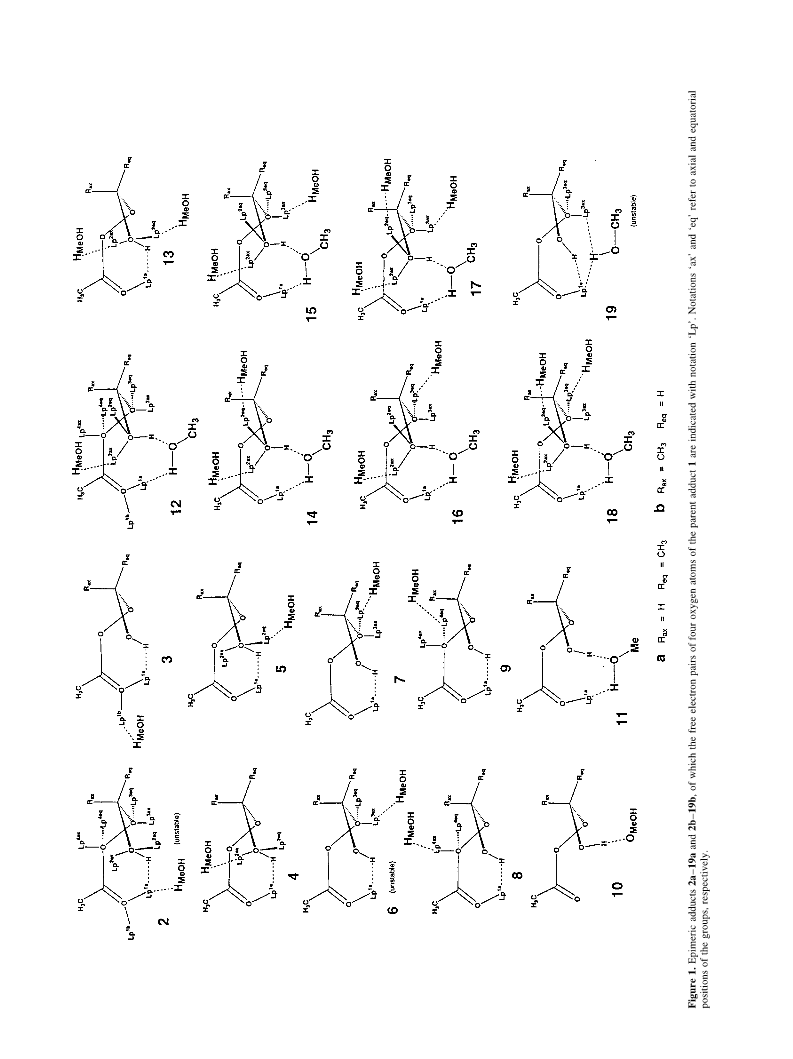
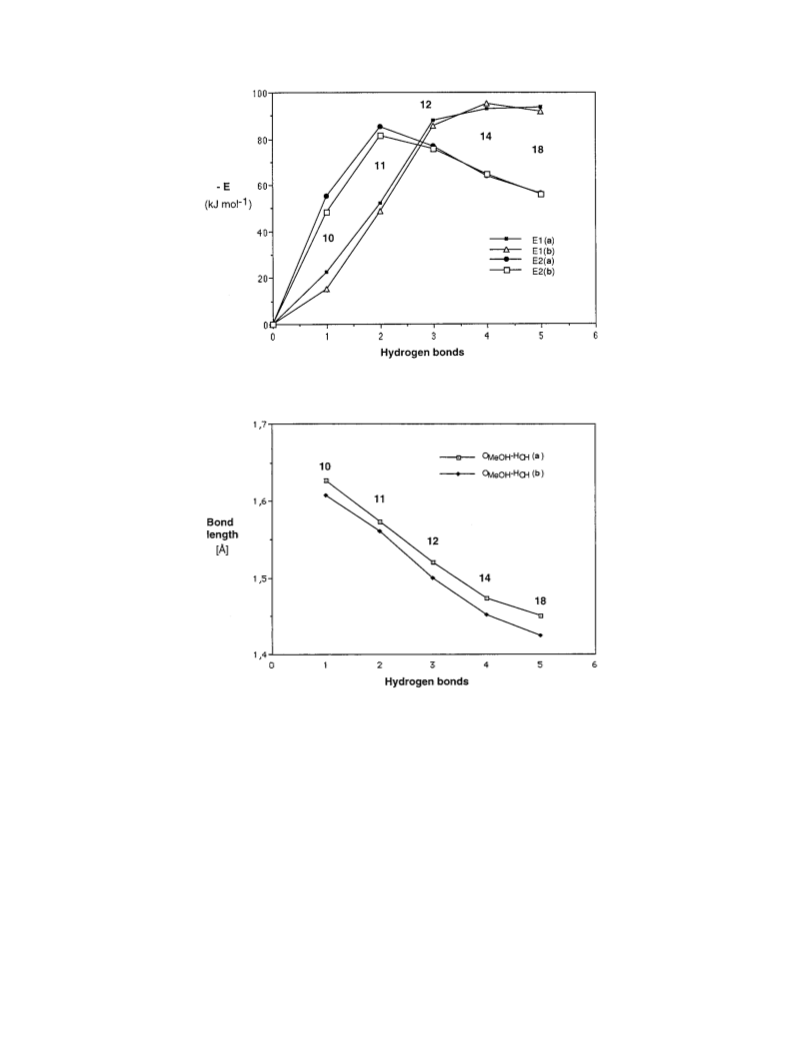
C. Lehtinen et al. / Tetrahedron 57 (2001) 4741±4751
4751
observed formation of acid (pathway a, Scheme 1) in the
presence of alcohol was rationalised on the basis of
the computational study and Hammond's postulate: the
structural similarity of adducts b (reacting via pathway a,
Scheme 1) to the transition state of the reaction (Scheme 2)
was higher than that of adducts a (reacting via pathway b,
Scheme 1).
and the electron multiplier voltage was adjusted to obtain
proper sensitivity in both cases.
4.2. Oxidation of aldehyde
A ¯at-bottomed glass vessel equipped with condenser and
oxygen balloon was charged with aldehyde, internal
standard and solvent if used. Magnetic stirring (1000±
1250 rpm) was commenced and the reaction mixture was
evacuated and oxygenated three times at the chosen
temperature. When reactions were done in air air¯ow in
synthesis was 34 ml/min.
The computational results of this study show that methods
of theoretical chemistry can be used to better understand
solvent effects at the molecular level. The importance of
computational inspections is enhanced in the case of
reactions of labile intermediates which are dif®cult to
study experimentally.
4.3. Preparation of 3-heptyl formate used as model
compounds
3-Heptyl formate was prepared from the corresponding
alcohol (20 mmol) with dimethylformamide (20 mmol)
and benzoyl chloride (20 mmol) in 12 ml dichloroethane
according to method of Barluenga et al.13 Besides formate,
a small amount of unreacted 3-heptanol was present in the
crude product. The formate was puri®ed by vacuum
distillation and the yield was 39%.
4. Experimental
4.1. General
2-Ethylhexanal, 2-ethylbutanal, pentanal, 2-phenylpropa-
nal, and all the solvents were dried, distilled and preserved
under inert atmosphere until use.
Gas chromatographic analyses were performed with an HP
6890 instrument: Polar Innowax column 30 m; initial
column temperature 408C ®nal column temp. 2508C;
progress rate 108C/min; constant ¯ow 6.3 ml/min of carrier
gas; initial pressure 0.93 bar. The main oxidation products
of aldehydes were identi®ed and quanti®ed by comparison
with authentic samples. Amount of 2-formyl butanal was
quanti®ed by using 3-formyl heptanal (For preparation
see below) as standard and amount of 2-phenyl propyl
formate by using 2-phenylpropanoic acid as standard.
Decane or tetradecane was used as internal standard to
calculate the exact amount of substance present in the
reaction mixture.
Acknowledgements
The TEKES foundation is acknowledged for partial
®nancial support.
References
1. Reviews include: (a) McNesby, J.; Heller, C. Chem. Rev.
1954, 54, 325. (b) Sajus, L.; Roch, S. Comprehensive
Chemical Kinetics, Bamford, C., Tipper, C., Eds.; Elsevier:
New York, 1980; Vol. 16, pp 89.
2. For example: (a) Godfrey, I.; Sargent, M.; Elix, J. J. Chem.
Soc., Perkin Trans. 1 1974, 1353. (b) Royer, J.; Beuglemans-
Verrier, M. C. R. Acad. Sc. Paris, Serie C 1971, 1818.
(c) Camps, F.; Coll, J.; Messeguer, J.; Pericas, M. Tetrahedron
Lett. 1981, 22, 3895.
The oxidation products were also identi®ed by GC-MS. The
sample components were identi®ed using gas chromato-
graphy-mass spectrometry in full scan mode with electron
impact (EI) and chemical ionisation (CI). The EI spectra
were identi®ed by using the NIST-EPA-NIH Mass Spectral
Library (version 1.5a) and manual interpretation. The CI
spectra were used to con®rm the molecular weight of
unknowns in ambiguous case.
3. DeBoer, A.; Ellwanger, R. J. Org. Chem. 1974, 39, 77.
4. Alcaide, B.; Aly, M.; Sierra, M. J. Org. Chem. 1996, 61, 8819.
5. Barrero, A.; Alvarez-Manzaneda, E.; Alvarez-Manzaneda, R.;
Chahboun, R.; Meneses, R.; Aparicio, M. Synlett 1999, 713.
6. Lehtinen, C.; Nevalainen, V.; Brunow, G. Tetrahedron 2000,
56, 9375.
7. Lehtinen, C.; Brunow, G. Organic Process & Development
2000, 4, 544.
The EI spectra were measured using an HP Mass Selective
detector interfaced to HP 6890 Gas chromatograph. The MS
scan range was m/e 25±500 and the speed 1 scan/sec. The
ion source temperature was 2308C and the quadrupole
temperature 1508C. The CI spectra were measured using a
VG Prospec sector mass spectrometer interfaced to an HP
5890 Series II Gas Chromatograph. The scan range was m/e
80±500 and scan speed 2 sec./decade. The ion source
temperature was 2008C. Isobutene was used as reagent gas
and the ion source pressure was maintained at 2£1025 mbar
by regulating the reagent gas ¯ow. In both ionisation cases
the GC columns and parameters were identical to those of
simple GC analyses. The GCMS interface was kept at 2808C
8. Yamada, T.; Takai, T.; Rhode, O.; Mukaiyama, T. Chem. Lett.
1991, 5.
9. Ohakahatsu, Y.; Takeda, M.; Hara, T.; Osa, T.; Misono, A.
Bull. Chem. Soc. Jpn 1967, 1423.
10. Maslow, S.; Bluymberg, E. Russ. Chem. Rev. 1976, 45, 155.
11. Wavefunction Inc., 18401 Von Karman Ave., Suite 370,
Irvine, CA 92612, USA.
12. Freccero, M.; Gandol®, R.; Sarzi-Amade, M.; Rastelli, A.
J. Org. Chem., 1999, 65, 2030.
Â
13. Barluenga, J.; Campos, P.; Gonzales-Nunez, E.; Asensio, G.
Synth. Commun. 1985, 426.

 Lehtinen, Christel
Lehtinen, Christel
 Nevalainen, Vesa
Nevalainen, Vesa
 Brunow, G?sta
Brunow, G?sta