Communication
ChemComm
Notes and references
1
(a) V. E. Ziffle and S. P. Fletcher, Reduction of Saturated Alkyl
Halides to Alkanes, in Comprehensive Organic Synthesis, ed. G. A.
Molander and P. Knochel, Elsevier, Oxford, 2nd edn, 2014, vol. 8,
pp. 999–1010; (b) J. D. Nguyen, E. M. D’Amato and C. R. J.
Stephenson, Reduction of Alkenyl Halides to Alkenes, and of Aryl
Halides and Related Compounds to Arenes, in Comprehensive
Organic Synthesis, ed. G. A. Molander and P. Knochel, Elsevier,
Oxford, 2nd edn, 2014, vol. 8, pp. 1123–1142.
2
3
S. Harrad, M. Robson, S. Hazrati, V. S. Baxter-Plant, K. Deplanche,
M. D. Redwood and L. E. Macaskie, J. Environ. Monit., 2007, 9, 314.
(a) H. G. Kuivila, Synthesis, 1970, 499; (b) C. Chatgilialoglu,
K. U. Ingold and J. C. Scaiano, J. Am. Chem. Soc., 1981, 103, 7739.
Fig. 1 DFT calculation of the reaction mechanism and partial potential
energy surface of the black pathway and the grey pathway. Some
intermediates are omitted. The Gibbs free energies of PhBr (GS) and
4 For selected reviews, please see: (a) F. Alonso, I. P. Beletskaya and
M. Yus, Chem. Rev., 2002, 102, 4009; (b) M. Szostak, M. Spain and
D. J. Procter, Chem. Soc. Rev., 2013, 42, 9155. For selected examples,
please see: (c) A. Zask and P. Helquist, J. Org. Chem., 1978, 43, 1619;
ꢁ1
isopropanol were set to 0.0 kcal mol as reference. Distances are in Å.
(
d) M. Narisada, I. Horibe, F. Watanabe and K. Takeda, J. Org. Chem.,
1989, 54, 5308; (e) C. A. Marques, M. Selva and P. Tundo, J. Org.
Chem., 1994, 59, 3830; ( f ) P. A. Baguley and J. C. Walton, Angew.
Chem., Int. Ed., 1998, 37, 3072; (g) K.-I. Fujita, M. Owaki and
R. Yamaguchi, Chem. Commun., 2002, 2964; (h) A. Bhattacharjya,
P. Klumphu and B. H. Lipshutz, Org. Lett., 2015, 17, 1122; (i) Q. Liu,
B. Han, W. Zhang, L. Yang, Z.-L. Liu and W. Yu, Synlett, 2005,
hydrogen bond of TS1, there is apparent charge separation (large
red and blue clouds), leading to large dipole moment. In contrast,
0
there is less charge separation in TS1 (please see ESI,† Fig. S2).
In conclusion, we have developed a simple and clean metal-
free reduction of C–X bond under air at room temperature.
Isopropanol was used as both reducing reagent and solvent.
A wide range of functional groups, such as acids, esters,
alcohols, anilines, phenols, indoles, pyridines, cyano group,
trifluoromethyl group and heterocyclic compounds, were all
tolerated. Different organic halides comprising C–I, C–Br and
C–Cl bonds were reduced with good to excellent yields. Poly-
halides were also chemoselectively reduced. DFT calculation
showed that a six-member ring transition state containing
C–Brꢀ ꢀ ꢀH–O hydrogen bonding has lower energy. This strategy
provides greener approach for C–halogen bond reduction and
detoxification of environmentally hazardous organic halides.
We thank the Recruitment Program of Global Experts (Short-
Term B) to C.-J. L., the Fundamental Research Funds for
the Central Universities (lzujbky-2018-62, lzujbky-2018-79),
the International Joint Research Centre for Green Catalysis
and Synthesis, Gansu Provincial Sci. & Tech. Department (No.
2248.
5 (a) A. Dewanji, C. M u¨ ck-Lichtenfeld and A. Studer, Angew. Chem., Int.
Ed., 2016, 55, 6749; (b) H.-X. Zheng, X.-H. Shan, J.-P. Qu and
Y.-B. Kang, Org. Lett., 2017, 19, 5114; (c) W. Liu and F. Hou,
Tetrahedron, 2017, 73, 931.
6 D. Y. Ong, C. Tejo, K. Xu, H. Hirao and S. Chiba, Angew. Chem.,
Int. Ed., 2017, 56, 1840.
(a) J. M. R. Narayanam, J. W. Tucker and C. R. J. Stephenson, J. Am.
Chem. Soc., 2009, 131, 8756; (b) J. D. Nguyen, E. M. D’Amato,
J. M. R. Narayanam and C. R. J. Stephenson, Nat. Chem., 2012,
7
4
, 854; (c) H. Kim and C. Lee, Angew. Chem., Int. Ed., 2012, 51, 12303;
d) J. J. Devery, J. D. Nguyen, C. Dai and C. R. J. Stephenson,
ACS Catal., 2016, 6, 5962.
8 (a) I. Ghosh, T. Ghosh, J. I. Bardagi and B. K o¨ nig, Science, 2014,
46, 725; (b) E. H. Discekici, N. J. Treat, S. O. Poelma, K. M. Mattson,
(
3
Z. M. Hudson, Y. Luo, C. J. Hawker and J. R. de Alaniz, Chem.
Commun., 2015, 51, 11705; (c) S. O. Poelma, G. L. Burnett,
E. H. Discekici, K. M. Mattson, N. J. Treat, Y. Luo, Z. M. Hudson,
S. L. Shankel, P. G. Clark, J. W. Kramer, C. J. Hawker and J. Read de
Alaniz, J. Org. Chem., 2016, 81, 7155.
S. Fukuzumi, K. Hironaka and T. Tanaka, J. Am. Chem. Soc., 1983,
105, 4722.
0 H. Jiang, J. R. Bak, F. J. L o´ pez-Delgado and K. A. Jørgensen, Green
Chem., 2013, 15, 3355.
9
1
2016B01017, 18JR3RA284, 18JR4RA003) and Lanzhou University
1
1 T. Kawamoto and I. Ryu, Org. Biomol. Chem., 2014, 12, 9733.
for support of our research. We also thank the Canada Research 12 T. Fukuyama, Y. Fujita, H. Miyoshi, I. Ryu, S.-C. Kao and Y.-K. Wu,
Chem. Commun., 2018, 54, 5582.
Chair (Tier I) foundation, the E. B. Eddy endowment fund, the
1
3 (a) H. Meerwein and R. Schmidt, Justus Liebigs Ann. Chem., 1925,
44, 221. For selected reviews, please see: (b) J. M. Clay, Meerwein-
CFI, NSERC, and FQRNT to C.-J. L. We thank Dr Haining Wang for
his helpful discussions about the DFT calculation and Mr Shuo
Zhang in this group for reproducing the results presented substrates
4
Ponndorf-Verley reduction, in Name Reactions for Functional Group
Transformations, ed. J. J. Li and E. J. Corey, John Wiley & Sons,
hoboken, NJ, 2007, pp. 123–128; (c) C. F. de Graauw, J. A. Peters,
H. van Bekkum and J. Huskens, Synthesis, 1994, 1007.
4 P. Boffetta, K. A. Mundt, H.-O. Adami, P. Cole and J. S. Mandel, Crit.
Rev. Toxicol., 2011, 41, 622.
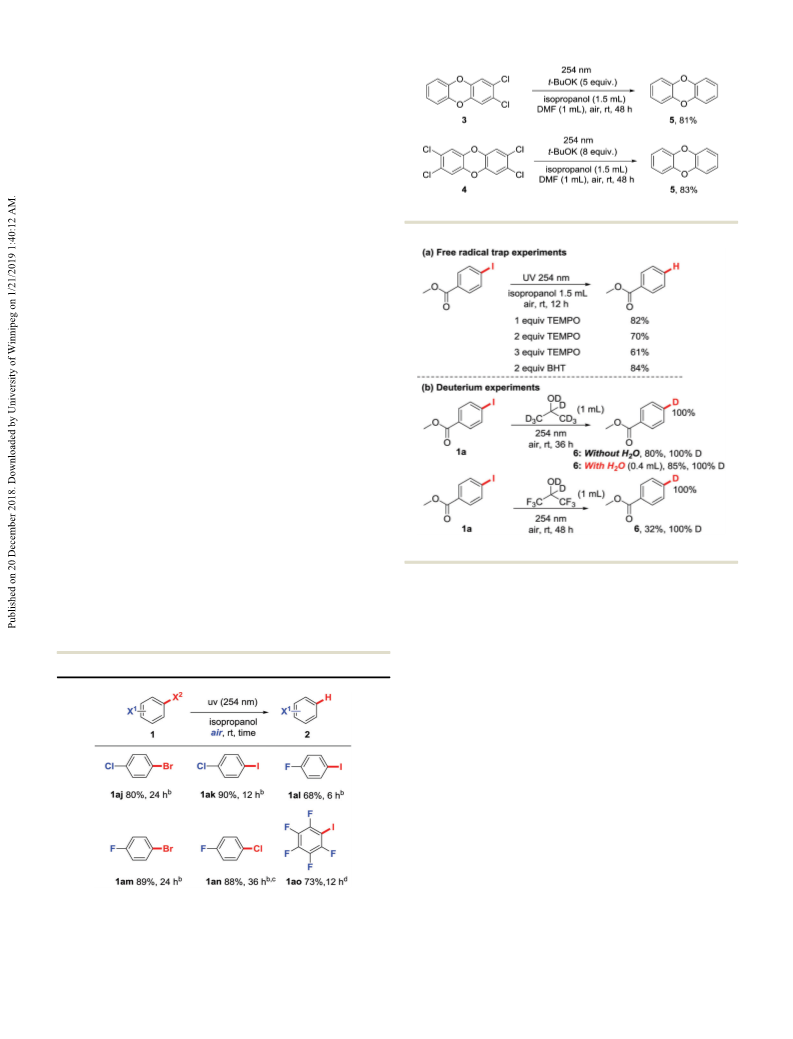
1r, 1v and 1ag in Table 2 and substrate 1aj in Table 3.
1
Conflicts of interest
1
1
5 Please see ESI† for detail.
6 Y. Zhao and D. G. Truhlar, Theor. Chem. Acc., 2008, 120, 215.
There are no conflicts to declare.
17 J. Tomasi, B. Mennucci and R. Cammi, Chem. Rev., 2005, 105, 2999.
770 | Chem. Commun., 2019, 55, 767--770
This journal is ©The Royal Society of Chemistry 2019

 Cao, Dawei
Cao, Dawei
 Yan, Chaoxian
Yan, Chaoxian
 Zhou, Panpan
Zhou, Panpan
 Zeng, Huiying
Zeng, Huiying
 Li, Chao-Jun
Li, Chao-Jun