Products Categories
| CAS No.: | 571-20-0 |
|---|---|
| Name: | 5alpha-Androstane-3b,17b-diol |
| Article Data: | 120 |
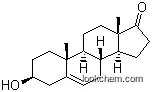
| Molecular Structure: | |
|
|
|
| Formula: | C19H32 O2 |
| Molecular Weight: | 292.462 |
| Synonyms: | 5a-Androstane-3b,17b-diol (7CI,8CI); 3b,17b-Androstanediol; 3b,17b-Dihydroxy-5a-androstane; 3b-Adiol; Maxterone; NSC 50891 |
| EINECS: | 209-334-3 |
| Density: | 1.09g/cm3 |
| Melting Point: | 161 °C |
| Boiling Point: | 415°Cat760mmHg |
| Flash Point: | 186°C |
| Hazard Symbols: | Xi |
| Risk Codes: | 36/38 |
| Safety: | Experimental reproductive effects. When heated to decomposition it emits acrid smoke and irritating vapors. |
| PSA: | 40.46000 |
| LogP: | 3.75090 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE

| Conditions | Yield |
|---|---|
| With diisobutylaluminium hydride; nickel dichloride In dichloromethane; toluene at -78℃; for 0.25h; Inert atmosphere; | 99% |
| Multi-step reaction with 2 steps 1: 97 percent / NaBH4 / methanol / 1 h / 0 - 20 °C 2: 95 percent / NaOH / methanol / 0.17 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: sodium tetrahydroborate; methanol / 0.42 h / 20 °C / Inert atmosphere 2: potassium carbonate / methanol / 24 h / 20 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 0 - 20℃; | 95% |
| With ethanol; nickel Hydrogenation; | |
| With hydrogenchloride; acetic acid; platinum Hydrogenation.; |
- 3090-70-8
(3S,5S,8R,9S,10S,13S,14S,17S)-17-hydroxy-10,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-3-yl acetate

- 571-20-0
5-androgen-3,17-diol

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol for 0.166667h; Heating; | 95% |
| With potassium carbonate In methanol at 20℃; for 24h; Inert atmosphere; | 89% |
| With sodium hydroxide In methanol Yield given; |
- 1239-31-2
3β-acetoxy-5α-androstan-17-one

A
- 3090-70-8
(3S,5S,8R,9S,10S,13S,14S,17S)-17-hydroxy-10,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-3-yl acetate

B
- 571-20-0
5-androgen-3,17-diol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In 1,4-dioxane; methanol for 6h; Ambient temperature; Yields of byproduct given; | A 86% B n/a |
| With sodium tetrahydroborate In 1,4-dioxane; methanol for 6h; Ambient temperature; Yields of byproduct given; | A n/a B 760 mg |
- 16992-89-5
3α-hydroxyandrost-4-en-17β-yl acetate

A
- 571-20-0
5-androgen-3,17-diol

B
- 1852-53-5
androstanediol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; dihydrogen peroxide; diborane Product distribution; multistep reaction: 1.) THF, 0 deg C, 4 h, 2.) overnight; hydroboration of androst-4-enes; stereochemical aspects; | A n/a B n/a C 21% D 63% |
| With sodium hydroxide; dihydrogen peroxide; diborane 1.) THF, 0 deg C, 4 h, 2.) overnight; Yield given. Multistep reaction. Yields of byproduct given. Title compound not separated from byproducts; | |
| With sodium hydroxide; dihydrogen peroxide; diborane 1.) THF, 0 deg C, 4 h, 2.) overnight; Yield given. Multistep reaction. Yields of byproduct given; |

- 846-46-8
androstanedione

A
- 481-29-8
Epiandrosterone

B
- 53-41-8
cis-androsterone

C
- 571-20-0
5-androgen-3,17-diol

| Conditions | Yield |
|---|---|
| for 504h; Rhodotorula mucilaginosa; | A 18% B 35% C 30% |

- 521-18-6
Stanolone

A
- 481-29-8
Epiandrosterone

B
- 53-41-8
cis-androsterone

C
- 846-46-8
androstanedione

D
- 571-20-0
5-androgen-3,17-diol

| Conditions | Yield |
|---|---|
| for 504h; Rhodotorula mucilaginosa; | A 21% B 33% C 5% D 33% |

- 521-18-6
Stanolone

A
- 571-20-0
5-androgen-3,17-diol

B
- 27261-27-4
5α-androstane-3β,17β,16β-triol

C
- 121209-70-9
5α-androstane-2β,3α,16α,17β-tetrol

| Conditions | Yield |
|---|---|
| With fungus Gnomonia fructicola In ethanol at 24 - 26℃; for 120h; | A 3% B 20% C 3% |
| With fungus Gnomonia fructicola In ethanol at 24 - 26℃; for 120h; | A 3% B 20% C 3% |

- 846-46-8
androstanedione

A
- 481-29-8
Epiandrosterone

B
- 29907-31-1
11α-hydroxy-5α-androstane-3,17-dione

C
- 17752-36-2
3β,5α-dihydroxy-5α-androstan-17-one

D
- 571-20-0
5-androgen-3,17-diol

| Conditions | Yield |
|---|---|
| With Cephalosporium aphidicola In ethanol; dimethyl sulfoxide for 168h; | A 9% B 3.5% C 1% D 5.5% |


| Conditions | Yield |
|---|---|
| With sodium |
- 126338-77-0miramistin
- 7440-33-7Tungsten
- 92113-31-0Collagen hydrolyzates
- 119520-06-8Imidazo[4,5,1-jk][1]benzazepin-2(1H)-one,4,5,6,7-tetrahydro-7-hydroxy-6-[(1-methylethyl)amino]-, hydrochloride (1:1),(6R,7R)-rel-
- 20018-09-1Benzene,1-[(diiodomethyl)sulfonyl]-4-methyl-
- 94-96-21,3-Hexanediol,2-ethyl-
- 555-45-3Tetradecanoic acid,1,1',1''-(1,2,3-propanetriyl) ester
- 11096-26-7Erythropoietin
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin

What can I do for you?
Get Best Price
Chemistry
IUPAC Name: (3S,5S,8R,9S,10S,13S,14S,17S)-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthrene-3,17-diol
Synonyms: 10,13-Dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-3,17-diol ; 10,13-Dimethylhexadecahydro-1H-cyclopenta[a]phenanthrene-3,17-diol ; 10,13-Diméthylhexadécahydro-1H-cyclopenta[a]phénanthrène-3,17-diol ; androstane-3,17-diol ; 3.alpha-17.beta.-Dihydroxy-5.alpha.-androstane ; 3alpha,17alpha-Dihydroxy-5beta-androstane ; 3alpha,17-Dihydroxy-5beta-androstane ; 3beta,17alpha-Dihydroxy-5alpha-androstane ; 3-beta,17-beta-Androstanediol ; 3beta,17beta-Dihydroxy-5alpha-androstane ; 3-beta,17-beta-Dihydroxy-5-alpha-androstane
The Molecular Formula of 5-alpha-Androstane-3-beta-17-beta-diol (571-20-0):C19H32O2
The Molecular Weight of 5-alpha-Androstane-3-beta-17-beta-diol (571-20-0):292.456180 g/mol
The Molecular Structure of 5-alpha-Androstane-3-beta-17-beta-diol (571-20-0):
Index of Refraction: 1.546
Molar Refractivity: 84.95 cm3
Molar Volume: 268.1 cm3
Polarizability: 33.67x 10-24cm3
Surface Tension: 42.6 dyne/cm
Density: 1.09 g/cm3
Flash Point: 186 °C
Enthalpy of Vaporization: 77.17 kJ/mol
Boiling Point: 415 °C at 760 mmHg
Vapour Pressure: 1.27E-08 mmHg at 25°C
Uses
5-alpha-Androstane-3-beta-17-beta-diol (571- 20-0) can be used as anabolic agent.
Safety Profile
Experimental reproductive effects. When heated to decomposition it emits acrid smoke and irritating vapors.