W. Zhang et al. / Journal of Molecular Catalysis A: Chemical 335 (2011) 71–81
81
of K is systematically investigated through several characterization
methods, which can be summarized as follows: (1) K doping has a
great influence on Pd2+ precursor phase and the reduced Pd atom.
TPR data indicates that there exists a strong interaction between K
and Pd2+, which inhibits the reduction of Pd2+ species. The results
of CO-FTIR and H2 chemisorption also suggest that the strong inter-
action still exists between K and the reduced Pd atoms. (2) K doping
has caused the red-shift of CO adsorption, which suggests that more
basic nature of K-doped Al2O3 results in an electron transfer to Pd,
leading to a higher Pd electron density, and ultimately promoting
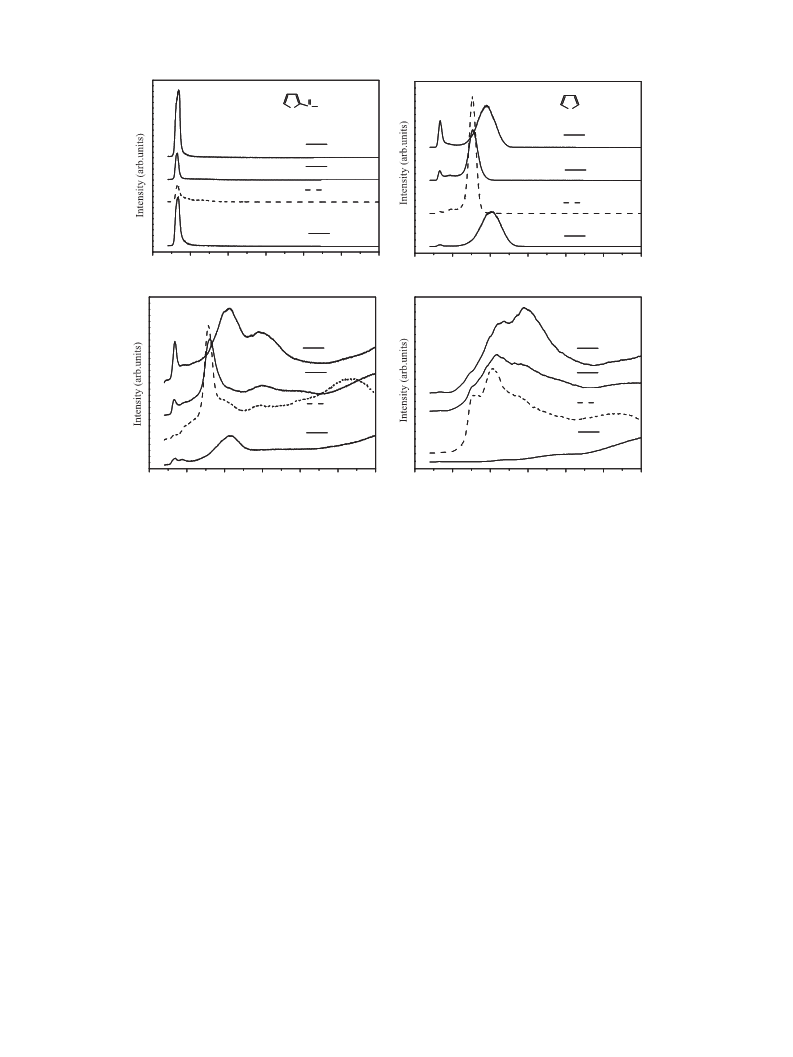
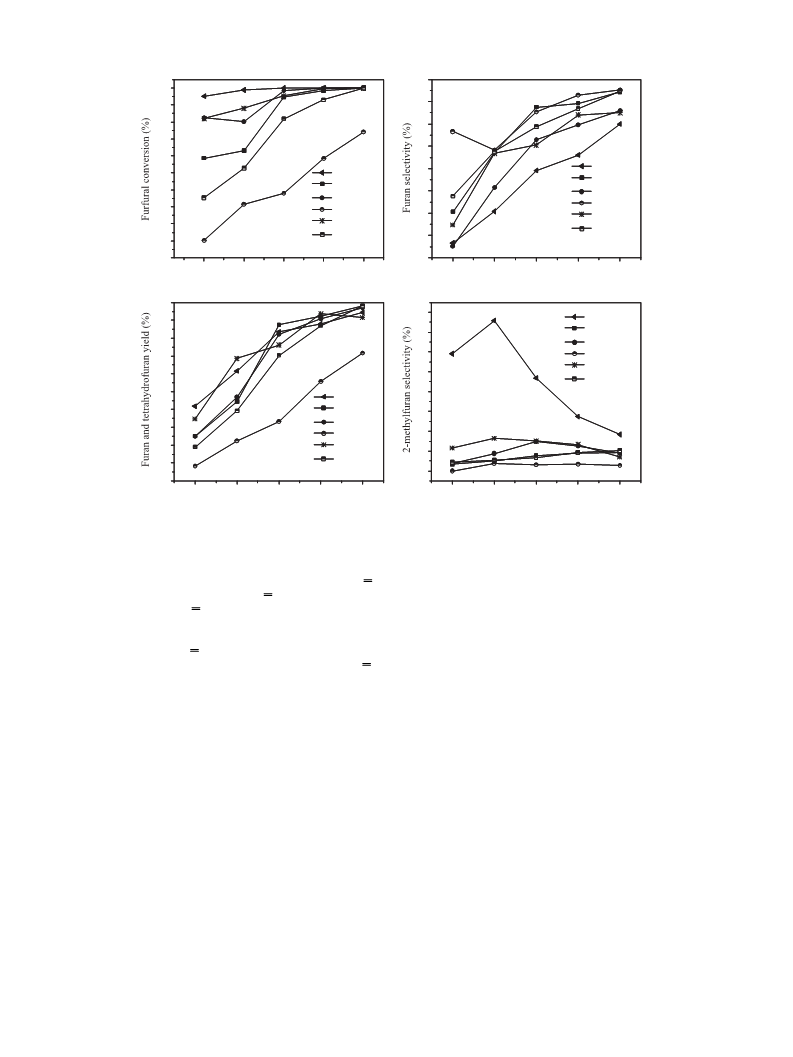
the decarbonylation of furfural. (3) The furfural-TPSR and furfural-
FTIR results indicate that the doping of K can change the adsorption
mode of furfural on Pd catalysts. Finally, it is important to mention
that the decrease of ꢀ2(C,O) configuration might be the main reason
for suppressing hydrogenation of furfural on K-doped Pd catalysts.
[10] P. Lejemble, A. Gaset, Philippe Kalck, Biomass 4 (1984) 263–274.
[11] A.P. Dunlop, G.W. Huffman, US 3,257,417 (1966).
[12] K.J. Jung, A. Gaset, Biomass 16 (1988) 89–96.
[13] R.D. Srivastava, A.K. Guha, J. Catal. 91 (1985) 254–262.
[14] Y. Yang, H.W. Xiang, Y.Y. Xu, L. Bai, Y.W. Li, Appl. Catal. A 266 (2004) 181–194.
[15] R. Pellegrini, G. Leofanti, G. Agostini, L. Bertinetti, S. Bertarione, E. Groppo, A.
Zecchina, C. Lamberti, J. Catal. 267 (2009) 40–49.
[16] G. Neri, G. Rizzo, L. De Luca, A. Donato, M.G. Musolino, R. Pietropaolo, Appl.
Catal. A 356 (2009) 113–120.
[17] A.H. Padmasri, A. Venugopal, J. Krishnamurthy, K.S. Rama Rao, P. Kanta Rao, J.
Mol. Catal. A 181 (2002) 73–80.
[18] P. Paraskevi, I.K. Dimitris, J. Catal. 267 (2009) 57–66.
[19] A.A. Davydov, Infrared Spectroscopy of Adsorbed Species on the Surface of
Transition Metal Oxides, Wiley, New York, 1990.
[20] X.Z. Jiang, Y.H. Sua, B.J. Lee, S.H. Chien, Appl. Catal. A 211 (2001) 47–51.
[21] W.J. Kim, J.H. Kang, I.Y. Ahn, S.H. Moon, Appl. Catal. A 268 (2004) 77–82.
[22] S. Bertarione, D. Scarano, A. Zecchina, V. Johanek, J. Hoffmann, S. Schauermann,
M.M. Frank, J. Libuda, G. Rupprechter, H.J. Freund, J. Phys. Chem. B 108 (2004)
3603.
[23] C. Keresszegi, D. Ferri, T. Mallat, A. Baiker, J. Catal. 234 (2005) 64–75.
[24] B.B. Baeza, I.R. Ramos, A.G. Ruiz, Appl. Catal. A 205 (2001) 227–237.
[25] R. Zanella, C. Louis, S. Giorgio, R. Touroude, J. Catal. 223 (2004) 328–339.
[26] T.B.L.W. Marinelli, S. nabuurs, V. ponec, J. Catal. 151 (1995) 431–438.
[27] A. Daz, D.R. Acosta, J.A. Odriozola, M. Montes, J. Phys. Chem. B 101 (1997)
1782–1790.
Acknowledgement
The authors gratefully thank the financial supports of Natural
Science Foundation of China (No. 20976185).
[28] W.D. Mross, Catal. Rev. 25 (4) (1983) 591–637.
[29] A.J. Urquhart, J.M. Keel, F.J. Williams, R.M. Lambert, J. Phys. Chem. B 107 (2003)
10591–10597.
References
[30] C.P. Huang, J.T. Richardson, J. Catal. 51 (1978) 1–8.
[31] M.P. Kiskinova, Studies in Surface Science and Catalysis, vol. 70, 1992, 149.
[32] G.D. Weatherbee, J.L. Rankin, C.H. Bartholimew, Appl. Catal. 11 (1984) 73.
[33] H. Praliaud, M. Primet, G.A. Matin, Appl. Surf. Sci. 17 (1983) 107.
[34] J.W. Federico, P. Alejandra, T. Samuel, S.T. Mintcho, M.L. Richard, J. Phys. Chem.
B 106 (2002) 5668–5672.
[35] F.L. Leonarda, A.M. Guy, G.D. Giulio, J. Catal. 164 (1996) 322–333.
[36] S. Stolbov, T.S. Rahman, Phys. Rev. Lett. 96 (2006) 186801–186804.
[37] H.P. Yeung, L.P. Geoffrey, Ind. Eng. Chem. Res. 31 (1992) 2.
[38] N.S. Figoli, P.C. L’argeniere, Catal. Today 5 (1989) 403.
[39] F. Figueras, R. Gomez, M. Primet, Adu. Chem. Ser. 121 (1973) 480.
[40] M. Mavrikakis, M.A. Barteau, J. Mol. Catal. A 131 (1998) 135.
[41] J.L. Davis, M.A. Barteau, J. Am. Chem. Soc. 111 (1989) 1782–1792.
[42] M. Bowker, R. Holroyd, N. Perkins, J. Bhantoo, J. Counsell, A. Carley, C. Morgan,
Surf. Sci. 601 (2007) 3651–3660.
[1] A. Corma, S. lborra, A. Velty, Chem. Rev. 107 (2007) 2411–2502.
[2] K.J. Zeitsch, The Chemistry and Technology of Furfural and Its Many By-
products, vol. 13, 1st ed., Elsevier, Amsterdam, 2000.
[3] H. Singh, M. Prasad, R.D. Srivastaval, J. Chem. Technol. Biotechnol. 30 (1980)
293–296.
[4] J. Coca, E.S. Morrondo, J.B. Parra, H. Sastre, React. Kinet. Catal. Lett. 20 (1982)
3–4.
[5] K.J. Jung, A. Gaset, Biomass 16 (1988) 63–76.
[6] P.A. Moshkin, E.A. Preobrazhenskaya, B.B. Berezina, V.E. Markovich, E.G. Kud-
rina, V.P. Papsueva, E.A. Sokolina, Khimiya Oeterotsiklicheskikh Soedinenii. 2
(1966) 3–7.
[7] J. Coca, E.S. Morrondo, H. Sastre, J. Chem. Technol. Biotechnol. 32 (1982)
904–908.
[8] G.M. Whitman, US 2,374,149 (1945).
[9] D.G. Manly, J.P. O’Halloran, US 3,223,714 (1965).
[43] R. Shekhar, M.A. Barteau, R.V. Plank, J.M. Vohs, J. Phys. Chem. B 101 (1997)
7939–7951.

 Zhang, Wei
Zhang, Wei
 Zhu, Yulei
Zhu, Yulei
 Niu, Shasha
Niu, Shasha
 Li, Yongwang
Li, Yongwang