PÖPPL, SZALLER: PHASE DIAGRAM OF Bi
2
O
3
– TeO
2
SYSTEM
385
tion rate is determined by the diffusion of MO . Bi O rich phases were detected as
2
2
3
reaction products.
In both cases the Bi O is the stationary phase and the other reactant oxide
2
3
(
MO ) is the moving phase. The composition of the product depends on the volatility
x
of MO . If it is very low, the Bi O rich phases arise. But if the volatility of MO is
x
2
3
x
sufficiently high, the MO -rich phases are the products. The vapor pressure of solid
x
TeO is 0.438 Pa at 605°C and 3.62 Pa at 650°C, respectively [19].
2
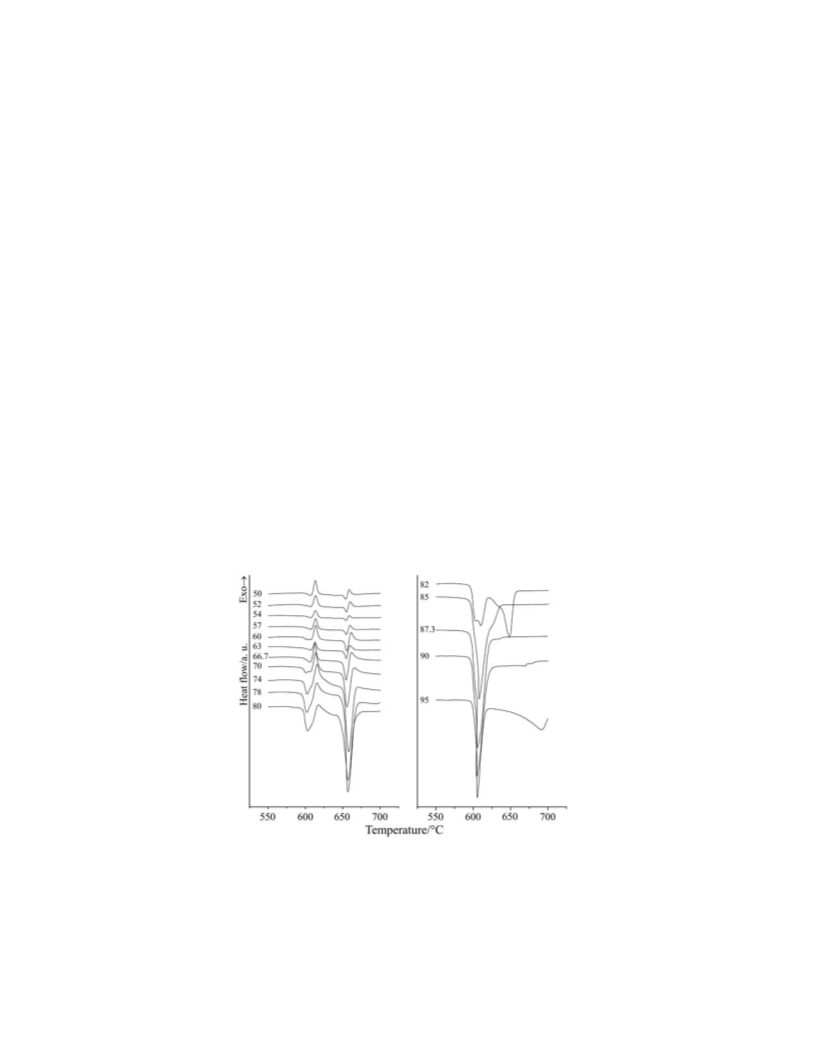
Applying linear heating rate the following reaction pathways can be established
on the base of DSC and electron microprobe analyses results. The reaction starts by
grain boundary diffusion. But at the same time, from 450°C the TeO evaporates onto
2
the Bi O grains producing TeO layer. This TeO layer reacts with the Bi O by bulk
2
3
2
2
2
3
diffusion forming Bi Te O . When the amount of Bi Te O is sufficient, the
2
4
11
2
4
11
eutectic forms and melts. The reaction rate increases in the liquid phase. The conver-
sion of Bi Te O completes and the melt is solidified. When increasing the tempera-
2
4
11
ture the Bi Te O melts and reacts with Bi O forming Bi Te O , which is solidified.
2
4
11
2
3
2
2
7
The Bi O +Bi Te O reaction is taking place by bulk diffusion at higher temperature.
2
3
2
2
7
The formation of structures of samples cooled down from melt is controlled by cat-
ion-ordering processes. Ions have to move from one site to another through strong poten-
tial walls of other ions. This ‘kinetic hindrance’ can be strong and require long annealing
–1
time [20]. The 10°C min cooling rate applied is relatively high and permits the forma-
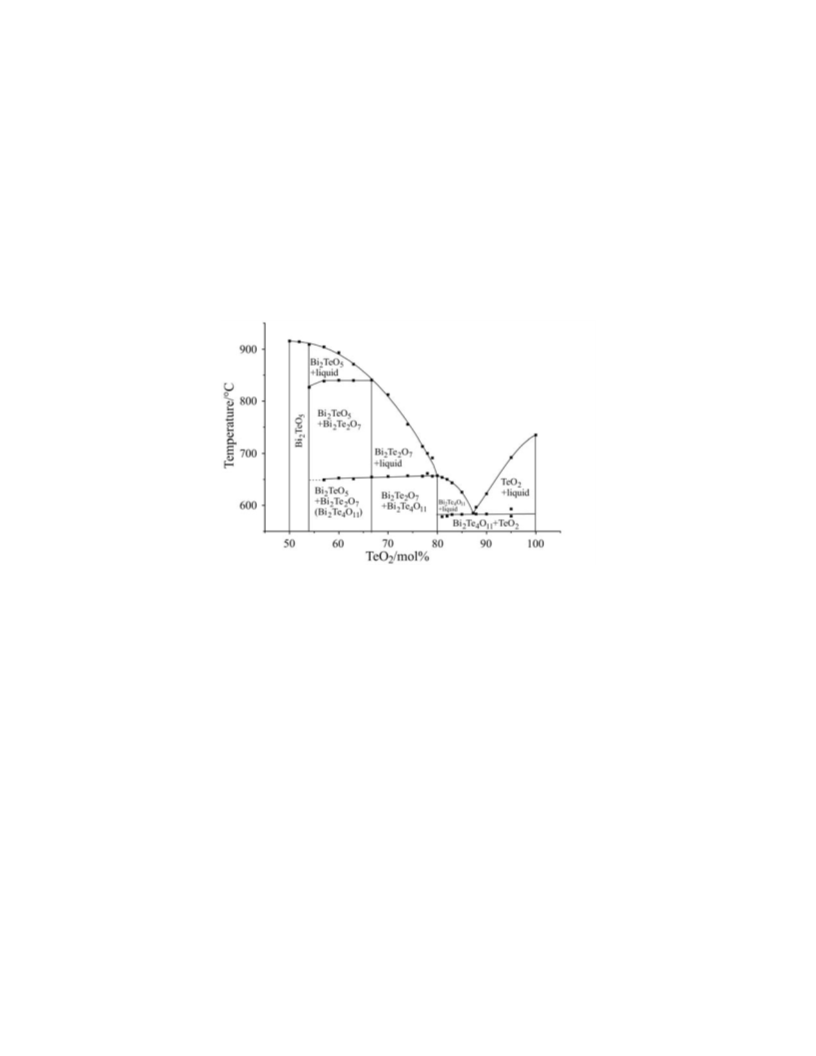
tion of metastable phases (β-Bi Te O , Bi Te O ) with cubic symmetry between
2
4
11
2
7
17
54–100% composition range. Though stable phases form during the subsequent heating,
the phase diagram has been determined under non-equilibrium conditions.
*
* *
This work was supported by the Hungarian Scientific Research Found OTKA (Grants T 026647).
References
1
2
3
4
5
6
I. Földvári, Á. Péter, R. Voszka and L. A. Kappers, J. Cryst. Growth, 100 (1990) 75.
B. Frit, M. Jaymes and P. Hagenmuller, Rev. Chim. Min., 8 (1971) 453.
B. Frit and M. Jaymes, Rev. Chim. Min., 9 (1972) 837.
D. Mercurio, M. El. Farassi, B. Frit and P. Goursat, Mater. Chem. Phys., 9 (1983) 467.
M. El. Farassi, D. Mercurio and B. Frit, Mater. Chem. Phys., 16 (1987) 133.
D. Mercurio, B. Frit, G. Harburn, B. H. Parry, R. P. Williams and R. J. D. Tilley,
Phys. Stat. Sol. (a), 108 (1988) 111.
7
8
9
D. Mercurio, B. H. Parry, B. Frit, G. Harburn, R. P. Williams and R. J. D. Tilley, J. Sol. State
Chem., 92 (1991) 449.
B. Frit, D. Mercurio, B. H. Parry, G. Harburn, R. P. Williams and R. J. D. Tilley, J. Sol. State
Chem., 116 (1995) 240.
D. Mercurio, J. C. Champarnaud-Mesjard, I. Gouby and B. Frit, Eur. J. Sol. State Inorg. Chem.,
35 (1998) 49.
1
0 A. K. Jakhkind, P. S. Martyshhenko, Izv. Akad. Nauk. SSSR Neorg. Matter., 9 (1973) 2186.
J. Therm. Anal. Cal., 74, 2003

 Poeppl
Poeppl
 Szaller, Zsuzsanna
Szaller, Zsuzsanna