4
6
Y. Ding, W. Zhao / Journal of Molecular Catalysis A: Chemical 337 (2011) 45–51
Table 1
Oxidation of 2-picoline under different solvents catalyzed by [(C18H37)2(CH3)2N]7[PW11O39] with hydrogen peroxide.
a
Entry
Solvent
Solubility of catalyst
Yieldb (%)
◦
◦
During reaction (65 C)
After reaction (25 C)
1c
2
3
4
5
6
7
8
1,4-Dioxane
1,4-Dioxane
Ethanol
Acetonitrile
Acetone
Ethyl acetate
Toluene
1,2-Dichloroethane
–
–
9
98
59
85
80
93
94
98
Soluble
Soluble
Soluble
Soluble
Soluble
Soluble
Soluble
Insoluble
Insoluble
Insoluble
Insoluble
Insoluble
Soluble
Soluble
a
◦
Reaction conditions: 1 mmol substrate, 5 mmol (30% aq.) H2O2, 8 mol catalyst, 2 ml solvent, 65 C, 3 h.
b
c
Yields were determined by GC analysis. Selectivity to the N-oxide was ≥99%. Assignments of products were analyzed by 1H NMR and 13C NMR.
Without catalyst.
ion, ([(C18H37) (CH ) N]7[PW11O39]), and it has temperature-
nal diameter = 0.25 mm, film thickness = 0.25 m, length = 30 m).
Chemical elemental analysis of the catalysts was done on an ICP-
atomic emission spectrometer (IRIS ER/S), and C, H, and N contents
were measured on a German Elementar Vario EL spectrometer.
2
3 2
responsive behavior in a biphasic mixture of aqueous H O2 and
2
1
,4-dioxane. The catalyst was insoluble in the system at room tem-
perature, but dissolved with the elevation of reaction temperature.
After the reaction finished with cooling, the catalyst precipitated
gradually from the system itself. The catalyst effectively catalyzed
the oxidation and could be easily recovered and reused.
2.4. Catalytic reaction
Catalyst (8 mol), 1,4-dioxane (2–3 ml), substrate (1 mmol), and
2
. Experimental
H O (5 mmol, 30% aq.) were charged in the reaction flask. The
2 2
reaction was carried out at proper temperature for 3–7 h. After reac-
tion finished with temperature dropping, the catalyst precipitated
itself from the solvent. The catalyst was separated by centrifu-
2.1. Reagents
All chemicals were analytical grade, commercially available and
gation and washed with Et O, which was then used for the next
2
used without further purification unless otherwise stated.
oxidation. The organic products were separated from the aqueous
phase by extraction and then the organic layer was analyzed by GC
with the internal standard method. Assignments of some products
were made by comparison with authentic samples. Selective prod-
ucts were obtained by evaporation or column chromatography and
analyzed by IR, melting point, 1H NMR and C NMR.
2
.2. Preparation of the catalyst [(C18H37) (CH ) N]7[PW11O39
]
2
3 2
First 30 g of Na WO ·2H O was dissolved in 37 ml of distilled
2
4
2
13
water with stirring. Then 0.75 ml of 85% H PO , followed by 5.5 ml
3
4
of glacial acetic acid, was added to the stirring solution. After a
few seconds the solution became cloudy and after about 1 min a
heavy white precipitate had formed. The solid was collected and
first dried under aspiration for 24 h, and then the aspirated solid
3
. Results and discussion
3.1. Pyridine oxidation
◦
was left in air for 24 h [41]. After that, the solid was dried at 140 C
for about 1.5 h. Then, 0.56 g of the dried solid was dissolved in 55 ml
of water. To this solution, 0.9 g of dioctadecyl dimethyl ammonium
chloride dissolved in 10 ml of tert-butanol was added slowly. The
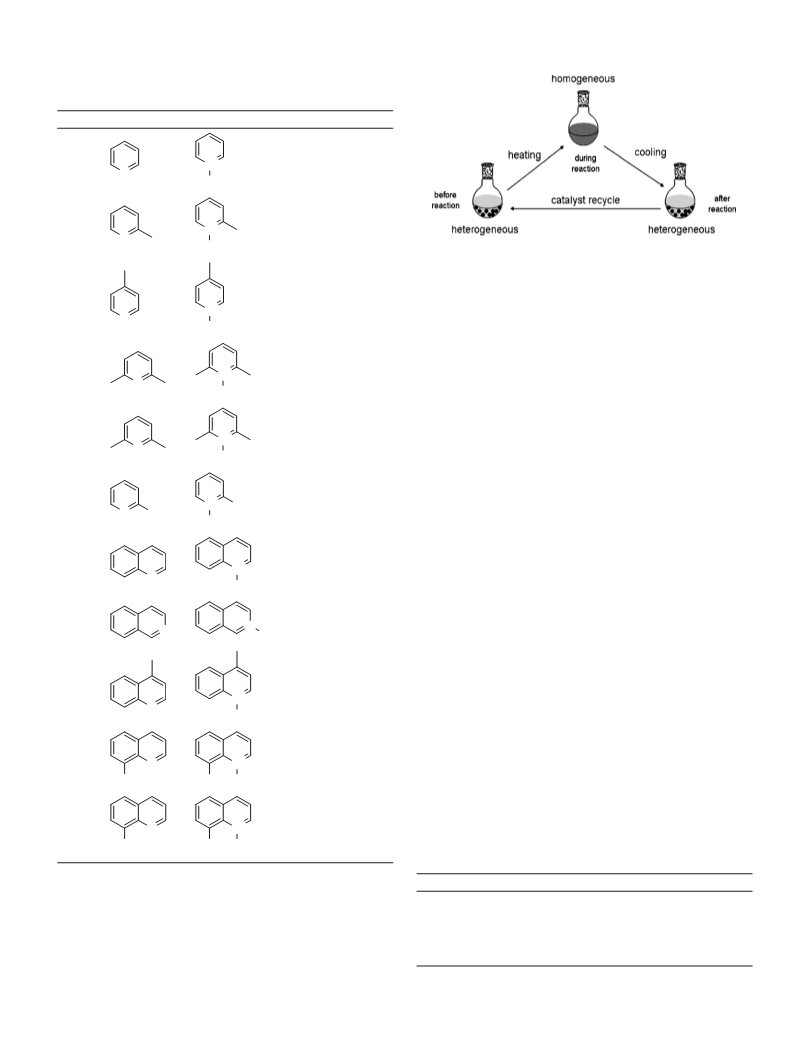
Using the temperature-controlled phase transfer catalyst, we
investigated the effect of the solvent on temperature phase trans-
formation for the oxidation of 2-picoline with hydrogen peroxide
◦
mixture was stirred vigorously for 4–5 h at 40 C. A white solid was
(
Table 1). Although the catalyst of [(C18H37) (CH ) N]7[PW11O39]
2 3 2
filtered off and then washed with an excess amount of water, then
was insoluble in most solvents at room temperature, a homoge-
neous medium was formed in all solvents during the oxidation
−
1
dried in vacuum. The yield was 85%. IR spectrum (KBr, cm ): 1079,
040, 945, 900, 849, 806, 759, 725, 591, 512, 409. 31P MAS NMR:
10.2 ppm. Calcd. for [(C18H37) (CH ) N]7[PW11O39]: C, 48.90; H,
1
◦
at reaction temperature (65 C). After the reaction with cooling,
−
2
3
2
the catalyst precipitated gradually from the system in some sol-
vents, including 1,4-dioxane, ethanol, acetonitrile, acetone and
ethyl acetate. In contrast, the catalyst was still soluble in toluene
and 1,2-dichloroethane when the temperature cooled to the room
temperature after reaction. The catalytic results for the oxidation
of 2-picoline to 2-picoline-N-oxide under different solvents are
also summarized in the table. The oxidation was effectively cat-
alyzed in most solvents investigated in short reaction time, and
the N-oxide was the only product. Especially in 1,4-dioxane and
1,2-dichloroethane, excellent yields of the N-oxide were obtained
(Table 1, entries 2 and 8). Considering that the catalyst could not
be reused in 1,2-dichloroethane, 1,4-dioxane was chosen as the
optimal solvent in this catalytic system. In addition, a control exper-
iment was also conducted in 1,4-dioxane with H O in the absence
8
.58; N, 1.50; P, 0.47; W, 30.98. Found: C, 50.03; H, 8.76; N, 1.47; P,
.43; W, 30.89.
0
2.3. Characterization techniques
Infrared spectra were recorded on a Nicolet FTIR-360 FT-IR
spectrometer. The catalysts were measured using 2–4% (w/w) KBr
pellets prepared by manual grinding. 31P MAS NMR spectra were
recorded at 9.4 T on a Bruker Avance-400 wide bore spectrometer.
31
The P MAS NMR spectra of solid catalyst with high-power proton
decoupling were performed at 161.9 MHz with BB MAS probe head
using 4 mm ZrO2 rotors and 3.8 s pulse and 2 s repetition time
and 4096 scans, with samples spun at 10 kHz and referenced to 85%
H PO . GC analyses were performed on Shimadzu GC-9AM with a
2
2
of catalyst. Only 9% yield of the product was observed; indicating
the strong activity of the catalyst used in this system.
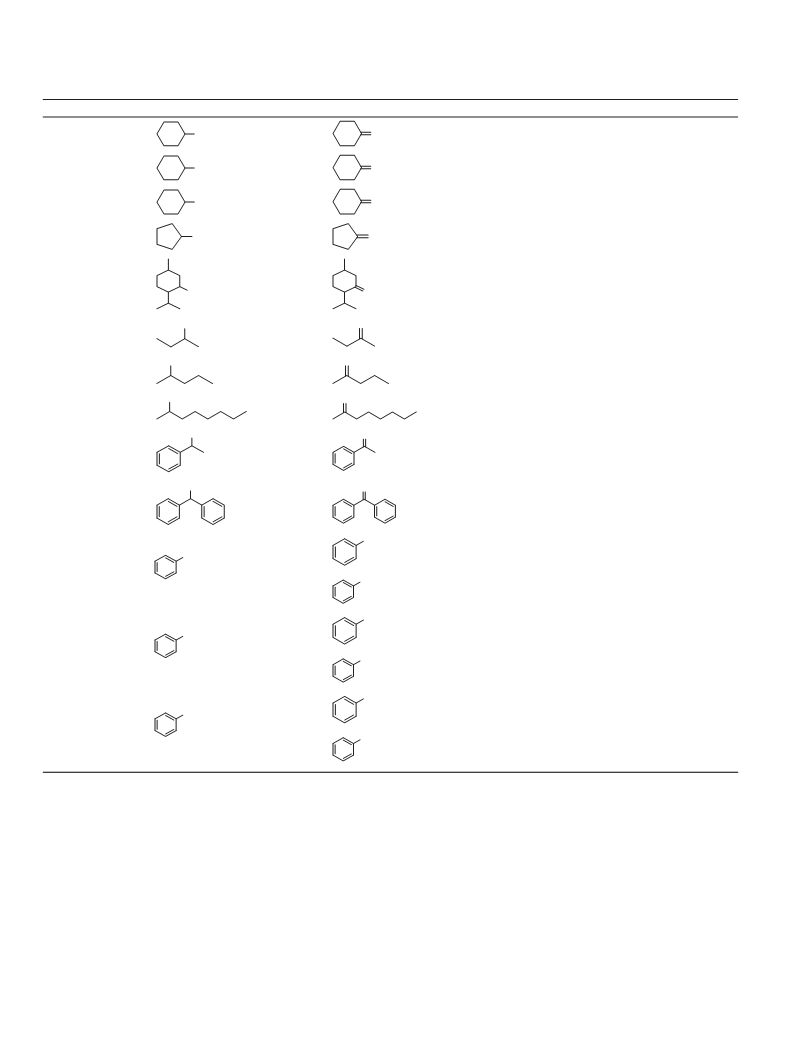
Having obtained these results, we extended our temperature-
controlled phase transfer catalytic system to the oxidation of other
pyridine derivatives. The results are listed in Table 2. The yields
3
4
flame ionization detector equipped with SE-54 capillary (internal
diameter = 0.25 mm, length = 30 m). Mass spectra were recorded on
Finnigan Trace DSQ (Thermo Electron Corporation) at an ionization
voltage of 70 eV equipped with a DB-5 capillary column (inter-

 Ding, Yong
Ding, Yong
 Zhao, Wei
Zhao, Wei