Products Categories
| CAS No.: | 87-51-4 |
|---|---|
| Name: | Indole-3-acetic acid |
| Article Data: | 107 |
| Molecular Structure: | |
|
|
|
| Formula: | C10H9NO2 |
| Molecular Weight: | 175.187 |
| Synonyms: | beta-Indolylacetic acid;1H-Indole-3-acetic-a-t acid (9CI);Indole-3-acetate;omega-Skatole carboxylic acid;Acetic acid, indolyl-;.alpha.-IAA;.beta.-Indole-3-acetic acid;2-(1H-indol-3-yl)acetate;alpha-IAA;GAP;meso-2,3-Butanediol-bis(iodoacetate);3-Indolylmethylcarboxylic acid;3-Iaa;3-Indolylacetic acid;beta-Indoleacetic acid;Indolyl-3-acetic acid;1H-indol-3-ylacetic acid;Heteroauxin;Indol-3-ylacetic acid;[(2R,3S)-3-(2-iodoacetyl)oxybutan-2-yl] 2-iodoacetate;[3H]-IAA;(1H-Indol-3-yl)acetic acid;.beta.-Indolylacetic acid;Hexteroauxin;Indoleacetate;2-(3-Indolyl)acetic acid 3-(Carboxymethyl)-1H-indole;3-(Carboxymethyl)indole;potassium 2-(1H-indol-3-yl)acetate;.alpha.-Indol-3-yl-acetic acid;2-(1H-Indol-3-yl)acetic acid;Indole-3-acetic-t acid;Rhizopon A;Heteroauxinhexteroauxiniaa;3-Indolebutyric acid(IBA);.beta.-Indoleacetic acid;1H-Indole-3-acetic acid (9CI);Rhizopin;3-Indole acetic acid;3-Indoleacetic acid(IAA);Indole-3- acetic acid;3-Indoleacetic Acid, Indole-3-acetic acid;indoleI-3-acetic acid;Indole-3-acetic acid (3-IAA);3-Indole propanoic acid; |
| EINECS: | 201-748-2 |
| Density: | 1.355 g/cm3 |
| Melting Point: | 165-169 °C(lit.) |
| Boiling Point: | 415.03 °C at 760 mmHg |
| Flash Point: | 204.803 °C |
| Solubility: | Soluble in ethanol, acetone, diethyl ether. Slightly soluble in chloroform. Insoluble in water |
| Appearance: | white to tan crystals |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 36/37/38 |
| Safety: | 22-24/25 |
| PSA: | 53.09000 |
| LogP: | 1.79500 |
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 90221-55-92-bromo-5-methylbenzaldehyde
- 885590-99-82,3-DIFLUORO-4-IODOBENZALDEHYDE
- 97730-31-9(S)-4'-(2-Methylbutyl)Biphenyl-4-Carbonitrile
- 926293-55-26-Bromo-2-methylpyridine-3-carboxaldehyde
- 911112-05-55-Iodo-3-(trifluoromethyl)-2-pyridinamine
- 886364-50-71-(5-Bromo-2-pyridinyl)-2,2,2-trifluoroethanone

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water at 250℃; under 34200 - 38000 Torr; for 12h; | 95% |

| Conditions | Yield |
|---|---|
| With sodium perborate In water for 48h; Ambient temperature; | 71% |
| With sodium hydroxide; dihydrogen peroxide |
- 40432-84-6
(5-bromo-1H-indol-3-yl)-acetic acid

- 87-51-4
indole-3-acetic acid

| Conditions | Yield |
|---|---|
| With hydrogen; sodium acetate; nickel In ethanol under 2327.2 Torr; for 4h; Ambient temperature; | 52% |

| Conditions | Yield |
|---|---|
| With diethyl ether; copper Erhitzen des Reaktionsprodukts mit methanol. KOH; |


| Conditions | Yield |
|---|---|
| With potassium hydroxide at 250℃; | |
| Stage #1: indole; glycolic Acid With potassium hydroxide at 250℃; for 18h; Stage #2: With hydrogenchloride In water at 10℃; | |
| With hydrogenchloride; potassium hydroxide In water |

| Conditions | Yield |
|---|---|
| With potassium permanganate; acetone | |
| With NAD; water; enzyme AldC from Pseudomonas syringae In aq. buffer at 25℃; pH=8.0; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| With potassium hydroxide | |
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| With potassium permanganate; sodium carbonate |

| Conditions | Yield |
|---|---|
| With ethanol; sodium cyanide |

| Conditions | Yield |
|---|---|
| With ethanol; sodium cyanide at 200℃; | |
| Multi-step reaction with 2 steps 1: NaCN; aqueous ethanol 2: aqueous NaOH View Scheme | |
| With ethanol; sodium cyanide at 200℃; |
- 58-93-5Hydrochlorothiazide
- 64485-93-4Cefotaxime sodium
- 54060-30-93-Aminophenylacetylene
- 54965-24-1Tamoxifen citrate
- 90776-59-3β-Methyl vinyl phosphate
- 1492-18-8Calcium folinate
- 39537-23-0L-Glutamine, L-alanyl-
- 2392-39-4Pregna-1,4-diene-3,20-dione,9-fluoro-11,17-dihydroxy-16-methyl-21-(phosphonooxy)-, sodium salt (1:2), (11b,16a)-
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Consensus Reports
Reported in EPA TSCA Inventory.
Specification
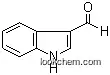
The Indole-3-acetic acid, with the CAS registry number 87-51-4, is also known as 3-Indolylacetic acid. It belongs to the product categories of Indole derivative; Indoles and derivatives; Carboxylic Acids; Pyrroles & Indoles; Indole; Organic acids; API intermediates; Indoles; Auxins; Biochemistry; Plant Growth Regulators; Simple Indoles; Plant Hormones; Carboxylic Acids; Heterocycles. Its EINECS number is 201-748-2. This chemical's molecular formula is C10H9NO2 and molecular weight is 175.19. What's more, its systematic name is 1H-Indol-3-ylacetic acid. Its classification codes are: (1)Agricultural Chemical; (2)Growth Substances; (3)Growth regulator / Fertilizer; (4)Mutation data; (5)Plant growth regulators; (6)Reproductive Effect; (7)Tumor data. This chemical should be sealed and stored in a cool and dry place. It is the most common, naturally-occurring, plant hormone of the auxin class. It is the best known of the auxins, and has been the subject of extensive studies by plant physiologists.
Physical properties of Indole-3-acetic acid are: (1)ACD/LogP: 1.526; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.48; (4)ACD/LogD (pH 7.4): -1.32; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 14.45; (8)ACD/KOC (pH 7.4): 1.00; (9)#H bond acceptors: 3; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 53.09 Å2; (13)Index of Refraction: 1.694; (14)Molar Refractivity: 49.642 cm3; (15)Molar Volume: 129.315 cm3; (16)Polarizability: 19.68×10-24cm3; (17)Surface Tension: 66.4 dyne/cm; (18)Density: 1.355 g/cm3; (19)Flash Point: 204.803 °C; (20)Enthalpy of Vaporization: 70.436 kJ/mol; (21)Boiling Point: 415.03 °C at 760 mmHg; (22)Vapour Pressure: 0 mmHg at 25°C.
Preparation: this chemical can be prepared by 3-indol-3-yl-2-oxo-propionic acid at the ambient temperature. This reaction will need reagent sodium perborate tetrahydrate and solvent H2O with the reaction time of 48 hours. The yield is about 71%.

Uses of Indole-3-acetic acid: it can be used to produce indol-3-yl-acetyl chloride at the temperature of 0 °C. It will need reagent PCl5 and solvent diethyl ether with the reaction time of 1 hour. The yield is about 72%.

When you are using this chemical, please be cautious about it as the following:
This chemical is irritating to eyes, respiratory system and skin. You should not breathe dust. When using it, you must avoid contact with skin and eyes.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)Cc2c1ccccc1nc2
(2)Std. InChI: InChI=1S/C10H9NO2/c12-10(13)5-7-6-11-9-4-2-1-3-8(7)9/h1-4,6,11H,5H2,(H,12,13)
(3)Std. InChIKey: SEOVTRFCIGRIMH-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 150mg/kg (150mg/kg) | National Technical Information Service. Vol. AD691-490, | |
| rat | LD | oral | > 500mg/kg (500mg/kg) | National Academy of Sciences, National Research Council, Chemical-Biological Coordination Center, Review. Vol. 5, Pg. 7, 1953. |