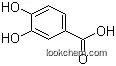
99-50-3Relevant academic research and scientific papers
Quercetin inhibits advanced glycation end product formation via chelating metal ions, trapping methylglyoxal, and trapping reactive oxygen species
Bhuiyan, Mohammad Nazrul Islam,Mitsuhashi, Shinya,Sigetomi, Kengo,Ubukata, Makoto
, p. 882 - 890 (2017)
Physiological concentration of Mg2+, Cu2+, and Zn2+ accelerated AGE formation only in glucosemediated conditions, which was effectively inhibited by chelating ligands. Only quercetin (10) inhibited MGO-mediated AGE formation as well as glucoseand ribose-mediated AGE formation among 10 polyphenols (1-10) tested. We performed an additional structure-activity relationship (SAR) study on flavanols (10, 11, 12, 13, and 14). Morin (12) and kaempherol (14) showed inhibitory activity against MGO-mediated AGE formation, whereas rutin (11) and fisetin (13) did not. These observations indicate that 3,5,7,4'-tetrahydroxy and 4-keto groups of 10 are important to yield newly revised mono-MGO adducts (16 and 17) and di-MGO adduct (18) having cyclic hemiacetals, while 3'-hydroxy group is not essential. We propose here a comprehensive inhibitory mechanism of 10 against AGE formation including chelation effect, trapping of MGO, and trapping of reactive oxygen species (ROS), which leads to oxidative degradation of 18 to 3,4-dihydroxybenzoic acid (15) and other fragments.
Investigating terephthalate biodegradation: Structural characterization of a putative decarboxylating cis-dihydrodiol dehydrogenase
Bains, Jasleen,Boulanger, Martin J.,Wulff, Jeremy E.
, p. 284 - 293,10 (2012)
As a highly coveted precursor molecule, terephthalate (Tph) continues to be used extensively for the production of polyethylene Tph bottles, polyester films, and textile fibers worldwide. Based on its detrimental physiological effects, Tph is now recognized as a serious environmental pollutant. While amenable to biodegradation and, in fact, traditionally neutralized by aerobic microbiological processes, our current lack of understanding of the enzymatic degradation of Tph at the molecular level presents a major impediment in the development of robust bioremediation strategies. The biodegradation of Tph proceeds through a single metabolic intermediate (a cis-dihydrodiol), which is subsequently converted to the end product (protocatechuate) by a decarboxylating cis-dihydrodiol dehydrogenase (TphB). Using iodide single-wavelength anomalous dispersion, we report the first structural characterization of TphB to 1.85 A resolution. Contrary to prior speculations, a fluorescent scan unambiguously shows that TphB coordinates Zn2 + and not Fe 2 +. The molecular architecture of TphB provides a rationale to the primary-level divergence observed between TphB and other cis-dihydrodiol dehydrogenases while explaining its intriguingly close evolutionary clustering with non-dihydrodiol dehydrogenases belonging to the isocitrate/isopropylmalate family of enzymes. Sequence and structural analyses reveal a putative substrate-binding pocket proximal to the bound Zn2 +. In silico substrate modeling in this putative binding pocket suggests a mechanistic sequence relying on H291, K295, and Zn2 + as core mediators of catalytic turnover. Overall, this study reveals novel structural and mechanistic insights into a decarboxylating cis-dihydrodiol dehydrogenase that mediates one of the two catalytic steps in the biodegradation of the environmental pollutant Tph.
Multi-Enzymatic Cascade Reactions for the Synthesis of cis,cis-Muconic Acid
Di Nardo, Giovanna,Gazzola, Silvia,Gilardi, Gianfranco,Pollegioni, Loredano,Rosini, Elena,Valetti, Francesca,Vignali, Elisa
, p. 114 - 123 (2021/10/07)
Lignin valorization allows the generation of a number of value-added products such as cis,cis-muconic acid (ccMA), which is widely used for the synthesis of chemicals for the production of biodegradable plastic materials. In the present work, we reported the first multi-enzymatic, one-pot bioconversion process of vanillin into ccMA. In details, we used four sequential reactions catalyzed by xanthine oxidase, O-demethylase LigM (and the tetrahydrofolate-regeneration enzyme methyl transferase MetE), decarboxylase AroY (based on the use of E. coli transformed cells) and catechol 1,2-dioxygenase CatA. The optimized lab-scale procedure allowed to reach, for the first time, the conversion of 5 mM vanillin into ccMA in ~30 h with a 90% yield: this achievement represents an improvement in terms of yields and time when compared to the use of a whole-cell system. This multi-enzymatic system represents a sustainable alternative for the production of a high value added product from a renewable resource. (Figure presented.).
Negative correlations between cultivable and active-yet-uncultivable pyrene degraders explain the postponed bioaugmentation
Jiang, Bo,Chen, Yating,Xing, Yi,Lian, Luning,Shen, Yaoxin,Zhang, Baogang,Zhang, Han,Sun, Guangdong,Li, Junyi,Wang, Xinzi,Zhang, Dayi
, (2021/09/24)
Bioaugmentation is an effective approach to remediate soils contaminated by polycyclic aromatic hydrocarbons (PAHs), but suffers from unsatisfactory performance in engineering practices, which is hypothetically explained by the complicated interactions between indigenous microbes and introduced degraders. This study isolated a cultivable pyrene degrader (Sphingomonas sp. YT1005) and an active pyrene degrading consortium (Gp16, Streptomyces, Pseudonocardia, Panacagrimonas, Methylotenera and Nitrospira) by magnetic-nanoparticle mediated isolation (MMI) from soils. Pyrene biodegradation was postponed in bioaugmentation with Sphingomonas sp. YT1005, whilst increased by 30.17% by the active pyrene degrading consortium. Pyrene dioxygenase encoding genes (nidA, nidA3 and PAH-RHDα-GP) were enriched in MMI isolates and positively correlated with pyrene degradation efficiency. Pyrene degradation by Sphingomonas sp. YT1005 only followed the phthalate pathway, whereas both phthalate and salicylate pathways were observed in the active pyrene degrading consortium. The results indicated that the uncultivable pyrene degraders were suitable for bioaugmentation, rather than cultivable Sphingomonas sp. YT1005. The negative correlations between Sphingomonas sp. YT1005 and the active-yet-uncultivable pyrene degraders were the underlying mechanisms of bioaugmentation postpone in engineering practices.
Iron-catalyzed arene C-H hydroxylation
Cheng, Lu,Wang, Huihui,Cai, Hengrui,Zhang, Jie,Gong, Xu,Han, Wei
, p. 77 - 81 (2021/10/05)
The sustainable, undirected, and selective catalytic hydroxylation of arenes remains an ongoing research challenge because of the relative inertness of aryl carbon-hydrogen bonds, the higher reactivity of the phenolic products leading to over-oxidized by-products, and the frequently insufficient regioselectivity. We report that iron coordinated by a bioinspired L-cystine-derived ligand can catalyze undirected arene carbon-hydrogen hydroxylation with hydrogen peroxide as the terminal oxidant. The reaction is distinguished by its broad substrate scope, excellent selectivity, and good yields, and it showcases compatibility with oxidation-sensitive functional groups, such as alcohols, polyphenols, aldehydes, and even a boronic acid. This method is well suited for the synthesis of polyphenols through multiple carbon-hydrogen hydroxylations, as well as the late-stage functionalization of natural products and drug molecules.
Ni-NiO heterojunctions: a versatile nanocatalyst for regioselective halogenation and oxidative esterification of aromatics
Bhardwaj, Nivedita,Goel, Bharat,Indra, Arindam,Jain, Shreyans K.,Singh, Ajit Kumar,Tripathi, Nancy
, p. 14177 - 14183 (2021/08/16)
Herein, we report a facile method for the synthesis of Ni-NiO heterojunction nanoparticles, which we utilized for the nuclear halogenation reaction of phenol and substituted phenols usingN-bromosuccinimide (NBS). A remarkablepara-selectivity was achieved for the halogenated products under semi-aqueous conditions. Interestingly, blocking of thepara-position of phenol offeredortho-selective halogenation. In addition, the Ni-NiO nanoparticles catalyzed the oxidative esterification of carbonyl compounds with alcohol, diol or dithiol in the presence of a catalytic amount of NBS. It was observed that the aromatic carbonyls substituted with an electron-donating group favoured nuclear halogenation, whereas an electron-withdrawing group substitution in carbonyl compounds facilitated the oxidation reaction. In addition, the catalyst was magnetically separated and recycled 10 times. The tuned electronic structure at the Ni-NiO heterojunction controlled selectivity and activity as no suchpara-selectivity was observed with commercially available NiO or Ni nanoparticles.
Thiols Act as Methyl Traps in the Biocatalytic Demethylation of Guaiacol Derivatives
Grimm, Christopher,Kroutil, Wolfgang,Pompei, Simona,Schiller, Christine,Schober, Lukas
supporting information, p. 16906 - 16910 (2021/07/02)
Demethylating methyl phenyl ethers is challenging, especially when the products are catechol derivatives prone to follow-up reactions. For biocatalytic demethylation, monooxygenases have previously been described requiring molecular oxygen which may cause oxidative side reactions. Here we show that such compounds can be demethylated anaerobically by using cobalamin-dependent methyltransferases exploiting thiols like ethyl 3-mercaptopropionate as a methyl trap. Using just two equivalents of this reagent, a broad spectrum of substituted guaiacol derivatives were demethylated, with conversions mostly above 90 %. This strategy was used to prepare the highly valuable antioxidant hydroxytyrosol on a one-gram scale in 97 % isolated yield.
Oxidative metabolism of typical phenolic compounds of Danshen by electrochemistry coupled to quadrupole time-of-flight tandem mass spectrometry
Cao, Jun,Chen, Yan,Dong, Xin,Yang, Juan,Ye, Li-Hong,Zhen, Xiao-Ting,Zheng, Hui
, (2020/02/04)
An electrochemistry coupled to online quadrupole time-of-flight tandem mass spectrometry (EC/Q-TOF/MS) was applied to investigate the oxidative transformation and metabolic pathway of five phenolic acids in Danshen sample. Simulation of the phase I oxidative metabolism was carried out in an electrochemical reactor equipped with a glassy carbon working electrode. The phase II reactivity of the generated oxidative products towards biomolecules (such as glutathione) was investigated by ways of covalent adduct formation experiments. The results obtained by EC/MS were compared with well-known in vitro studies by conducting rat liver microsome incubations. Structures of the electrochemically produced metabolites were identified by accurate mass measurement and previously results in vivo metabolites. It was indicated that the electrochemical oxidation was in good accordance with similar products found in vivo experiments. In conclusion, this work confirmed that EC/Q-TOF/MS was a promising analytical tool in the prediction of metabolic transformations of functional foods.
Method for industrially producing protocatechuic acid
-
Paragraph 0034; 0036; 0037; 0039; 0040; 0042, (2020/07/15)
The invention discloses a method for industrially producing protocatechuic acid. According to the invention, vanillic aldehyde is used as a raw material. The method comprises the following steps: firstly, performing catalytic oxidation on the raw material in an organic solvent to obtain 4-hydroxy-3-methoxybenzoic acid; and then removing a methyl group under the action of a demethylation reagent soas to finally obtain a target product, namely protocatechuic acid. The method is simple in process, has separation yield reaching 98% or above (metered by vanillin), enables product purity to reach 99% or above, greatly reduces synthesis cost, and is suitable for industrial large-scale production.
The fate of acylated anthocyanins in mildly heated neutral solution
Collins, Thomas M.,Dangles, Olivier,Fenger, Julie-Anne,Robbins, Rebecca J.
, (2020/03/23)
In neutral solution, anthocyanins acylated by hydroxycinnamic acids typically exhibit attractive blue colors and a higher resistance to color loss compared to their nonacylated homologs. However, they remain vulnerable to a poorly understood combination of oxidative and hydrolytic reactions that strongly contribute to color loss and limits their industrial applications. In this work, the thermal degradation of isolated red cabbage anthocyanins (0, 1 or 2 acyl groups) at pH 7 was investigated by UPLC-DAD-MS (low- and high-resolution). Non-oxidative alterations, including deacylation and intramolecular acyl transfer, were observed and found very dependent on the number and position of the acyl group(s) as well as on the presence of iron ions. At intermediate and advanced thermal degradation, several oxidative mechanisms were evidenced that lead to protocatechuic acid, phloroglucinaldehyde 2-O-glucoside, acylglycosides and derivatives of 2,4,6-trihydroxyphenylacetic acid and 3,5,7-trihydroxycoumarin. Based on the product distribution observed and on the impact of added Fe2+ ions and H2O2, possible degradation mechanisms are discussed. They likely start with a one- or two-electron transfer from the anionic base (a major colored form in neutral solution) to O2. The hydrogen peroxide produced could then further react as an electrophile with the anionic base and/or the hemiketal (major colorless hydrated form). This contribution to understanding the degradation mechanisms of anthocyanins around neutrality can open up new stabilization strategies to extend the range of their food applications to neutral media.