A. Ryckebusch et al. / Bioorg. Med. Chem. Lett. 15 (2005) 297–302
301
compounds display generally good inhibition of hematin
References and notes
formation and high intravacuolar accumulation by
weak-base character, no direct correlation could be
established between these parameters and antimalarial
activity. Exceptions such as aromatic carboxamide
derivatives made us suppose the existence of additional
mechanisms. Fluorescence studies conducted on sulfon-
amide derivatives made us assume that some of our
compounds could have additional targets outside the
digestive vacuole, possibly involving an original mecha-
nism of action. The next step will consist in the search of
these putative biological targets by the affinity chroma-
tography technique, using a derivative of compound 1.
Compounds 9 or 12 can be considered as good candi-
dates for this technique, as their respective amino termi-
nal function enable fixation on solid support while
preserving significant antimalarial activities (respective
IC50 398nM and 152nM).
1. Ridley, R. G. Nature 2002, 415, 686–693.
2
. Stocks, P. A.; Raynes, K. J.; Bray, P. G.; Park, B. K.;
OÕNeill, P. M.; Ward, S. A. J. Med. Chem. 2002, 45, 4975–
4
983.
3
. OÕNeill, P. M.; Mukhtar, A.; Stocks, P. A.; Randle, L. E.;
Hindley, S.; Ward, S. A.; Storr, R. C.; Bickley, J. F.;
OÕNeil, I. A.; Maggs, J. L.; Hughes, R. H.; Winstanley, P.
A.; Bray, P. G.; Park, B. K. J. Med. Chem. 2003, 46, 4933–
1
0
4
945.
4. De, D.; Krogstad, F. M.; Cogswell, F. B.; Krogstad, D. L.
Am. J. Trop. Med. Hyg. 1995, 55, 579–583.
5. De, D.; Krogstad, F. M.; Byers, L. D.; Krogstad, D. L. J.
Med. Chem. 1998, 41, 4926–4941.
6
. Ridley, R. G.; Hofheinz, W.; Matile, H.; Jaquet, C.; Dorn,
A.; Masciadri, R.; Jolidon, S.; Richter, W. F.; Guenzi, A.;
Girometta, M. A.; Urwyler, H.; Huber, W.; Thaithong, S.;
Peters, W. Antimicrob. Agents Chemother. 1996, 40, 1846–
1
854.
7
. Hawley, S. R.; Bray, P. G.; OÕNeill, P. M.; Park, B. K.;
Ward, S. A. Mol. Biochem. Parasitol. 1996, 80, 15–25.
. Krogstad, D. J.; Gluzman, I. Y.; Kyle, D. E.; Oduola, A.
M.; Martin, S. K.; Milhous, W. K.; Schlesinger, P. H.
Science 1987, 238, 1283–1285.
8
5. Conclusion
This work provided us with additional structure–activity
and structure–cytotoxicity information in the N -
9
. Egan, T. J. Exp. Opin. Ther. Pat. 2001, 11, 185–209.
1
1
0. Ryckebusch, A.; D e´ prez-Poulain, R.; Debreu-Fontaine,
M.-A.; Vandaele, R.; Mouray, E.; Grellier, P.; Serghera-
ert, C. Bioorg. Med. Chem. Lett. 2002, 12, 2595–2598.
(
7-chloro-4-quinolyl)-1,4-bis(3-aminopropyl)piperazine
family. Indeed, this study proved that a number of subs-
titutions lead to compounds with high activities and
reduced cytotoxicities.
11. Ryckebusch, A.; D e´ prez-Poulain, R.; Maes, L.; Debreu-
Fontaine, M.-A.; Mouray, E.; Grellier, P.; Sergheraert, C.
J. Med. Chem. 2003, 46, 542–557.
1
2. Two mice out of three died owing to toxicity at 40mg/kg
of compound 1.
3. General procedure of the preparation of cyclic amines 13–
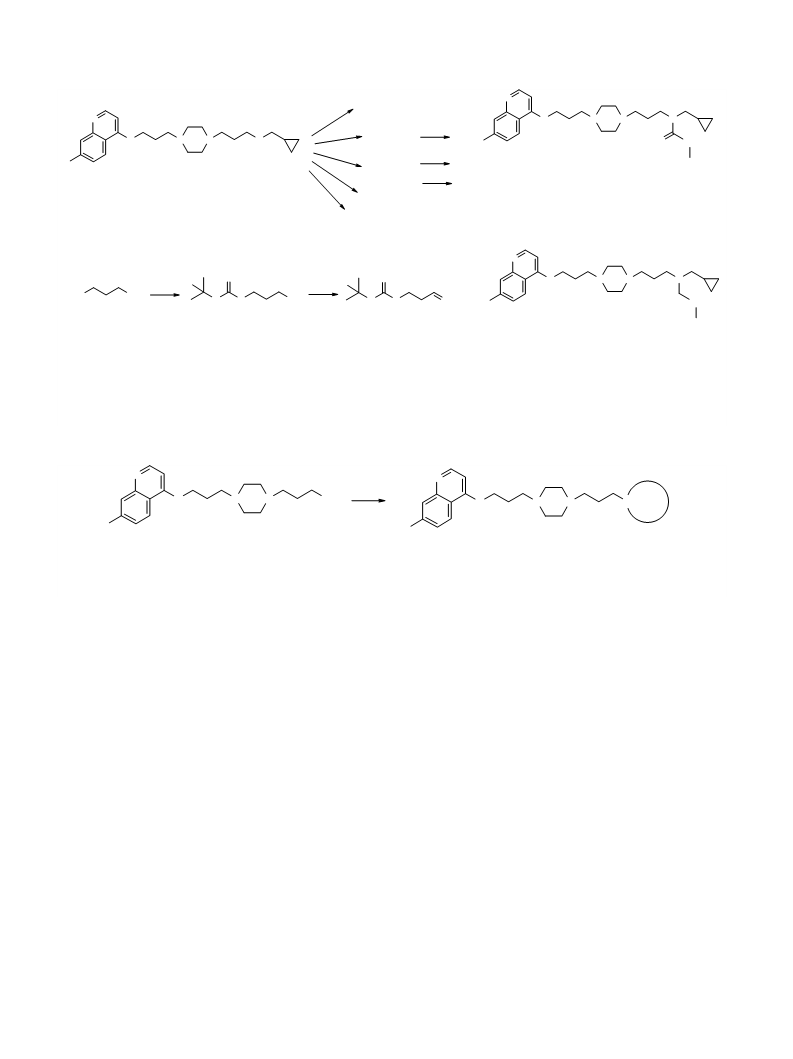
Synthesis and evaluation of the compounds in Series 1
showed that lipophilic substituents could be introduced
in the place of one of the cyclopropyl methylene moieties
in compound 1 while maintaining a high antimalarial
activity. Introduction of cyclic tertiary amines on the
1
1
5: To a solution of amine 2 (150mg, 0.41mmol) in 5mL
of DMF were added the appropriated dibromoalcane
1.2equiv) and K CO (287mg, 5equiv). After the mixture
(
2
3
1
N -(7-chloro-4-quinolyl)-1,4-bis(3-aminopropyl)piper-
was stirred at room temperature for 48h, dichloromethane
was added and the mixture was washed with aqueous 1M
NaHCO . The organic layer was separated and dried over
azine template provided three additional compounds
with high antimalarial activities, among which one dis-
played improved selectivity index compared with refer-
ence compound 1.
3
4
MgSO , the solvent was evaporated and the residue was
purified by thick layer chromatography to yield the desired
product.
4. H and NMR spectra were obtained using a Bruker
1
1
Further investigation is required in order to enlarge
diversity on the second methylenecyclopropyl
member in compound 1. Parallel synthesis of two
libraries of amides and tertiary amines compounds
are being undertaken using compound 3 as a common
precursor. Taking into account our first structure–
activity relationship, these libraries will include on
the terminal region of the molecule derivatives of
primary amines and alcohols (i.e., etheroxides, esters,
secondary and tertiary amines and amides, carbamic
acid esters) and derivatives of carboxylic and hydroxa-
mic acids.
3
00MHz spectrometer. Chemical shifts (d) were expressed
in ppm relative to TMS used as an internal standard. Mass
spectra were recorded on a time-of-flight (TOF) plasma
desorption spectrometer using a californium source or on
a MALDI-TOF Voyager-DE-STR spectrometer. Exam-
ples are given for compounds 4 and 13.
4 H NMR (CDCl ), d (ppm): 8.35 (d, J = 5.4Hz, 1H, Ar–
1
3
H), 8.27–8.20 (m, 1H, Ar–H), 8.01 (d, J = 1.9Hz, 1H, Ar–
H), 7.39–7.34 (m, 1H, Ar–H), 6.39 (d, J = 5.5Hz, 1H, Ar–
H), 3.49–3.45 (m, 4H, CH
2
), 3.30–3.27 (m, 2H, CH
), 2.55–2.51 (m, 2H, CH ), 2.07–
.05 (m, 2H, CH ), 1.98–1.62 (m, 3H, CH and CH ), 0.96–
.79 (m, 5H, CH and CH ), 0.53–0.48 (m, 2H, CH ), 0.27–
2
.23 (m, 2H, CH
2
),
2
2
0
0
1
.80–2.75 (m, 10H, CH
2
2
2
2
2
2
); TOFMS m/z = 484.4.
3 H NMR (CDCl ), d (ppm): 8.42 (d, J = 5.4Hz, 1H,
3
1
Ar–H), 7.86 (d, J = 2.1Hz, 1H, Ar–H), 7.80 (d, J = 8.9Hz,
Acknowledgements
1
2
CH
H, Ar–H), 6.24 (d, J = 5.4Hz, 1H, Ar–H), 3.33–3.27 (m,
H, CH ), 2.58–2.40 (m, 18H, CH ), 1.93–1.83 (m, 2H,
), 1.79–1.68 (m, 6H, CH ); MALDI-MS m/z = 416.2.
2
2
These works are supported by CNRS (GDR 1077, FR
CNRS 63, UMR CNRS 8525) and Universit e´ de
Lille II-Droit et Sant e´ . A.R. was on scholarship
granted by CNRS/R e´ gion Nord-Pas de Calais, France.
The authors thank G e´ rard Montagne for NMR experi-
ments and Herv e´ Drobecq for MS spectra.
2
2
1
5. Analytical HPLC was performed on a Shimadzu system
equipped with a UV detector set at 254nm. HPLC
column: C18 nucleosil using the following eluent system:
A (H O/TFA:100/0.05) and B (CH CN/H O/TFA: 80/20/
2
3
2
0.05). HPLC retention times (HPLC t ) were obtained at
R

 Ryckebusch, Adina
Ryckebusch, Adina
 Debreu-Fontaine, Marie-Ange
Debreu-Fontaine, Marie-Ange
 Mouray, Elisabeth
Mouray, Elisabeth
 Grellier, Philippe
Grellier, Philippe
 Sergheraert, Christian
Sergheraert, Christian
 Melnyk, Patricia
Melnyk, Patricia