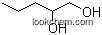
5343-92-0Relevant articles and documents
Baeyer-villiger oxidation of acyl carrier protein-tethered thioester to acyl carrier protein-linked thiocarbonate catalyzed by a monooxygenase domain in FR901464 biosynthesis
Tang, Man-Cheng,He, Hai-Yan,Zhang, Feng,Tang, Gong-Li
, p. 444 - 447 (2013)
Baeyer-Villiger monooxygenases (BVMOs), generally catalyzing the transformation of carbonylic compounds into the corresponding esters or lactones known as Baeyer-Villiger oxidation in organic chemistry, are widely distributed among microorganisms and have stimulated great interest as biocatalysts for organic synthesis. The physiological roles of this type of MOs are usually classified as degradation of organic compounds involved in primary metabolism. Recently, increasing numbers of BVMOs have been found to be involved in the biosynthesis of secondary metabolites, especially for postmodification; however, to date, none of them has been reported functionally as a tailoring domain within polyketide synthase (PKS) acting on carrier protein-tethered substrates. FR901464, an antitumor natural product that targets spliceosome and inhibits both splicing and nuclear retention of pre-mRNA, was elucidated to be biosynthesized by a hybrid acyltransferase-less PKS/nonribosomal peptide synthetase (NRPS) system. Within the hybrid system, an unprecedented domain that was proposed to mediate the chain release process was located in the termination module. In this paper, we report the in vitro biochemical characterization of this domain to be a BVMO tailoring domain that catalyzes the BV oxidation of an acyl carrier protein (ACP)-tethered thioester to an ACP-linked thiocarbonate, which represents the first example of BVMOs operating in cis within the PKS and NRPS biosynthetic paradigm.
Chromium-Catalyzed Production of Diols From Olefins
-
Paragraph 0111, (2021/03/19)
Processes for converting an olefin reactant into a diol compound are disclosed, and these processes include the steps of contacting the olefin reactant and a supported chromium catalyst comprising chromium in a hexavalent oxidation state to reduce at least a portion of the supported chromium catalyst to form a reduced chromium catalyst, and hydrolyzing the reduced chromium catalyst to form a reaction product comprising the diol compound. While being contacted, the olefin reactant and the supported chromium catalyst can be irradiated with a light beam at a wavelength in the UV-visible spectrum. Optionally, these processes can further comprise a step of calcining at least a portion of the reduced chromium catalyst to regenerate the supported chromium catalyst.
Unravelling the one-pot conversion of biomass-derived furfural and levulinic acid to 1,4-pentanediol catalysed by supported RANEY Ni-Sn alloy catalysts
Ansyah, Fathur Razi,Astuti, Maria Dewi,Hara, Takayoshi,Husain, Sadang,Mustikasari, Kamilia,Rodiansono,Shimazu, Shogo
, p. 241 - 250 (2022/01/19)
Bimetallic Ni-Sn alloys have been recognised as promising catalysts for the transformation of furanic compounds and their derivatives into valuable chemicals. Herein, we report the utilisation of a supported bimetallic RANEY nickel-tin alloy supported on aluminium hydroxide (RNi-Sn(x)/AlOH; x is Ni/Sn molar ratio) catalysts for the one-pot conversion of biomass-derived furfural and levulinic acid to 1,4-pentanediol (1,4-PeD). The as prepared RNi-Sn(1.4)/AlOH catalyst exhibited the highest yield of 1,4-PeD (78%). The reduction of RNi-Sn(x)/AlOH with H2 at 673-873 K for 1.5 h resulted in the formation of Ni-Sn alloy phases (e.g., Ni3Sn and Ni3Sn2) and caused the transformation of aluminium hydroxide (AlOH) to amorphous alumina (AA). The RNi-Sn(1.4)/AA 673 K/H2 catalyst contained a Ni3Sn2 alloy as the major phase, which exhibited the best yield of 1,4-PeD from furfural (87%) at 433 K, H2 3.0 MPa for 12 h and from levulinic acid (up to 90%) at 503 K, H2 4.0 MPa, for 12 h. Supported RANEY Ni-Sn(1.5)/AC and three types of supported Ni-Sn(1.5) alloy (e.g., Ni-Sn(1.5)/AC, Ni-Sn(1.5)/c-AlOH, and Ni-Sn(1.5)/γ-Al2O3) catalysts afforded high yields of 1,4-PeD (65-87%) both from furfural and levulinic acid under the optimised reaction conditions.
Hydrogenolysis of Furfuryl Alcohol to 1,2-Pentanediol Over Supported Ruthenium Catalysts
Yamaguchi, Aritomo,Murakami, Yuka,Imura, Tomohiro,Wakita, Kazuaki
, p. 731 - 736 (2021/06/12)
Hydrogenolysis of the furan rings of furfural and furfuryl alcohol, which can be obtained from biomass, has attracted attention as a method for obtaining valuable chemicals such as 1,2-pentanediol. In this study, we examined the hydrogenolysis of furfuryl alcohol to 1,2-pentanediol over Pd/C, Pt/C, Rh/C, and various supported Ru catalysts in several solvents. In particular, we investigated the effects of combinations of solvents and supports on the reaction outcome. Of all the tested combinations, Ru/MgO in water gave the best selectivity for 1,2-pentanediol: with this catalyst, 42 % selectivity for 1,2-pentanediol was achieved upon hydrogenolysis of furfuryl alcohol for 1 h at 463 K. In contrast, reaction in water in the presence of Ru/Al2O3 afforded cyclopentanone and cyclopentanol by means of hydrogenation and rearrangement reactions.
Selective Activation of C-OH, C-O-C, or Ca? C in Furfuryl Alcohol by Engineered Pt Sites Supported on Layered Double Oxides
An, Zhe,He, Jing,Jiang, Yitao,Ma, Xiaodan,Shu, Xin,Song, Hongyan,Xiang, Xu,Zhang, Jian,Zhang, Zhijun,Zhao, Wenfang,Zheng, Lirong,Zhu, Yanru
, p. 8032 - 8041 (2020/09/23)
The selective activation of targeted bonds in biomass-derived furfural or furfuryl alcohol with complex chemical linkages (C-C/C-H/C-O, Ca? C/Ca? O, or C-O-H/C-O-C) is of great challenge for biomass upgrading, expecting well-defined catalyst and definite catalytically active sites. This work demonstrates an efficient targeted activation to C-OH, C-O-C, or Ca? C by engineering the structure of catalytic Pt sites, affording 2-methylfuran (2-MF), tetrahydrofurfuryl alcohol (THFA), or 1,2-pentanediol (1,2-PeD) as product in the hydroconversion of furfuryl alcohol. The catalytic Pt sites have been engineered as atomic Pt, coordination unsaturated Pt-Pt in atom-thick dispersion, or coordination unsaturated 3D Pt-Pt by tailoring the Pt dispersion (single atom, 2D cluster, or 3D cluster) on Mg and Al-containing layered double oxide (Mg(Al)O) support. The selective activation of C-OH, C-O-C, or Ca? C has been traced with the FT-IR spectra recorded surface reaction. On atomic Pt, C-O-H is easily activated, with the assistance of Mg(Al)O support, with O-terminal adsorption without affecting furan C-O and Ca? C. However, Ca? C in the furan ring is easier to be activated on coordination-unsaturated Pt-Pt in atom-thick dispersion, resulting in a step-by-step hydrogenation to generate THFA. On coordination-unsaturated 3D Pt-Pt, the hydrogenolysis of furan ring is favored, resulting in the cleavage of furan C-O to produce 1,2-PeD. Also, the Mg(Al)O supports derived from Mg and Al layered double hydroxides (LDHs) here also play a key role in promoting the selectivity to 1,2-PeD by providing basic sites.
Highly selective 1-pentene epoxidation over Ti-MWW with modified microenvironment of Ti active sites
Jiang, Jingang,Tian, Wenwen,Wang, Bowen,Wu, Peng,Xu, Hao,Yin, Jianyong,Yin, Jinpeng
, p. 6050 - 6064 (2020/09/23)
A titanosilicate/H2O2catalytic system was applied to process the liquid-phase selective epoxidation of 1-pentene to 1,2-epoxypentane (EP). The effects of titanosilicate topology (MWW, MFI, MSE, MEL, MOR, and *BEA), solvent, H2O/H2O2ratio, catalyst amount, reaction temperature, pressure, and time on the EP production were investigated systematically. The Ti-MWW/H2O2/acetonitrile system exhibited the highest 1-pentene conversion of 72.9% together with high EP selectivity of 99.9% and H2O2utilization efficiency of 91.5%. Moreover, it was proved that the Ti active sites located inside the intralayer 10-membered ring sinusoidal channels catalyzed the epoxidation process primarily owing to their supplying more steric fitness for 1-pentene molecules. A piperidine (PI)-assisted structural rearrangement of Ti-MWW was performed to further enhance the catalytic activity, almost doubling the turnover number value. The evolution of the microenvironment of Ti active sites in this structural rearrangement process was carefully investigated, revealing the coordination of N atoms in PI molecules to the Ti atoms. More importantly, we identified that the hexa-coordinated Ti sites with the PI molecules as ligand could significantly accelerate H2O2activation, the effect of which far exceeded the inhibition effect caused by the electronegativity increase of Ti active sites.
Hydrogenation of furfural by noble metal-free nickel modified tungsten carbide catalysts
Bretzler, Patrick,Huber, Michael,K?hler, Klaus,Nickl, Simon
, p. 27323 - 27330 (2020/09/01)
Nickel-tungsten carbide catalysts convert furfural to high value products in a liquid phase catalytic reaction. The product distribution depends on the solvent and the Ni-W-ratio of the catalyst. In isopropyl alcohol a combination of Ni and WxC enables the opening of the furan ring to yield 1,2-pentanediol. Nickel accelerates the tungsten oxide reduction in the tungsten carbide catalyst synthesis and facilitates the carbon insertion. Nickel modified tungsten carbide is a promising, noble metal-free catalyst system for the upgrading of furfural based renewable resources. Its preparation is facilitated compared to unmodified tungsten carbide catalysts.
PROCESS OF PREPARATION OF 1,2-PENTANEDIOL FROM FURFURAL
-
Page/Page column 10-15, (2020/08/22)
The present invention relates to hydrogenolysis process for preparation of 1,2-pentanediol from furfural. In particular, the present invention provides a conversion of 1,2-pentanediol from furfural in presence of methanol and using a catalyst based on Rhodium on porous Manganese dioxide octahedral molecular sieve in a single step carried out at temperature range between 130 °C to 170 °C. The advantages of present invention is that it avoids formation of Intermediate-2 and directly give product 1,2-pentanediol with good selectivity over 1,5-pentanediol.
COMPOSITIONS COMPRISING ODORLESS 1,2-PENTANEDIOL
-
Paragraph 00316, (2020/04/25)
Suggested is a cosmetic or pharmaceutical or detergent composition comprising 1,2 pentanediol, wherein said 1,2-pentanediol is obtained from a process comprising the follow ing steps: (a) providing at least one starting material selected from furfuryl alcohol and furfural; (b) reacting at least one of said starting materials with hydrogen in the presence of a heterogeneous catalyst to form 1,2-pentanediol, wherein said heterogeneous catalyst comprises: one or more metals selected from the group consisting of platinum, rhodium, ruthenium, nickel, palladium and iridium in metallic form and/or one or more compounds of metals selected from the group consisting of platinum, rhodium, ruthenium, nickel, palladium and iridium; and one or more support materials selected from the group consisting of activated carbon, aluminum oxide, silicon dioxide, and silicon carbide; and (c) removing the 1,2-pentanediol thus obtained from the reaction mixture.
Hydro-Oxygenation of Furfural in the Presence of Ruthenium Catalysts Based on Al-HMS Mesoporous Support
Roldugina,Shayakhmetov,Maksimov,Karakhanov
, p. 1306 - 1315 (2019/11/03)
Ru-containing catalyst based on an Al-HMS mesoporous aluminosilicate was synthesized. The mesoporous support and the catalyst on its basis were characterized by the methods of low-temperature desorption/adsorption of nitrogen, temperature-programmed desorption of ammonia, transmission electron microscopy, X-ray photoelectron microscopy, and energy-dispersive X-ray fluorescence analysis. The synthesized catalyst was investigated in the hydrodeoxygenation of the model compound of bio-oil, furfural, in the presence of H2O. The reaction was carried out at initial hydrogen pressures of 1–7 MPa at 200°C–300°C temperature range. The results revealed that the synthesized catalyst displayed a high activity in the hydrotransformation of furfural. The conversion was 100% in 1 hr at a 5 MPa hydrogen pressure and 200°C.