53-16-7 Usage
Description
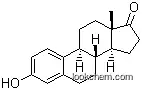
Estrone is one of the three naturally occurring estrogens, the others being estradiol and estriol. Estrone is synthesized from androstenedione by the aromatase enzyme system in the ovaries and placenta, and is also synthesized from estradiol by 17-hydroxy steroid dehydrogenase in the liver.Serum concentrations of estrone in premenopausal women fluctuate according to the menstrual cycle and becomes the most predominant estrogen in postmenopausal women.The binding affinities of estrone to the estrogen receptors α and β are approximately 60% and 37% relative to estradiol.
Chemical Properties
Estrone is an odorless white crystalline powder.Estrone is supplied as a crystalline solid. A stock solution may be made by dissolving the estrone in an organic solvent purged with an inert gas. Estrone is soluble in organic solvents such as DMSO and dimethyl formamide (DMF). The solubility of estrone in these solvents is approximately 20 mg/ml.Estrone is sparingly soluble in aqueous buffers. For maximum solubility in aqueous buffers, estrone should first be dissolved in DMF and then diluted with the aqueous buffer of choice. Estrone has a solubility of approximately 0.15 mg/ml in a1:5 solution of DMF:PBS (pH 7.2) using this method. We do not recommend storing the aqueous solution for more than one day.
Originator
Estrone,Abbott
Uses
Different sources of media describe the Uses of 53-16-7 differently. You can refer to the following data:
1. Estrone is a metabolite of 17β-Estradiol (E888000). During the metabolism, it is in rapid equilibrium with Estriol (E888960) and 17β-Estradiol (E888000) (1). Causes the feminization of male fish at human and animal waste sites (2).��This compound is a contaminant of emerging concern (CECs). Drinking water contaminant candidate list 3 (CCL 3) compound as per United States Environmental Protection Agency (EPA), environmental, and food contaminants.
2. Estrone is a weak form of estrogen and exists as estrone sulphate. Estrone is a luteolytic estrogen produced by the corpus luteum. In the follicle, estrone is synthesized from androstenedione by the action of cytochrome P450 aromatase.Estrone has been used:as medium supplement for hormone based degranulation studies of natural killer cells.as an endocrine disrupting compound for screening bacterial biosensor in toxic water.as medium component for monitoring fatty acid synthase (FASN) activity in breast adenocarcinoma cell lines.
Definition
ChEBI: A 17-oxo steroid that is estra-1,3,5(10)-triene substituted by an hydroxy group at position 3 and an oxo group at position 17.
Manufacturing Process
1-Vinyl-1,2,3,4-tetrahydronaphthalene-1,6-diol reacts with 2-
methylcyclopentane-1,3-dione in the presence of Triton B in tert-butanol gives
a good yield of δ1,3,5(10),9(11)-8,14-secoestratetraen-3-ol-14,17-dione, melting
point 124°-126°C (from methanol).δ1,3,5(10),9(11)-8,14-Secoestratetraen-3-ol-14,17-dione under influence of
hydrochloric acid in tetrahydrofurane cyclises into δ1,3,5(10),8,14-estrapentaen-
3-ol-17-one, melting point 216°-218°C.δ1,3,5(10),8,14-Estrapentaen-3-ol-17-one is converted to d,l-8-dehydroestrone
by selective hydrogenation with hydrogen, melting point 251°-254°C (from
methanol). Exhaustive hydrogenation of δ1,3,5(10),8,14-estrapentaen-3-ol-17-
one give d,l-8-isoestrone.d,l-8-Isoestrone in the presence of hydrochloric acid in tetrahydrofurane
isomerizes into d,l-9(11)-dehydroestrone, melting point 262°-265°C (from
alcohol).Hydrogenation of d,l-9(11)-dehydroestrone in tetrahydrofuran in the presence
of Pd/CaCO3 yields the estrone, melting point 251°-252°C (from acetone).
Brand name
Estrogenic Substance (Wyeth); Theelin (Parkdale).
Therapeutic Function
Estrogen
General Description
Estrone, 3-hydroxyestra-1,3,5(10)-trien-17-one, is less active than estradiol but more active than itsmetabolite, estriol. As the salt of its 3-sulfate ester, estroneis the primary ingredient in conjugated estrogens, USP, andesterified estrogens, USP. Although originally obtainedfrom the urine of pregnant mares (about 10 mg/L), estroneis now prepared synthetically. Estrone itself is not availablein commercial oral formulations, but can be obtained at compounding pharmacies as a topical formulation. Oleoylestrone,the C3 ester of estrone with oleic acid, is in phase IIclinical trials for the treatment of obesity. This acyl estronederivative reduces fat stores by a mechanism not involvingthe ER, although some of the oleoyl-estrone is hydrolyzedto estrone in vivo.
Biochem/physiol Actions
Estrone is an agonist for the estrogen receptor. The estradiol to estrone interconversion is favourable in menopause. Oral hormone replacement therapy (HRT) of estradiol-17β increases circulating levels of estrone.
Safety Profile
Confirmed carcinogen
with experimental carcinogenic,
neoplastigenic, tumorigenic, and teratogenic
data. A poison by intraperitoneal and
subcutaneous routes. Human reproductive
effects by implantation: spermatogenesis
and impotence. Mutation data reported. A
steroid drug for the treatment of menopause
and ovariectomy symptoms. When heated to
decomposition it emits acrid smoke and
irritating fumes.
Synthesis
Estrone, 3-hydroxyestra-1,3,5(10)-trien-17-one (28.1.9), has been made synthetically
in various ways. According to one of the first and most simple schemes, synthesis
was carried out in the following manner. Condensation of 3-methoxyphenylacetylene
with bicyclohexane-1,5-dione in a Favorskii reaction conditions lead to the corresponding
carbinol (28.1.1). The triple bond was reduced by hydrogen over a palladium catalyst,
forming the tertiary alcohol (28.1.2), which was then dehydrated in acidic conditions to
give the compound (28.1.3). Intramolecular alkylation of this compound in the presence of
anhydrous aluminum chloride formed a tetracyclic ketone (28.1.4), which during condensation
with benzaldehyde was transformed into an eneone (28.1.5). This was methylated
at the β-position relative to the keto-group by methyl iodide in the presence of potassium
tert-butylate, and the resulting compound (28.1.6) underwent ozonolysis, forming the
dicarboxylic acid (28.1.7). Cyclization of this compound to a cyclopentanone derivative
lead to the formation of methyl ester of the desired estrone (28.1.8), and demethylation of the phenolic hydroxyl group by hydrobromic acid formed the desired estrone (28.1.9).
Potential Exposure
Synthesized from ergosterol. Used in
combination with progestogen as an oral contraceptive.
Shipping
UN3249 Medicine, solid, toxic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials.
Purification Methods
Purify estrone by chromatography on silica gel, eluting with 2:1 hexane/EtOAc and recrystallising from EtOH or Et2O/EtOH. [Danishefsky & Cain J Am Chem Soc 98 4975 1976.] The acetate [901-93-9] crystallises from EtOH with m 125-127o. [Beilstein 8 III 1171.]
Incompatibilities
May react exothermically with reducing
agents to generate flammable gaseous hydrogen.
Incompatible with oxidizers (chlorates, nitrates, peroxides,
permanganates, perchlorates, chlorine, bromine, fluorine,
etc.); contact may cause fires or explosions. Keep away
from alkaline materials, strong bases, strong acids, oxoacids,
and epoxides.
Check Digit Verification of cas no
The CAS Registry Mumber 53-16-7 includes 5 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 2 digits, 5 and 3 respectively; the second part has 2 digits, 1 and 6 respectively.
Calculate Digit Verification of CAS Registry Number 53-16:
(4*5)+(3*3)+(2*1)+(1*6)=37
37 % 10 = 7
So 53-16-7 is a valid CAS Registry Number.
InChI:InChI=1/C18H22O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-16,19H,2,4,6-9H2,1H3/t14?,15-,16?,18+/m1/s1
53-16-7Relevant articles and documents
Mechanism of the hydrolysis of the sulfamate EMATE-an irreversible steroid sulfatase inhibitor
Spillane, William J.,Malaubier, Jean-Baptiste
, p. 2059 - 2062 (2010)
The kinetics of hydrolysis of the medicinally important sulfamate ester EMATE have been probed over a wide pH range and into moderately strong base (H_ region). Analysis of the pH/H_-rate profile, measurements of pKas, solvent-reactivity, kinetic isotope effects and determination of activation data reveal that in the pH range from ~1 to ~8 an SN2 (S) solvolytic mechanism is followed and after the pKa of EMATE (pKa ~9) is passed, a second pathway showing a first-order dependence on base operates and an E1cB mechanism is supported.
Marker et al.
, p. 1503 (1936)
Spiran isolation in the dienone-phenol rearrangement of steroidal p-quinols
Planas,Tomas,Bonet
, p. 471 - 474 (1987)
-
Synthetic approaches towards 4-functionalized estrone derivatives
Schoen, Uwe,Messinger, Josef,Solodenko, Wladimir,Kirschning, Andreas
, p. 3822 - 3828 (2012)
Directed ortho-lithiation of estrone carbamate followed by reaction with electrophiles afforded 2-substituted estrone derivatives. Reductive cleavage of the carbamate group followed by O-allylation and Claisen rearrangement led to new 4-functionalized est
An efficient steroid pharmacophore-based strategy to identify new aromatase inhibitors
Neves, Marco A.C.,Dinis, Teresa C.P.,Colombo, Giorgio,Sa e Melo, M. Luisa
, p. 4121 - 4127 (2009)
Aromatase, an enzyme involved in the conversion of androgens into estrogens, is an important target for the endocrine treatment of breast cancer. Aromatase inhibition is usually achieved with steroids structurally related to the substrate of catalysis or,
Me3SI-promoted chemoselective deacetylation: a general and mild protocol
Gurawa, Aakanksha,Kashyap, Sudhir,Kumar, Manoj
, p. 19310 - 19315 (2021/06/03)
A Me3SI-mediated simple and efficient protocol for the chemoselective deprotection of acetyl groups has been developedviaemploying KMnO4as an additive. This chemoselective deacetylation is amenable to a wide range of substrates, tolerating diverse and sensitive functional groups in carbohydrates, amino acids, natural products, heterocycles, and general scaffolds. The protocol is attractive because it uses an environmentally benign reagent system to perform quantitative and clean transformations under ambient conditions.
PhIO-Mediated oxidative dethioacetalization/dethioketalization under water-free conditions
Du, Yunfei,Ouyang, Yaxin,Wang, Xi,Wang, Xiaofan,Yu, Zhenyang,Zhao, Bingyue,Zhao, Kang
, p. 48 - 65 (2021/06/16)
Treatment of thioacetals and thioketals with iodosobenzene in anhydrous DCM conveniently afforded the corresponding carbonyl compounds in high yields under water-free conditions. The mechanistic studies indicate that this dethioacetalization/dethioketalization process does not need water and the oxygen of the carbonyl products comes from the hypervalent iodine reagent.
Solvent- and Wavelength-Dependent Photolysis of Estrone
Adriano, Natalie,Ahearn, Ceilidh,Black, Cory,Cracchiolo, Michael,Ghere, Daniel,Hare, Patrick M.,Nu?ez, Alexandra,Olivan, Lars,Patel, Raj,Saner, Stephanie,Smith, Krista R.,Watkins, Barbie
, (2021/11/08)
The direct photolysis of estrone in solvents ranging from water to cyclohexane is reported. The photodegradation is dominated by lumiestrone, an epimer of estrone resulting from the inversion of the methyl group at carbon 13, regardless of solvent and pho