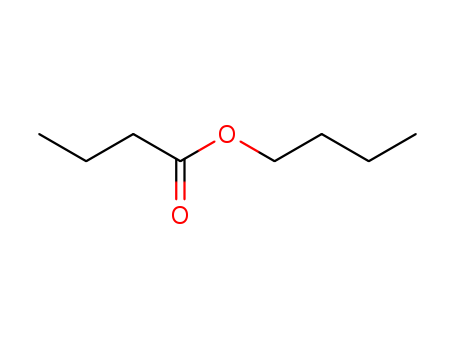
Chemical Property of Butyl butyrate
Chemical Property:
- Appearance/Colour:clear colorless to pale yellowish liquid
- Vapor Pressure:0.027mmHg at 25°C
- Melting Point:-92 °C
- Refractive Index:n20/D 1.406(lit.)
- Boiling Point:165 °C at 760 mmHg
- Flash Point:49.4 °C
- PSA:26.30000
- Density:0.8675 g/cm3
- LogP:2.12980
- Storage Temp.:Flammables area
- Solubility.:0.50g/l
- Water Solubility.:Soluble in water. (1 g/L).
- XLogP3:2.2
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:2
- Rotatable Bond Count:6
- Exact Mass:144.115029749
- Heavy Atom Count:10
- Complexity:89.3
- Transport DOT Label:Class 9
- Purity/Quality:
-
99% *data from raw suppliers
Butyl Butyrate >99.0%(GC) *data from reagent suppliers
Safty Information:
- Pictogram(s):
R10:;
- Hazard Codes:R10:;
- Statements:
10
- Safety Statements:
2-24/25
- MSDS Files:
-
SDS file from LookChem
Useful:
- Chemical Classes:Solvents -> Esters (
- Canonical SMILES:CCCCOC(=O)CCC
-
Description
Butyl butyrate is a kind of ester formed through the condensation of butyric acid and n-butanol. It has a pleasant flavor, and thus being used in the flavor industry to generate a sweet fruity flavor of pineapple-like. It occurs naturally in many kinds of fruits including apple, banana, berries, pear, plum, and strawberry. It is also found in alcoholic beverages. However, it should be noted that it is a marine pollutant, posing a threat to the marine environment. It may also penetrate into soil, contaminating groundwater and other nearby waterways.
-
Uses
It is very important for the food and beverages industries and used as perfuming agents. It is a applied as a solvent for resins. Butyl butyrate can be used as a reactant to synthesize: N-(Phenylmethyl)butanamide by reacting with benzylamine via ester-amide exchange reaction in the presence of supported graphene oxide catalyst.Cyclohexyl butyrate by acylation reaction with cyclohexanol using a ruthenium pincer PNN complex catalyst.Butyl ether by triiron dodecacarbonyl catalyzed hydrosilylation reaction. Flavoring.