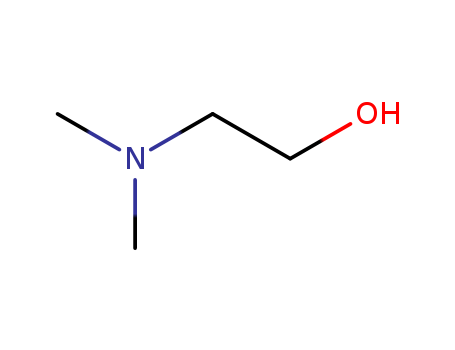
- Chemical Name:N,N-Dimethylethanolamine
- CAS No.:108-01-0
- Deprecated CAS:116134-09-9,156681-25-3,1404326-81-3
- Molecular Formula:C4H11NO
- Molecular Weight:89.1374
- Hs Code.:2922.19 Oral rat LD50: 2000 mg/kg
- European Community (EC) Number:203-542-8
- ICSC Number:0654
- NSC Number:2652
- UN Number:2051
- UNII:2N6K9DRA24
- DSSTox Substance ID:DTXSID2020505
- Nikkaji Number:J5.068G
- Wikipedia:Dimethylethanolamine
- Wikidata:Q241049
- NCI Thesaurus Code:C72733
- RXCUI:3116
- Metabolomics Workbench ID:45347
- ChEMBL ID:CHEMBL1135
- Mol file:108-01-0.mol
Synonyms:(2-Hydroxyethyl)dimethylamine;(Dimethylamino)ethanol;(N,N-Dimethylamino)ethanol;2-(Dimethylamino)ethanol;2-(Dimethylamino)ethyl alcohol;2-(N,N-Dimethylamino)ethanol;Amietol M 21;Dimethyl(2-hydroxyethyl)amine;Dimethyl(hydroxyethyl)amine;Dimethylethanolamine;Dimethylmonoethanolamine;N,N-Dimethyl(2-hydroxyethyl)amine;N,N-Dimethyl-2-aminoethanol;N,N-Dimethyl-N-(b-hydroxyethyl)amine;N,N-Dimethyl-b-hydroxyethylamine;N-(2-Hydroxyethyl)-N,N-dimethylamine;N-(2-Hydroxyethyl)dimethylamine;b-Dimethylaminoethyl alcohol;b-Hydroxyethyldimethylamine;


 C
C




