Full Paper
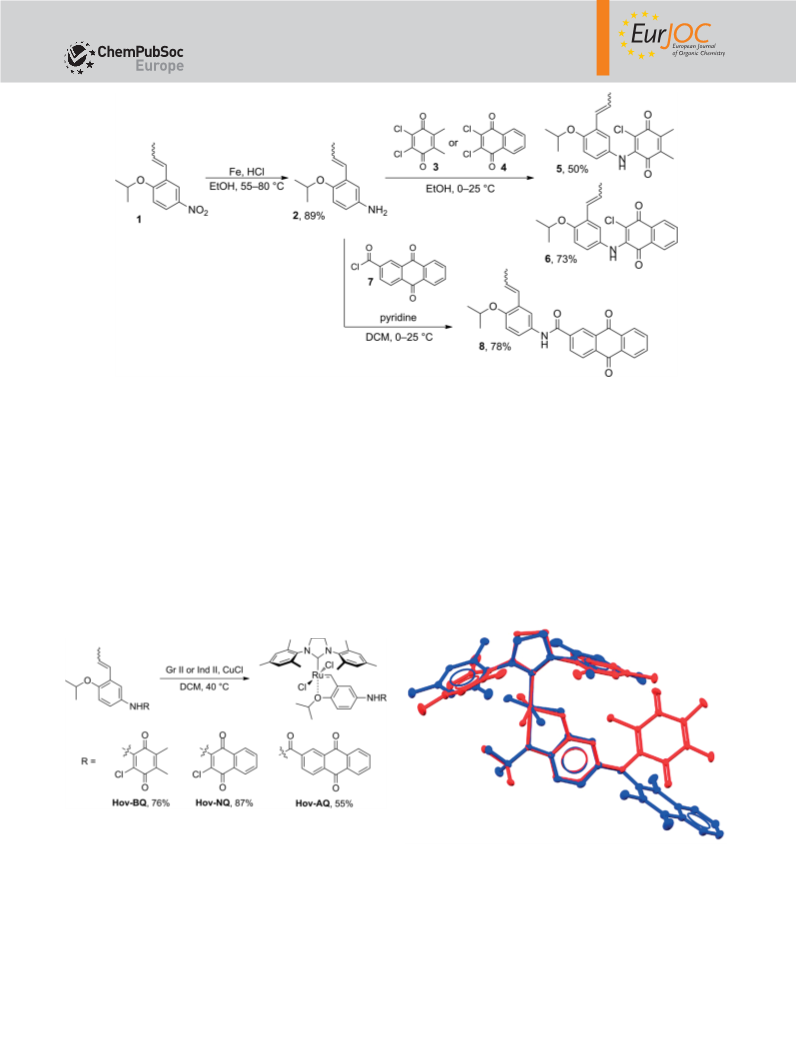
Hov-BQ: This compound was obtained according to the general
procedure by using 2-chloro-3-{[4-isopropoxy-3-(prop-1-en-1-
yl)phenyl]amino}-5,6-dimethylcyclohexa-2,5-diene-1,4-dione (5;
0.12 mmol, 43.2 mg) and Gr II. The product was obtained as purple
were washed with brine, dried with MgSO4 and the organic phase
was concentrated to dryness in vacuo. The residue was purified by
column chromatography (5–20 % EtOAc/c-hex) to obtain the de-
sired product as a white crystalline solid (8.43 g, 89 %). 1H NMR
crystals (0.087 mmol, 70.9 mg, 76 %). 1H NMR (500 MHz, CDCl3): δ = (400 MHz, CDCl3): δ = 5.74–5.86 (m, 2 H), 5.09–5.14 (m, 2 H), 4.16–
16.43 (s, 1 H), 7.38 (s, 1 H), 7.14 (dd, J = 2.7, 8.9 Hz, 1 H), 7.07 (s, 4
H), 6.72 (d, J = 8.8 Hz, 1 H), 6.58 (d, J = 2.5 Hz, 1 H), 4.86 (sept., J =
6.1 Hz, 1 H), 4.17 (s, 4 H), 2.46 (s, 12 H), 2.36 (s, 6 H), 2.13 (s, 3 H),
2.08 (s, 3 H), 1.27 (d, J = 6 Hz, 6 H) ppm. 13C NMR (125 MHz, CDCl3):
δ = 294.6, 210.5, 182.6, 179.5, 149.7, 144.7, 143.9, 139.0, 136.8, 131.9,
129.4, 116.9, 112.1, 110.8, 75.6, 51.5, 31.6, 26.9, 26.2, 22.6, 21.1, 19.4,
4.18 (m, 1 H), 3.68 (dd, J = 2.4, 7.6 Hz, 1 H), 2.77 (dt, J = 2.4, 6.6 Hz,
1 H), 2.30–2.5 (m, 2 H), 1.20 (d, J = 6.3 Hz, 3 H), 0.86 (s, 9 H), 0.06
(s, 6 H) ppm.[65]
Synthesis of (3S,4R)-1,4-Diallyl-3-{(R)-1-[tert-butyl(dimethyl)-
silyloxy]ethyl}azetidin-2-one: (3S,4R)-4-Allyl-3-{(R)-1-[tert-
butyl(dimethyl)silyloxy]ethyl}azetidin-2-one (3.3 mmol, 0.889 g) was
dissolved in DMF (23 mL) in a dried Schlenk tube, followed by so-
dium hydride (6.6 mmol, 0.264 g, 2 equiv.). After 5 min, allyl brom-
ide (9.9 mmol, 0.857 mL, 3 equiv.) was added dropwise and the
mixture was stirred at room temperature for 2 h. The reaction mix-
ture was diluted with methyl tert-butyl ether, washed with brine,
dried with MgSO4 and the organic phase concentrated to dryness
in vacuo. The residue was purified by column chromatography (5–
20 % EtOAc/c-hex) to obtain the desired product as a colourless oil
14.1, 13.4, 12.3 ppm. IR (CH2Cl2 film): ν = 3320, 2918, 1659, 1597,
˜
1485, 1419, 1259, 1220, 1103, 935, 854, 580 cm–1. MS (ESI, CHCl3):
m/z = 809.15 [M]+. C39H44Cl3N3O3Ru (810.22): calcd. C 57.82, H 5.47,
Cl 13.13, N 5.19; found C 57.87, H 5.67, Cl 13.22, N 4.92.
Hov-NQ: This compound was obtained according to the general
procedure by using 2-chloro-3-{[4-isopropoxy-3-(prop-1-en-1-
yl)phenyl]amino}naphthalene-1,4-dione (6; 0.12 mmol, 45.8 mg)
and Gr II. The product was obtained as purple crystals (0.076 mmol,
1
(0.776 g, 76 %). H NMR (500 MHz, CDCl3): δ = 5.82–5.72 (m, 2 H),
1
63.6 mg, 87 %). H NMR (500 MHz, CDCl3): δ = 16.47 (s, 1 H), 8.21
5.26–5.09 (m, 4 H), 4.27 (qd, J = 6.2, 4.6 Hz, 1 H), 3.99 (ddt, J = 15.7,
5.5, 1.5 Hz, 1 H), 3.70 (ddd, J = 7.2, 5.1, 1.1 Hz, 1 H), 3.62 (ddq, J =
15.6, 6.9, 1.0 Hz, 1 H), 2.78 (dd, J = 4.4, 2.1 Hz, 1 H), 2.51–2.45 (m, 1
H), 2.33–2.26 (m, 1 H), 1.18 (d, J = 6.3 Hz, 3 H), 0.88 (s, 9 H), 0.07 (s,
3 H), 0.06 (s, 3 H) ppm.[66]
(d, J = 7.1 Hz, 1 H), 8.13 (d, J = 7.8 Hz, 1 H), 7.78 (dt, J = 1.1, 7.6 Hz,
1 H), 7.73–7.68 (m, 2 H), 7.21 (dd, J = 2.6, 8.7 Hz, 1 H), 7.07 (s, 4 H),
6.76 (d, J = 8.9 Hz, 1 H), 6.65 (d, J = 2.4 Hz, 1 H), 4.88 (sept., J =
6.1 Hz, 1 H), 4.18 (s, 4 H), 2.47 (s, 12 H), 2.35 (s, 6 H), 1.28 (d, J =
6.1 Hz, 6 H) ppm. 13C NMR (125 MHz, CDCl3): δ = 294.5, 210.5, 180.5,
177.3, 163.6, 149.9, 144.8, 141.2, 139.0, 135.1, 132.4, 129.6, 127.1,
124.4, 117.2, 112.2, 78.7, 78.2, 76.0, 51.5, 21.1, 19.5 ppm. IR (CH2Cl2
Self-Metathesis Reaction of Dodec-1-ene (9): Dodec-1-ene (9;
4.5 mmol, 757 mg, 0.999 mL) was added to a dry Schlenk tube,
followed by the Ru complex (0.005 equiv., 0.0225 mmol) and the
mixture was stirred at 60 °C under vacuum (40 mbar) for 6 h. The
reaction mixture was analysed by gas chromatography.
film): ν = 3312, 2920, 1674, 1599, 1571, 1484, 1287, 1261, 1221,
˜
1311, 931, 848, 721, 580 cm–1. MS (ESI, CHCl3): m/z = 831.13 [M]+.
C41H42Cl3N3O3Ru (832.22): calcd. C 59.17, H 5.09, Cl 12.78, N 5.05;
found C 59.23, H 5.31, N 4.80.
Self-Metathesis Reaction of Methyl Oleate (11):[64] Methyl oleate
(11; 0.5 mmol, 148 mg) and dodecane (0.3 mmol, 51 mg, internal
standard) were dissolved in toluene (3 mL) in a dried Schlenk tube.
The Ru complex (Hov II, Hov-NQ, Hov-BQ or Hov-AQ; 0.01 equiv.,
0.005 mmol) was added and the mixture was stirred at 50 °C. Sam-
ples were removed after 1, 5 and 24 h and analysed by gas chroma-
tography.
Hov-AQ: This compound was obtained according to the general
procedure by using N-[4-isopropoxy-3-(prop-1-en-1-yl)phenyl]-9,10-
dioxo-9,10-dihydroanthracene-2-carboxamide (8; 0.12 mmol,
51.1 mg) and Ind II. The product was obtained as green crystals
1
(0.055 mmol, 48.2 mg, 55 %). H NMR (400 MHz, CD2Cl2): δ = 16.50
(s, 1 H), 8.71 (d, J = 1.7 Hz, 1 H), 8.46–8.43 (m, 2 H), 8.38–8.33 (m, 2
H), 8.07 (br. s 1 H), 7.90–7.86 (m, 2 H), 7.82 (dd, J = 8.8, 2.6 Hz, 1 H),
7.43 (d, J = 2.6 Hz, 1 H), 7.10 (s, 3 H), 6.86 (d, J = 8.9 Hz, 1 H), 4.89
(sept., J = 6.1 Hz, 1 H), 4.18 (s, 4 H), 2.46 (br. s, 12 H), 1.54 (s, 6 H),
1.25 (d, J = 6.1 Hz, 6 H) ppm. 13C NMR (101 MHz, CD2Cl2): δ = 214.3,
205.1, 192.1, 182.4, 182.3, 163.6, 149.0, 139.8, 139.0, 135.3, 134.5,
134.4, 133.6, 133.4, 133.0, 132.6, 129.2, 127.9, 127.2, 125.0, 121.2,
114.1, 113.0, 75.5, 29.7, 20.9, 20.8, 13.8, 4.2, 0.7 ppm. IR (CH2Cl2 film):
Cross-Metathesis Reactions of Methyl Oleate (11) with (Z)-But-
2-ene-1,4-diol Diacetate (14):[67] Methyl oleate (11; 0.5 mmol,
148 mg), (Z)-but-2-ene-1,4-diol diacetate (14; 1 mmol, 172 mg) and
tetradecane (internal standard; 0.3 mmol, 51 mg) were dissolved in
toluene (3 mL) in a dried Schlenk tube under argon. The Ru com-
plex (Hov II, Hov-NQ, Hov-BQ or Hov-AQ; 0.01 equiv., 0.005 mmol)
was added and the mixture was stirred at 50 °C. Samples were
removed after 1, 5 and 24 h and analysed by gas chromatography.
ν = 2984, 2960, 2913, 1672, 1588, 1532, 1491, 1448, 1421, 1409,
˜
1335, 1322, 1299, 1254, 1240, 1216, 1136, 1104, 932, 903, 851, 807,
797, 739, 705, 650, 636, 570 cm–1. MS (ESI, MeOH): m/z = 875.18
[M]+. HRMS: calcd. for C46H45Cl2N3O4Ru 875.1831; found 875.1821.
C46H45Cl2N3O4Ru (875.85): calcd. C 63.08, H 5.18, Cl 8.09, N 4.80;
found C 63.38, H 5.26, Cl 7.99, N 4.96.
Ring-Closing Metathesis of Diallyl Ether (12):[47] The Ru complex
(Hov II, Hov-NQ, Hov-BQ or Hov-AQ; 0.0016 mmol) was dissolved
in CD2Cl2 (0.7 mL) in a 5 mL vial under argon. Diallyl ether
(0.16 mmol, 15.7 mg, 19.6 mL) was added to the solution and the
reaction mixture was transferred to an NMR tube fitted with a screw
cap. The NMR tube was heated to 40 °C in an oil bath and the
Synthesis of (3S,4R)-4-Allyl-3-{(R)-1-[tert-butyl(dimethyl)-
silyloxy]ethyl}azetidin-2-one: Potassium iodide (105 mmol, 17.4 g)
and DMF (400 mL) were placed in a 1 L round-bottomed flask fol-
lowed by allyl bromide (105 mmol, 12.7 g) and indium powder
(70 mmol, 10.5 g). After 1 h, a solution of (2R,3R)-3-{(1R)-1-[tert-
butyl(dimethyl)silyloxy]ethyl}-4-oxo-2-azetidinyl acetate (35 mmol,
10.1 g) in DMF (100 mL) was added and the reaction mixture was
1
reaction was monitored by H NMR spectroscopy.
Synthesis of Diene 20: A solution of 1-cyclopropyl-6,8-difluoro-4-
oxo-7-(1-undec-10-enoyloctahydropyrrolo[3,4-b]pyridin-6-yl)-1.4-di-
hydroquinoline-3-carboxylic acid[64] (4.99 mmol, 2.77g), N,N-di-
methylaminopyridine (DMAP; 4.99 mmol, 0.61 g) and undec-10-en-
stirred at room temperature for 24 h. The reaction mixture was 1-ol (5.49 mmol, 0.935 g, 1.1 mL) in DCM (15 mL) was cooled with
diluted with ammonium chloride (250 mL) and extracted with
methyl tert-butyl ether (3 × 150 mL). The combined organic phases
stirring in an ice bath. 1-Ethyl-3-[3-(dimethylamino)propyl]carbodi-
imide (EDCI; 15 mmol, 2.87 g) dissolved in DCM (5 mL) was added
Eur. J. Org. Chem. 2017, 626–638
635
© 2017 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Kajetanowicz, Anna
Kajetanowicz, Anna
 Milewski, Mariusz
Milewski, Mariusz
 Rogińska, Joanna
Rogińska, Joanna
 Gajda, Roman
Gajda, Roman
 Wo?niak, Krzysztof
Wo?niak, Krzysztof