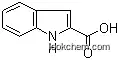
1477-50-5Relevant articles and documents
Improved preparation method of indole-2-formic acid as starting material of perindopril
-
Paragraph 0039-0049, (2021/07/10)
The invention discloses an improved preparation method of indole-2-formic acid by using perindopril as a starting raw material, which comprises the following steps: S1, taking 2-bromobenzaldehyde and ethyl acetate as raw materials, under the action of sodium ethoxide, carrying out Claisen-Schmidt condensation reaction to synthesize ethyl 2-bromocinnamate; s2, carrying out cyclization reaction on the ethyl 2-bromocinnamate in an amide solvent by using cuprous halide as a coupling catalyst and sodium azide as a nitrogen source to synthesize the indole-2-formic acid in one pot. According to the invention, 2-bromobenzaldehyde is used as a raw material for preparation, ethyl 2-bromocinnamate is firstly prepared, then cuprous halide is used as a coupling catalyst, sodium azide is used as a nitrogen source, cyclization reaction is carried out in an amide solvent, indole-2-formic acid is synthesized in one pot, and the HPLC content is 99% or above.
Visible-Light-Promoted Efficient Aerobic Dehydrogenation of N-Heterocycles by a Tiny Organic Semiconductor Under Ambient Conditions
Su, Chenliang,Yu, Kunyi,Zhang, Hanjie,Zhu, Yongfa
supporting information, p. 1956 - 1960 (2020/04/10)
An efficient reusable catalytic system has been developed based on perylene diimide (PDI) organic semiconductor for the aerobic dehydrogenation of N-heterocycles with visible light. This practical catalytic system without any additives proceeds under ambient conditions. The minute aggregates of PDI molecules on the surface of SiO2 nanospheres form tiny organic semiconductors, resulting in high-efficiency photo-oxidative activity. Notably, the robustness of this method is demonstrated by the synthesis of a wide range of N-heteroarenes, gram-scale experiments as well as reusability tests.
Design and synthesis of thiadiazolo-carboxamide bridged β-carboline-indole hybrids: DNA intercalative topo-IIα inhibition with promising antiproliferative activity
Tokala, Ramya,Sana, Sravani,Lakshmi, Uppu Jaya,Sankarana, Prasanthi,Sigalapalli, Dilep Kumar,Gadewal, Nikhil,Kode, Jyoti,Shankaraiah, Nagula
, (2020/10/27)
The conjoining of salient pharmacophoric properties directing the development of prominent cytotoxic agents was executed by constructing thiadiazolo-carboxamide bridged β-carboline-indole hybrids. On the evaluation of in vitro cytotoxic potential, 12c exhibited prodigious cytotoxicity among the synthesized new molecules 12a–k, with an IC50 50 value of 2.82 ± 0.10 μM. Besides, another compound 12a also displayed impressive cytotoxicity against A549 cell line (IC50: 3.00 ± 1.40 μM). Further target-based assay of these two compounds 12c and 12a revealed their potential as DNA intercalative topoisomerase-IIα inhibitors. Additionally, the antiproliferative activity of compound 12c was measured in A549 cells by traditional apoptosis assays revealing the nuclear, morphological alterations, and depolarization of membrane potential in mitochondria and externalization of phosphatidylserine in a concentration-dependent manner. Cell cycle analysis unveiled the G0/G1 phase inhibition and wound healing assay inferred the inhibition of in vitro cell migration by compound 12c in lung cancer cells. Remarkably, the safety profile of compound 12c was disclosed by screening against normal human lung epithelial cell line (BEAS-2B: IC50: 71.2 ± 7.95 μM) with a selectivity index range of 14.9–25.26. Moreover, Molecular modeling studies affirm the intercalative binding of compound 12c and 12a in the active pocket of topo-IIα. Furthermore, in silico prediction of physico-chemical parameters divulged the propitious drug-like properties of the synthesized derivatives.