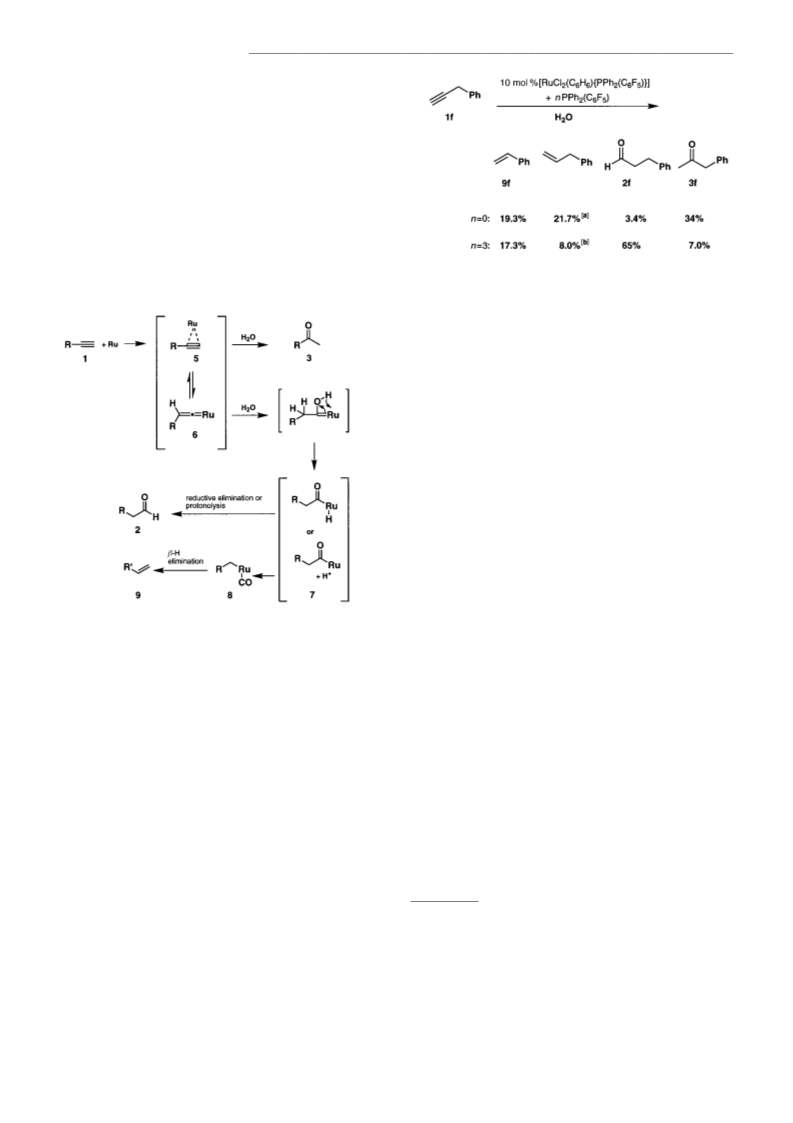
COMMUNICATIONS
[
4] a) J. Halpern, B. R. James, A. L. W. Kemp, J. Am. Chem. Soc. 1961, 83,
tions. Compound 1 turned out to be more susceptible towards
oxidizing agents; initially, however, the tetrahedrane could
only be oxidized to complex product mixtures. We have now
found that upon reaction with iodine in equimolar amounts at
room temperature, 1 quantitatively yields a compound with
the molecular formula R*Si I , whose structure, reactivity,
4097; b) J. Halpern, B. R. James, A. L. W. Kemp, J. Am. Chem. Soc.
1966, 88, 5142; c) M. M. Taqui Khan, S. B. Halligudi, S. Shukla, J. Mol.
Cat. 1990, 58, 299.
[
[
5] J. Blum, H. Huminer, H. Alper, J. Mol. Catal. 1992, 75, 153.
6] a) W. Hiscox, P. W. Jennings, Organometallics 1990, 9, 1997; b) J. W.
Hartman, W. C. Hiscox, P. W. Jennings, J. Org. Chem. 1993, 58, 7613.
7] a) N. X. Hu, Y, Aso, T. Otsubo, F. Ogura, Tetrahedron Lett. 1986, 27,
4
4 2
[
and synthesis are described here.
6
099; b) I. K. Meier, J. A. Marsella, J. Mol. Catal. 1993, 78, 31.
8] M. A. Bennett, A. K. Smith, J. Chem. Soc. Dalton Trans. 1974, 233.
9] The complex [RuCl (PPh (C )) ] was prepared separately and
As long as migrations of the supersilyl groups during the
iodination of R*Si are excluded, the four isomeric structures
[
[
2
2
6
F
5
3
4
4
showed similar catalytic activity without added phosphane. The
catalytic activity, however, was lower than that of the catalyst system
of method A.
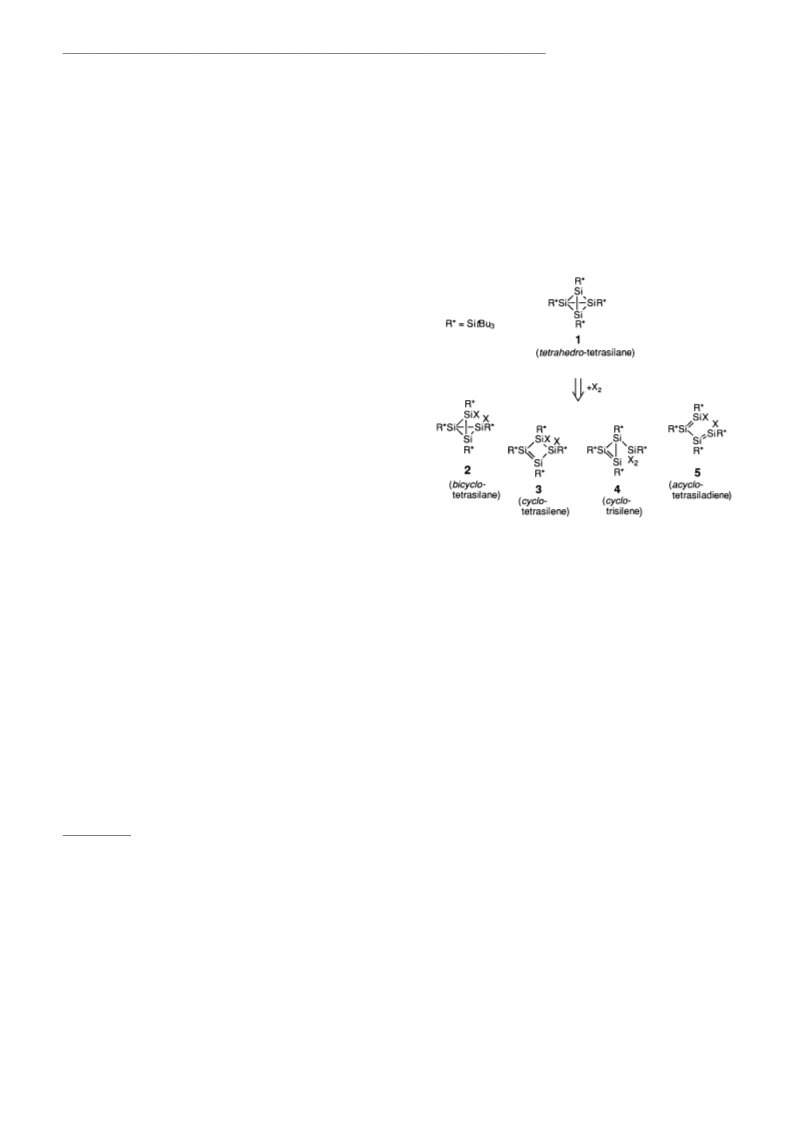
2 ± 5 (X I) are possible for R*Si I (Scheme 1). A bicyclo-
4
4 2
tetrasilane of type 2 was first proven by Masamune et al. in the
[
[
10] W. A. Herrmann, C. W. Kohlpaintner, Angew. Chem. 1993, 105, 1588;
Angew. Chem. Int. Ed. Engl. 1993, 32, 1524.
11] a) O. M. Abu Salah, M. I. Bruce, J. Chem. Soc. Dalton Trans. 1974,
2302; b) M. I. Bruce, A. G. Swincer, Aust. J. Chem. 1980, 33, 1471;
c) B. P. Sullivan, R. S. Smythe, E. M. Kober, T. J. Meyer, J. Am. Chem.
Soc. 1982, 104, 4701; d) C. Mountassir, T. B. Hadda, H. L. Bozec, J.
Organomet. Chem. 1990, 388, C13; e) S. G. Davies, J. P. Mcnally, A. J.
Smallridge, Adv. Organomet. Chem. 1990, 30, 1; f) W. Knaup, H.
Werner, J. Organomet. Chem. 1991, 411, 471; g) C. Bianchini, J. A.
Casares, M. Peruzzini, A. Romerosa, F. Zanobini, J. Am. Chem. Soc.
1996, 118, 4585; h) G. Poignant, F. Martin, V. Guerchais, Synlett 1997,
913.
[
[
[
12] M. I. Bruce, Chem. Rev. 1991, 91, 197; R. Mah e , Y. Sasaki, C. Bruneau,
P. H. Dixneuf, J. Org. Chem. 1989, 54, 1518; B. M. Trost, G. Dyker,
R. J. Kulawiec, J. Am. Chem. Soc. 1990, 112, 7809; C. A. Merlic, M. E.
Pauly J. Am. Chem. Soc. 1996, 118, 11319.
13] N. M. Kosti c , R. F. Fenske, Organometallics 1982, 1, 974; Y. Wakatsu-
ki, N. Koga, H. Yamazaki, K. Morokuma, J. Am. Chem. Soc. 1994, 116,
8105; Y. Wakatsuki, N. Koga, H. Werner, K. Morokuma, J. Am. Chem.
Soc. 1997, 119, 360.
14] Y. Wakatsuki, H. Yamazaki, N. Kumegawa, T. Satoh, J. Y. Satoh, J.
Am. Chem. Soc. 1991, 113, 9604.
Scheme 1. Possible structural isomers for the molecular formula R*
Si X .
4 4 2
form of the pale yellow species tBu (2,6-Et C H ) Si (the tBu
2
2
6
3
4
4
groups are located on the central Si atoms of the ªSi
4
butterflyº).[ We prepared a further species of type 2,
2]
[
3]
colorless R*H Si (X H in 2). The deep orange, air-
4
2
4
Diiodotetrasupersilylcyclotetrasilene
sensitive (tBuMe Si) Si obtained by Kira et al. in low yields
2
6
4
(
tBu Si) Si I ÐA Molecule Containing an
3
4
4 2
[4]
represents a cyclo-tetrasilene of type 3. Upon irradiation, it
is supposed to rearrange to a red-brown bicyclo-tetrasilane
isomer 2, which in the dark slowly reverts to 3. The red-brown
Unsaturated Si Ring**
4
Nils Wiberg,* Harald Auer, Heinrich Nöth,
Jörg Knizek, and Kurt Polborn
species (2,4,6-iPr
C H ) Si , recently synthesized by Weiden-
3
6 2 6 4
bruch et al.,[ represents a tetrasiladiene of type 5. A cyclo-
5]
A few years ago we were able to synthesize the first
trisilene of type 4 has so far only been obtained as the
molecular silicon compound containing a Si tetrahedron, the
homologous germanium compound R
4
3
(Ge instead of Si
4
and R* instead of SiX
R*
Si
R* in 4).[
6]
orange tetrasupersilyl-tetrahedro-tetrasilane R*Si (1; R*
4
4
2
[
1]
tri-tert-butylsilyl, SitBu , supersilyl). We noted at the time
4
4
I
2
crystallizes from benzene at room temperature in
3
that 1 is very stable towards water, air, light, and high
temperatures and cannot be reduced by sodium even in the
presence of [18]crown-6 in benzene under standard condi-
the form of red-orange, light- and moisture-sensitive platelets
which are stable in air. They melt with decomposition at 128 ±
1308C. The structure of this compound is presented in
Figure 1. It was obtained by an X-ray structure analysis of a
[
*] Prof. Dr. N. Wiberg, Dipl.-Chem. H. Auer, Prof. Dr. H. Nöth,
Dipl.-Chem. J. Knizek, Dr. K. Polborn
crystal (monoclinic, space group P2 /c) that also contained
1
[7]
one molecule of benzene per molecule of R*Si I . Accord-
4
4 2
Institut für Anorganische Chemie der Universität
Meiserstrasse 1, D-80333 München (Germany)
Fax: (49)-89-5902-578
ing to the analysis, R*Si I exhibits the structure 3 (X I) and
4
4 2
can be termed 1,2-diiodo-1,2,3,4-tetrasupersilylcyclotetrasil-3-
ene. The central structural element, which has not been
[
**] Compounds of Silicon, Part 124. Unsaturated Compounds of Silicon
and Its Homologues, Part 53. Sterically Overloaded Compounds of
Silicon, Part 17. This research was supported by the Deutsche
Forschungsgemeinschaft. Parts 123 and 52: N. Wiberg, S. Wagner,
S.-K. Vasisht, Chem. Eur. J., in press; part 16: N. Wiberg, K.
Amelunxen, H.-W. Lerner, H. Nöth, J. Knizek, I. Krossing, Z.
Naturforsch. B. 1998, 53, 333.
observed before, is a bent unsaturated Si ring with iodine
4
substituents (angle between the planes Si1-Si2-Si3/Si3-Si4-Si1
2
2
7.88 and Si2-Si3-Si4/Si4-Si1-Si2 28.18). The SiSi distance is
.257(2) and is therefore similar to that in the disilene
(iPr Si) Si (2.251(1) ), which carries very bulky substitu-
3
4
2
Angew. Chem. Int. Ed. 1998, 37, No. 20
ꢀ WILEY-VCH Verlag GmbH, D-69451 Weinheim, 1998
1433-7851/98/3720-2869 $ 17.50+.50/0
2869

 Tokunaga, Makoto
Tokunaga, Makoto
 Wakatsuki, Yasuo
Wakatsuki, Yasuo