
Journal of Physical Chemistry p. 457 - 462 (1989)
Update date:2022-08-17
Topics:
 Piermarini, G. J.
Piermarini, G. J.
 Block, S.
Block, S.
 Miller, P. J.
Miller, P. J.
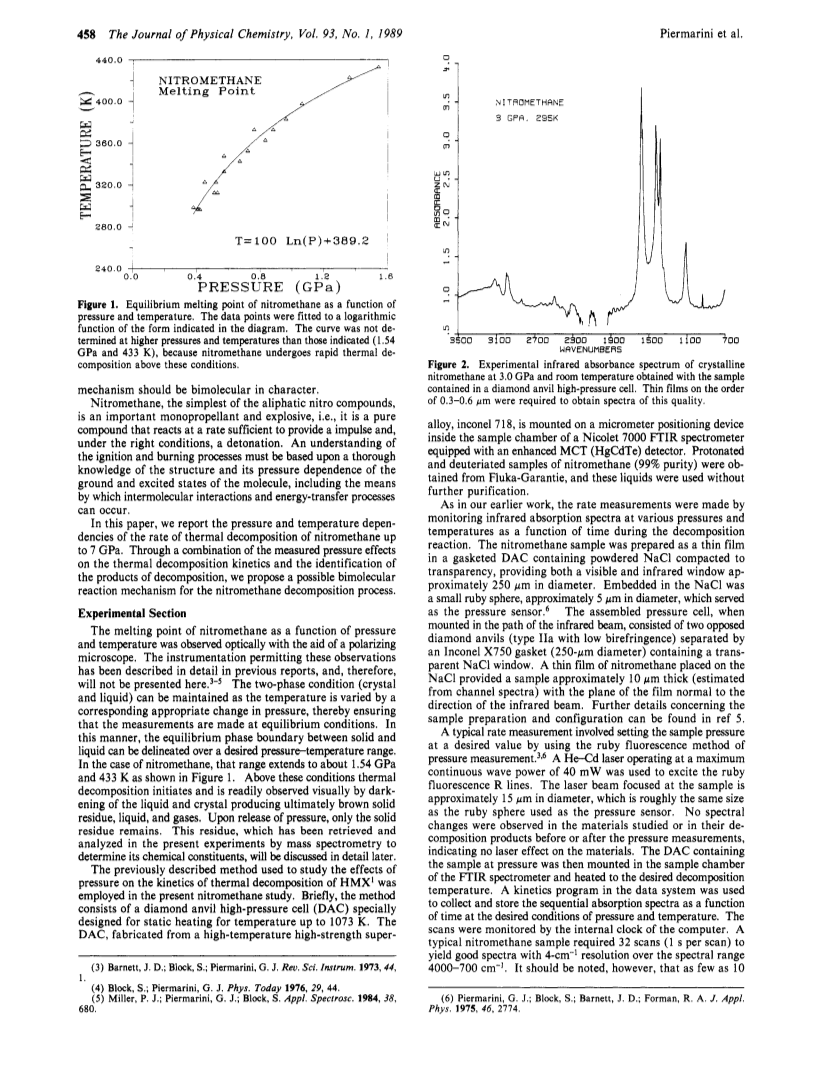
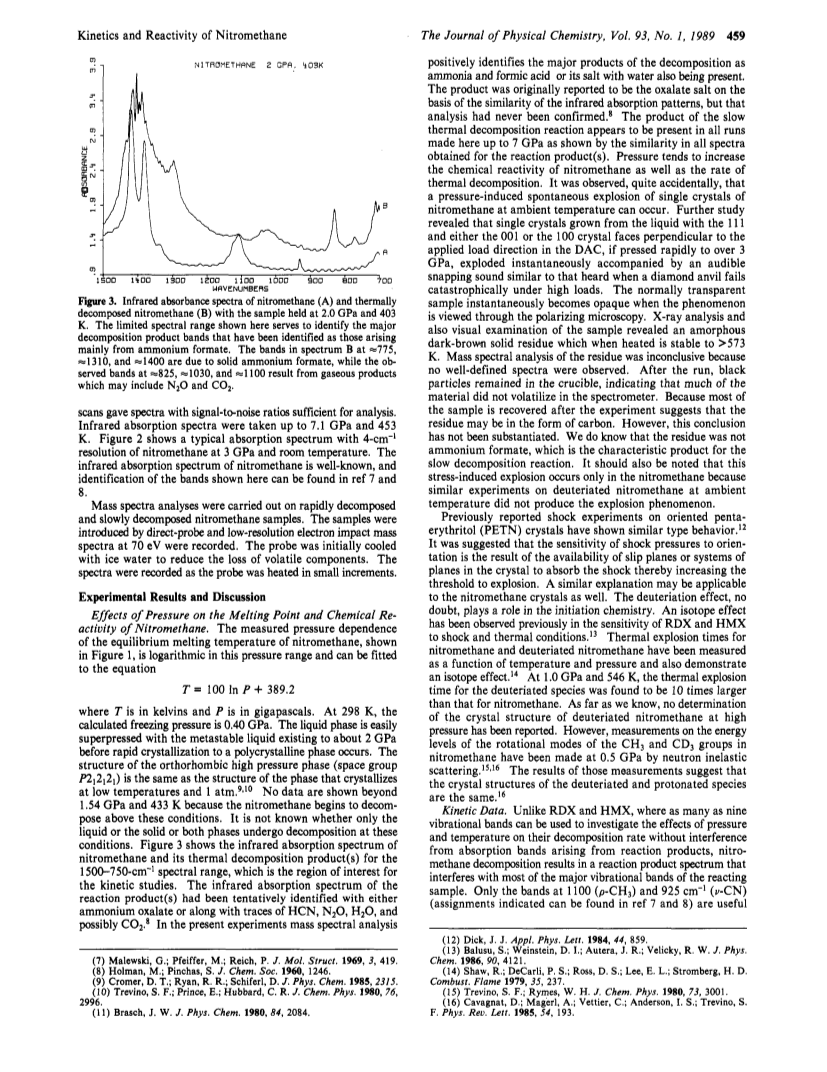
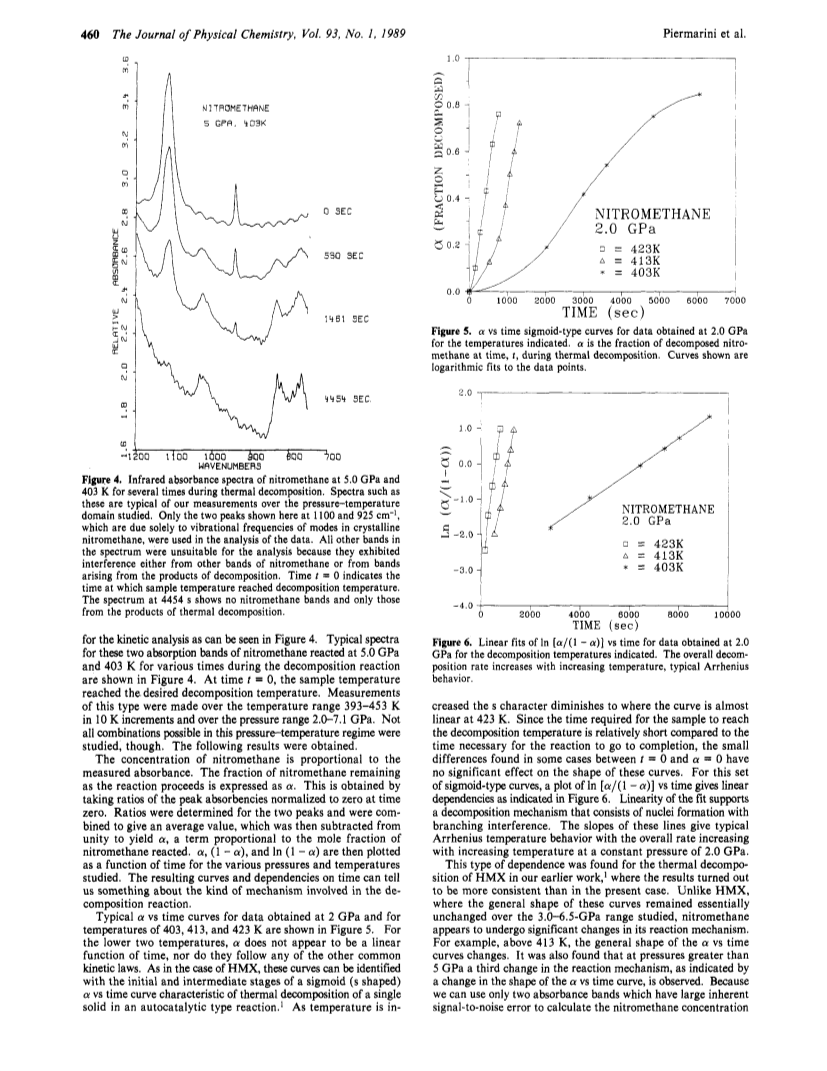
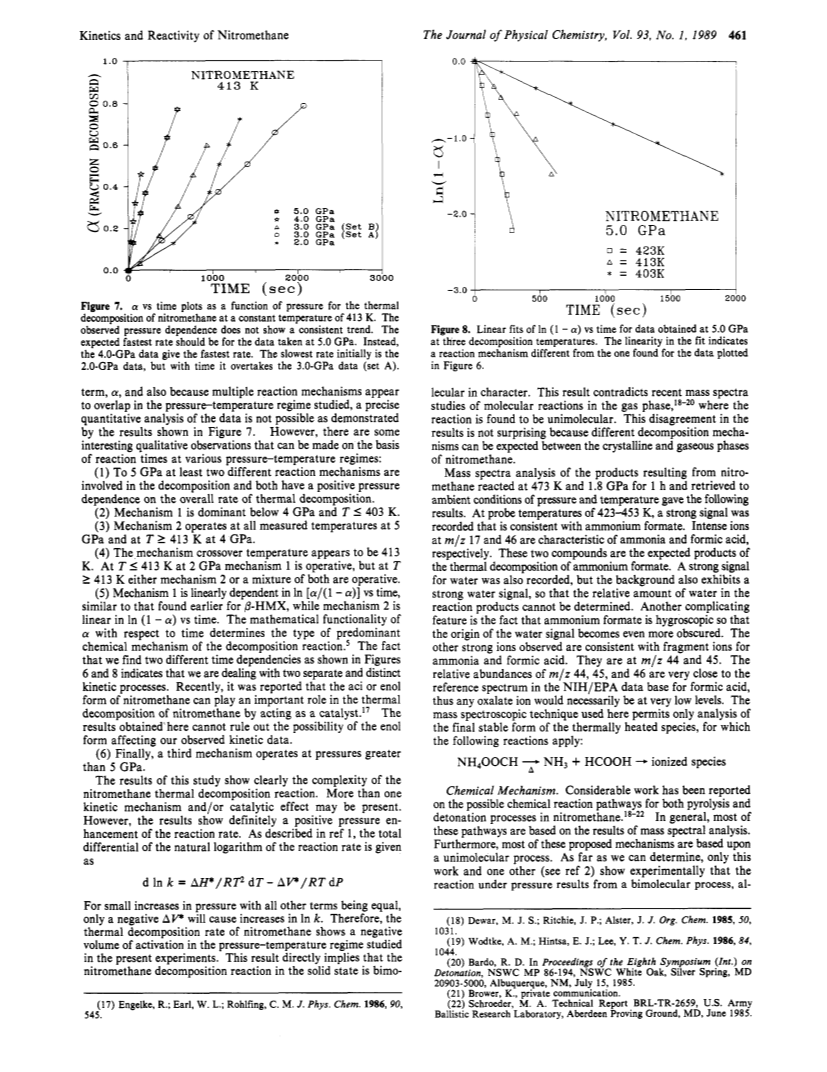
The effect of pressure and temperature on the melting point, thermal decomposition rate, and chemical reactivity of nitromethane have been studied.A diamond anvil high-pressure cell in conjunction with (1) an optical polarizing microscope for melting point and chemical reactivity observations and (2) an automated Fourier transform infrared instrument for thermal decomposition kinetic measurement was used.Both pressure and temperature were found to increase the rate of thermal decomposition, indicating an overall bimolecular reaction mechanism.However, the mechanism is complex and appears to vary over large changes in pressure.Nevertheless, a chemical mechanism is proposed that explains the bimulecularity and also accounts for the observed decomposition products, ammonium formate and water.A dynamic stress-induced catastrophic reaction in nitromethane, which appears to be crystal orientation dependent with respect to the applied stress, was found.Under the same conditions, deuteriated nitromethane did not exhibit this catastrophic reaction.


Contact:021-36356756
Address:Room601,Building No.14,280 Yangcheng Road,Shanghai
Contact:+86-27-87204219, +86-27-87215023
Address:2402, HuiGu Space-time Building, 8 Forest Road, East Lake Hi-Tech Development Zone
Contact:+86-10-62651721
Address:29 Yongxing Road, Daxing District,Beijing China
Contact:+86-518-81061113
Address:No. 8 Lingzhou Road, Lianyungang, Jiangsu, China
WEIFANG RICHEM INTERNATIONAL LTD
Contact:86-536-2222176
Address:weifang,shandong
Doi:10.1002/jhet.3611
(2019)Doi:10.1055/S-0029-1218635
(2010)Doi:10.1246/cl.1985.411
(1985)Doi:10.1002/hlca.193301601107
(1933)Doi:10.1002/anie.201307406
(2013)Doi:10.1016/S0022-328X(03)00266-3
(2003)