T. Ishida et al. / Journal of Catalysis 374 (2019) 320–327
321
we examined the recyclability of Pd/ZrO2 and Pd/CeO2. Pd/CeO2
was deactivated with consecutive runs due to the aggregation of
single Pd atoms to Pd nanoparticles. The question then arises what
is the real active size of Pd on CeO2; can single Pd atoms catalyze
the decarbonylation (just less reactive than small Pd clusters) or
are aggregated Pd clusters formed in-situ the active species?
In-situ X-ray absorption fine structure (XAFS) analysis has been
attracting growing interest as a powerful tool to elucidate both
changes in oxidation states and size of metal particles under sim-
ilar conditions in catalyst preparation [9] or catalytic reactions
[10]. To answer the above question, we exploited the in-situ XAFS
technique to disclose the active size of Pd clusters and aggregation
behavior of single Pd atoms on CeO2 during the reaction. We inves-
tigated a difference between ZrO2 and CeO2 as supports in terms of
stabilization of Pd atoms or clusters during the decarbonylation by
in-situ XAFS. We found that ZrO2 stabilized Pd clusters played an
important role to keep the Pd particle size, whereas Pd single
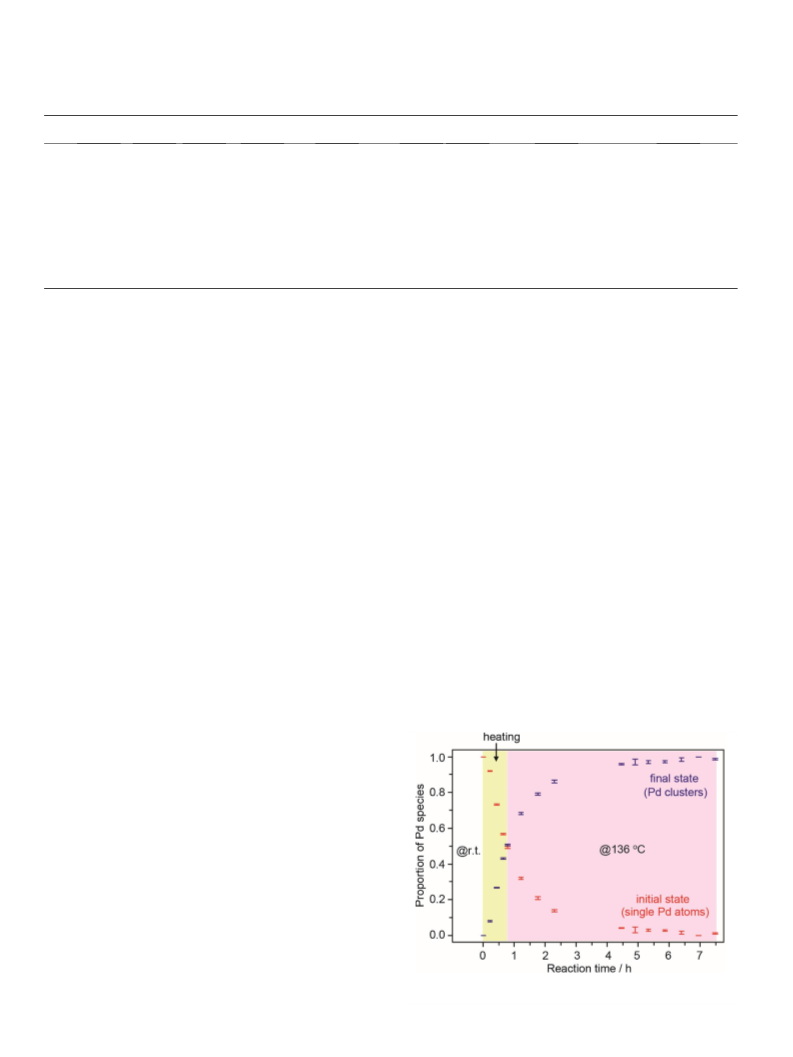
atoms on CeO2 were aggregated to form Pd13 clusters during heat-
ing and the reaction. Although single Pd atoms on CeO2 were
aggregated during the reaction, the results of in-situ XAFS analysis
suggested that the size of Pd clusters did not increase gradually.
Accordingly, Pd clusters having a certain size (i.e., Pd13 clusters)
were formed, and the proportion of Pd13 clusters increased with
time. Therefore, the formation of Pd13 clusters is responsible for
the decarbonylation in the case of Pd/CeO2. A different feature
depending on the support was also discussed by density functional
theory (DFT) study.
with a typical current of 99.5 mA. Pd K-edge (24.32 keV) XAFS
spectra were measured using a Si(3 1 1) double crystal monochro-
mator in a transmission mode. Ionization chambers were used to
measure the intensity of the incident and transmitted X-rays and
the quick scan technique (QXAFS) was used. Spectral analyses were
carried out using the XAFS analysis software, Athena and Artemis
[12]. In the curve-fitting analysis, backscattering amplitude, phase
shift, and mean free path of the photoelectrons were calculated by
FEFF8.4 [13] and then the other parameters, including the number
of neighboring atoms, interatomic distance between the adsorbed
atom to the neighboring atom, the Debye-Waller factor, and
absorption edge energy, were treated as fitting parameters. The
intrinsic loss factor was obtained by curve-fitting analysis of the
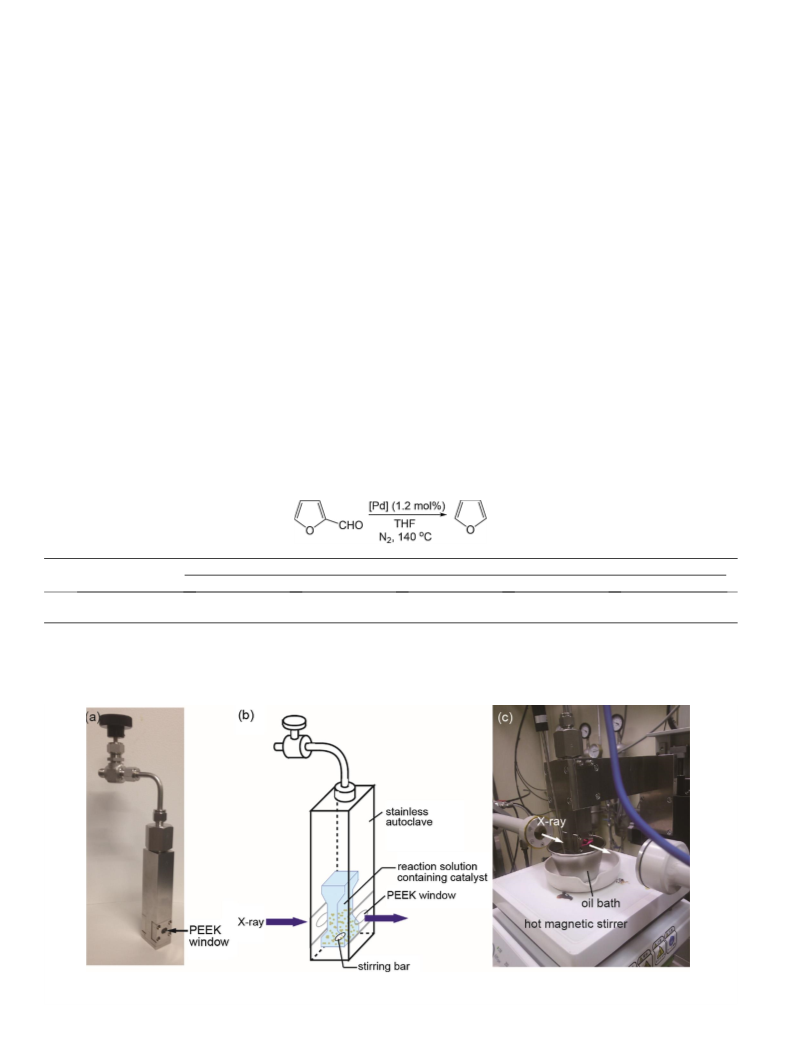
EXAFS data for the Pd foil. Autoclaves with PEEK windows for in-
situ XAFS were purchased from UNISOKU Co., Ltd. The optical path
length was 2 mm. PdO/ZrO2 and PdO/CeO2 were pretreated in THF
(1.5 mL) under H2 (2 MPa) at 120 °C for 2 h in an autoclave to give
Pd/ZrO2 and Pd/CeO2, respectively. After removing THF under vac-
uum, the Pd catalysts were transferred into the autoclave for in-
situ XAFS. Furfural (1 mmol), mesitylene (1.5 mL), and a magnetic
stir bar were added. The autoclave was purged and filled with N2
(0.1 MPa). The mixture was stirred at 140 °C in an oil bath. XAFS
were obtained without stirring while the measurement. Pd particle
size was estimated from the coordination number (CN) as an icosa-
hedron spherical particle according to the literature [14].
2.5. Temperature-programmed reduction (H2-TPR)
2. Experimental
H2-TPR measurements were performed using a BELCAT (Micro-
tracBEL Corp.) equipped with a thermal conductivity detector. The
catalysts (PdO/ZrO2 and PdO/CeO2) (0.1 g) were placed in a quartz
tube. The H2-TPR measurements were carried out without pre-
treatment in a flow of 5% H2/Ar (50 mL/min) up to 200 °C.
2.1. Materials
ZrO2 (JRC-ZRO-6, 280 m2/g) and CeO2 (HS, 173 m2/g) were sup-
plied from the Catalysis Society of Japan (Tokyo, Japan), and Daiichi
Kigenso Kagaku Kogyo (Osaka, Japan), respectively. An aqueous
solution of Pd(NO3)2 (200 g/L) was purchased from Tanaka Kikin-
zoku Kogyo (Tokyo, Japan). Furfural was passed through a short
activated alumina column (Wako, 300 mesh) before use. Other
reagents were purchased and used without further purifications.
2.6. Calculation
Ab-initio calculations were performed to determine the struc-
tural relaxation and electronic states. Vienna Ab initio simulation
package (VASP) [15] code was used based on the projector-
augmented-wave (PAW) method [16] for the actual calculations.
2.2. Catalyst preparation
Perdew-Burke-Ernzerhof (PBE) [17] with
a cut-off energy of
500 eV was used to determine the exchange-correlation term.
PdO/ZrO2 and PdO/CeO2 (2 wt% of Pd loading) were prepared by
impregnation. An aqueous solution of Pd(NO3)2 (Tanaka Kikinzoku
Kogyo) was impregnated into the metal oxides, and the mixture
was stirred at room temperature for 30 min. Water was removed
by vacuum-freeze-drying. The solid was calcined in air at 550 °C
for 4 h to obtain PdO/MOx.
3. Results and discussion
For liquid phase decarbonylation of furfural, activated carbons
and Al2O3 have been frequently used as supports. In our previous
report [7], Pd/ZrO2 and Pd/CeO2 exhibited superior catalytic activ-
ity to Pd/C or Pd/Al2O3. The size of Pd particles decreased with an
increase in specific surface area of ZrO2 and CeO2 Pd clusters con-
sisting of less than 10 Pd atoms and single Pd atoms could be sta-
bilized on high-surface-area ZrO2 and CeO2, respectively. Other
metal oxides and carbonaceous materials could not stabilize Pd
as clusters even though their surface areas were as high as those
of ZrO2 and CeO2. The obtained small Pd clusters on ZrO2 exhibited
high catalytic activity for decarbonylation of furfural without addi-
tives such as a base. According to ex-situ XAFS, clusters consisting
of 3–5 Pd atoms were deposited on ZrO2. On the other hand, the
peaks corresponding to PdAO and PdACe (second coordination
shell) bond distances were observed despite the lack of the peak
for PdAPd bond, suggesting that Pd atoms were present as single
atoms and attached to CeO2 surface.
2.3. Typical procedure for decarbonylation of furfural
An autoclave was charged PdO/MOx (Pd 0.4 mol% to furfural),
THF (1.5 mL), and a magnetic stir bar. The autoclave was pressur-
ized by H2 (2 MPa), and the mixture was stirred at 120 °C for 3 h
to obtain Pd/MOx. After cooling, furfural (1.0 mmol) was added,
and the autoclave was purged and filled with N2. The mixture
was stirred under N2 (0.1 MPa) at 150 °C for 12 h. The catalyst
was removed by filtration, and the filtrate was analyzed by gas
chromatography (Agilent GC6850 Series II equipped with a CO2
gas cylinder, flame ionization detector, and a capillary column
(J&W HP-1)) using tridecane as an internal standard.
2.4. In-situ XAFS measurement
In this work, we initially examine the durability of Pd/ZrO2 and
Pd/CeO2, and recycling tests were carried out. Prior to the reaction,
PdO/ZrO2 and PdO/CeO2 prepared by impregnation followed by
Pd K-edge XAFS measurements were carried out at BL14B2 of
SPring-8 (Hyogo, Japan) [11]. The storage ring energy was 8 GeV

 Ishida, Tamao
Ishida, Tamao
 Honma, Tetsuo
Honma, Tetsuo
 Nakada, Kengo
Nakada, Kengo
 Murayama, Haruno
Murayama, Haruno
 Mamba, Tetsuya
Mamba, Tetsuya
 Kume, Kurumi
Kume, Kurumi
 Izawa, Yusuke
Izawa, Yusuke
 Utsunomiya, Masaru
Utsunomiya, Masaru
 Tokunaga, Makoto
Tokunaga, Makoto