Products Categories
Basic Information
| CAS No.: | 10294-34-5 |
|---|---|
| Name: | Boron trichloride |
| Molecular Structure: | |
|
|
|
| Formula: | BCl3 |
| Molecular Weight: | 117.17 |
| Synonyms: | Boronchloride (BCl3) (8CI);Trichloroborane;Trichloroboron; |
| EINECS: | 233-658-4 |
| Density: | 1.384 g/cm3 |
| Melting Point: | -107 °C(lit.) |
| Boiling Point: | 12.5 °C at 760 mmHg |
| Flash Point: | -17 °C |
| Solubility: | Decomposes in water |
| Appearance: | colourless gas |
| Hazard Symbols: |
 T+, T+,  T, T,  N, N,  F F
|
| Risk Codes: | 14-26/28-36/37/38-40-67-65-62-51/53-48/20-34-11-50/53-26/27/28-63-39/23/24/25-24-21-10 |
| Safety: | 9-26-28-36/37/39-45-8-61-38-28A-16-1-60-33-23-7/9-62 |
| Transport Information: | UN 3390 6.1/PG 1 |
| PSA: | 0.00000 |
| LogP: | 1.68770 |
Related products
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 1219080-61-1IMIDAZOLE-2-BORONIC ACID
Synthetic route


- 10294-34-5
boron trichloride
Conditions

| Conditions | Yield |
|---|---|
| In melt Electrolysis; electrolysis in a chloride melt (400-500°C); | 100% |
| With chlorine 400°C;; 6-8 times vac. distn. spect. purity product obtained; | |
| With silver(I) chloride |
Conditions

| Conditions | Yield |
|---|---|
| byproducts: HCl, ClCOCl; at room temp.;; | A n/a B 96% |
...Expand


A

- 10294-34-5
boron trichloride

B

- 64486-90-4
1,3-di-tert-butyl-2-chloro-2,3-dihydro-1H-1,3,2-diazaborole
Conditions

| Conditions | Yield |
|---|---|
| With sodium amalgam In hexane Ar atmosphere, stiring (overnight in the dark); decantation, filtration, removement of volatiles (vacuum), sublimation (40 to 60°C, 0.01 Torr); | A n/a B 91% |
Conditions

| Conditions | Yield |
|---|---|
| With hydrogen condensation of HBCl2/BCl3/B2H6 mixt. on molecular sieve, heating in H2 stream, BCl3 not adsorbed, B2H6 easily desorbed, strongly adsorbed HBCl2 slowly symmetrysied; | A n/a B 90% |
| With H2 condensation of HBCl2/BCl3/B2H6 mixt. on molecular sieve, heating in H2 stream, BCl3 not adsorbed, B2H6 easily desorbed, strongly adsorbed HBCl2 slowly symmetrysied; | A n/a B 90% |
| equil. const. Kx determined at 273 K; |
Conditions

| Conditions | Yield |
|---|---|
| In tetrachloromethane 100°C, 2 weeks; NMR monitoring; not isolated, detected by NMR; | A 88% B n/a C n/a |
...Expand


- 7446-70-0
aluminium trichloride


- 7637-07-2
boron trifluoride

A

- 7784-18-1
aluminum(III) fluoride

B

- 10294-34-5
boron trichloride
Conditions

| Conditions | Yield |
|---|---|
| heating with excess of BF3; condensation of BCl3 in an U-tube cooled the mixt. of CO2 and alcohol; fractionated distn.; | A n/a B 80% |
| heating with excess of BF3; condensation of BCl3 in an U-tube cooled the mixt. of CO2 and alcohol; fractionated distn.; | A n/a B 80% |
...Expand


- 89033-11-4
1,3-bis(dichloroboryl)propane


- 13517-10-7
boron triiodide

A

- 157426-91-0
1,3-bis(diiodoboryl)propane

B

- 10294-34-5
boron trichloride
Conditions

| Conditions | Yield |
|---|---|
| removal of BCl3 in vacuo;; distillation in vacuo after 4 h;; | A 78% B n/a |
...Expand



- 13701-67-2
diboron tetrachloride

A

- 17927-57-0
monochlorodiborane

C

- 10325-39-0
dichloroborane

D

- 10294-34-5
boron trichloride

E

- 19287-45-7
diborane
Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction carried out in Pyrex tubes to which Teflon valves had been sealed or in a N2-filled glovebox; reaction of B2Cl4 with excess B5H9 in a 1:3 mole ratio; vessel allowed to warm from -196 ° C to ambient temp., 20 min at ambient temp.,; fractionation, yield of 1-BCl2B5H8 based upon amt. of B2Cl4 employed: 73%, based upon amt. of B5H9 consumed: 95%; small amts. of B2H6, BCl2H, B2H5Cl, also material insufficiently volatileto move into vac. line formed;; | A n/a B 73% C n/a D n/a E n/a |
...Expand


- 3677-81-4
diphenylboronchloride


- 13517-10-7
boron triiodide

A

- 17933-05-0
(diphenyl)iodoborane

B

- 10294-34-5
boron trichloride
Conditions

| Conditions | Yield |
|---|---|
| heating at 130-140°C for 4 h; distn.; | A 72% B n/a |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| 800-1000°C; ratio B2O3:NaCl=3:1; | 60% |
| 800-1000°C; ratio B2O3:NaCl=1:1; | 30% |
| 0% |
Raw Materials
Hot Products
- 25038-59-9Poly(oxy-1,2-ethanediyloxycarbonyl-1,4-phenylenecarbonyl)
- 10040-45-6Sodium picosulfate
- 55399-93-44-Hydroxyisoleucine
- 73398-61-5Glycerides, mixeddecanoyl and octanoyl
- 15914-84-81-Naphthalenemethanol, a-methyl-, (aS)-
- 98-95-3Benzene, nitro-
- 143-19-1Sodium oleate
- 140-95-4Urea,N,N'-bis(hydroxymethyl)-
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
- Total:22 Page 1 of 1 1
Chemistry
Boron trichloride(10294-34-5) exists as clear, colorless liquid or gas, fumes in air. Its colorless gas is valuable in organic synthesis.
Molecular Structure: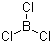
Molecular Formula:BCl3
Molecular Weight:117.16
EINECS:233-658-4
Density:0.738 g/cm3
Melting Point:-107 ℃
Boiling Point:12.5 ℃ at 760 mmHg
Flash Point:-17 ℃
Solubility:Decomposes in water
Molecular Structure:
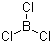
Molecular Formula:BCl3
Molecular Weight:117.16
EINECS:233-658-4
Density:0.738 g/cm3
Melting Point:-107 ℃
Boiling Point:12.5 ℃ at 760 mmHg
Flash Point:-17 ℃
Solubility:Decomposes in water
Uses
Boron trichloride(10294-34-5) is originally used for production of elemental boron.
Toxicity Data With Reference
| 1. | ihl-rat LCLo:20 ppm/7H | 14KTAK Boron, Metallo-Boron Compounds and Boranes R.M. Adams,New York, NY.: Wiley,1964,726. | ||
| 2. | ihl-mus LCLo:20 ppm/7H | 14KTAK Boron, Metallo-Boron Compounds and Boranes R.M. Adams,New York, NY.: Wiley,1964,726. |
Consensus Reports
Reported in EPA TSCA Inventory. EPA Extremely Hazardous Substances List.
Safety Profile
A poison by inhalation. A corrosive and severe irritant to skin, eyes, and mucous membranes. Reacts with water or steam to produce heat, toxic and corrosive fumes. Violent reaction with aniline or phosphine. Incompatible with hexafluorisopropylidene amino lithium, NO2, grease, organic matter, O2. When heated to decomposition it emits toxic fumes of Cl−. See also BORON COMPOUNDS and HYDROCHLORIC ACID.
Hazard Codes: T+,T,
T+,T, N,
N, F
F
Risk Statements: 14-26/28-36/37/38-40-67-65-62-51/53-48/20-34-11-50/53-26/27/28-63-39/23/24/25-24-21-10
14: Reacts violently with water
26/28: Very Toxic by inhalation and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
40: Limited evidence of a carcinogenic effect
67: Vapors may cause drowsiness and dizziness
65: Harmful: May cause lung damage if swallowed
62: Possible risk of impaired fertility
51/53: Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
48/20: Harmful: danger of serious damage to health by prolonged exposure through inhalation
34: Causes burns
11: Highly Flammable
50/53: Very Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
26/27/28: Very Toxic by inhalation, in contact with skin and if swallowed
63: Possible risk of harm to the unborn child
39/23/24/25: Toxic: danger of very serious irreversible effects through inhalation, in contact with skin and if swallowed
24: Toxic in contact with skin
21: Harmful in contact with skin
10: Flammable
Safety Statements: 9-26-28-36/37/39-45-8-61-38-28A-16-1-60-33-23-7/9-62
9: Keep container in a well-ventilated place
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
28: After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
8: Keep container dry
61: Avoid release to the environment. Refer to special instructions safety data sheet
38: In case of insufficient ventilation, wear suitable respiratory equipment
28: After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
16: Keep away from sources of ignition - No smoking
1: Keep locked up
60: This material and/or its container must be disposed of as hazardous waste
33: Take precautionary measures against static discharges
23: Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer)
7/9: Keep container tightly closed and in a well-ventilated place
62: If swallowed, do not induce vomiting: seek medical advice immediately and show this container or label
RIDADR: UN 3390 6.1/PG 1
WGK Germany: 3
RTECS: ED1925000
F: 10-21
F 10 Keep under argon
F 12 Shake before use
Hazard Note: Very toxic
HazardClass: 2.3
Hazard Codes:
 T+,T,
T+,T, N,
N, F
F Risk Statements: 14-26/28-36/37/38-40-67-65-62-51/53-48/20-34-11-50/53-26/27/28-63-39/23/24/25-24-21-10
14: Reacts violently with water
26/28: Very Toxic by inhalation and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
40: Limited evidence of a carcinogenic effect
67: Vapors may cause drowsiness and dizziness
65: Harmful: May cause lung damage if swallowed
62: Possible risk of impaired fertility
51/53: Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
48/20: Harmful: danger of serious damage to health by prolonged exposure through inhalation
34: Causes burns
11: Highly Flammable
50/53: Very Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
26/27/28: Very Toxic by inhalation, in contact with skin and if swallowed
63: Possible risk of harm to the unborn child
39/23/24/25: Toxic: danger of very serious irreversible effects through inhalation, in contact with skin and if swallowed
24: Toxic in contact with skin
21: Harmful in contact with skin
10: Flammable
Safety Statements: 9-26-28-36/37/39-45-8-61-38-28A-16-1-60-33-23-7/9-62
9: Keep container in a well-ventilated place
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
28: After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
8: Keep container dry
61: Avoid release to the environment. Refer to special instructions safety data sheet
38: In case of insufficient ventilation, wear suitable respiratory equipment
28: After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
16: Keep away from sources of ignition - No smoking
1: Keep locked up
60: This material and/or its container must be disposed of as hazardous waste
33: Take precautionary measures against static discharges
23: Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer)
7/9: Keep container tightly closed and in a well-ventilated place
62: If swallowed, do not induce vomiting: seek medical advice immediately and show this container or label
RIDADR: UN 3390 6.1/PG 1
WGK Germany: 3
RTECS: ED1925000
F: 10-21
F 10 Keep under argon
F 12 Shake before use
Hazard Note: Very toxic
HazardClass: 2.3
Standards and Recommendations
DOT Classification: 2.3; Label: Poison Gas, Corrosive

