120-51-4Relevant articles and documents
HOMOMOLECULAR ESTERIFICATION OF ALDEHYDES CATALYZED BY HYDRIDOTETRAKIS(TRIPHENYLPHOSPHINE)RHODIUM(I)
Massoui, M.,Beaupere, D.,Nadjo, L.,Uzan, R.
, p. 345 - 354 (1983)
Homomolecular esterification of benzaldehyde has been carried out under mild conditions using RhH(PPh3)4 as catalyst.A kinetic study of the reaction has revealed a law rate of the form r=k2.The rate-limiting step in the mechanism is the complexation of the second aldehyde molecule with the catalyst.As expected from this result, the lactonisation of ortho-phthalaldehyde is instantaneous at 19 deg C.In this homomolecular esterification RhH(PPh3)3 is the catalytic species and slowly transfers its hydrogen ligand to benzaldehyde to form benzyl alcohol in concentration less than half of that of the catalyst.The complex Rh(PPh3)3 which is formed also catalyses the homomolecular esterification, but 1.6 times less readily than RhH(PPH3s)3.
Palladium nanoparticles immobilized on magnetic nanoparticles: An efficient semi-heterogeneous catalyst for carbonylation of aryl bromides
Dutta, Bishnu,Omar, Suheir,Natour, Suzana,Abu-Reziq, Raed
, p. 31 - 36 (2015)
We describe a method for supporting palladium nanoparticles on magnetic nanoparticles modified with amino-functionalized dihydro-imidazolium groups. This catalytic system is very efficient in alkoxy- and amino-carbonylation reactions of aryl bromides. The catalyst can be easily recovered from the reaction mixture by applying an external magnetic field and reused for over five consecutive cycles without a significant decrease in its activity.
Bisphosphine phenol and phenolate complexes of Mn(i): Manganese(i) catalyzed Tishchenko reaction
Kadassery, Karthika J.,MacMillan, Samantha N.,Lacy, David C.
, p. 12652 - 12655 (2018)
We synthesized new organomanganese complexes using the phenolic "pincer" type ligand H-POP. The coordination chemistry of H-POP with Mn(i) was explored, revealing a wide range of binding motifs. Finally, we found that complex 1 catalyzes the formation of benzyl benzoate from benzaldehyde in a Tishchenko reaction.
Catalytic, high-speed Tishchenko reaction using (2,7-dimethyl-1,8-biphenylenedioxy)bis(diisopropoxyaluminum) as a powerful bidentate catalyst
Ooi, Takashi,Miura, Tomoya,Takaya, Keisuke,Maruoka, Keiji
, p. 7695 - 7698 (1999)
Exceedingly high-speed Tishchenko reaction of various aldehydes can be realized by using a powerful, bidentate aluminum catalyst. This catalytic system is also applicable to the highly stereoselective intramolecular Tishchenko reduction of β-hydroxy ketones.
Modification of mesoporous silicate SBA-15 with tris[bis(trimethylsilyl) amido]samarium and its utility in Tishchenko reaction
Chen, Yinghua,Zhu, Zhenyu,Zhang, Jie,Shen, Jianzhong,Zhou, Xigeng
, p. 3783 - 3789 (2005)
Treatment of mesoporous silicate SBA-15 with Sm[N(SiMe3) 2]3 led to the formation of a novel organolanthanide/ inorganic hybrid material [SBA-15]Sm[N(SiMe3)2] x via abstraction of N(SiMe3)2 by terminal silanol groups and subsequent surface silylation. The hybrid material was characterized by elemental analyses, IR spectroscopy, X-ray diffraction, and nitrogen sorption, indicating a successful tailoring inside the silicate SBA-15 and the maintenance of the well-ordered mesostructure. This hybrid material is a promising heterogeneous catalyst for the Tishchenko reaction, where it is superior to the homogeneous correspondent in deactivation behavior, reusability and relative tolerance to oxygen, particularly in the control of selectivity of mixed Tishchenko reaction due to the steric hindrance and the diffusion control derived from the surface confinement.
Superbase-promoted acylation of hindered alcohols
D'Sa, Bosco A.,Verkade, John G.
, p. 2963 - 2966 (1996)
The commercially available nonionic superbase P(MeNCH2CH2)3N (1a) is very useful for the acylation of unreactive hindered alcohols as well as acid-sensitive alcohols. The reactions proceed in high yields using an acid anhydride, and 1a can be regenerated in a single step. The relative rates for benzoylation of (±)-menthol in C6D6 using conventional acylation reagents and strong nonionic bases are compared. In general, acetylation with 1a is accelerated in the polar solvent CH3CN whereas benzoylation is faster in the nonpolar solvent C6H6. The benzoylation intermediate RC(O)P(MeNCH2CH2)3N+ was found to be in equilibrium with 1a, with lower temperatures favoring the intermediate. The relative stabilities of several known acylating intermediates are compared.
Novel and efficient method for esterification catalyzed by 1-glycyl-3-methyl imidazolium chloride-iron (III) complex
Karthikeyan, Parasuraman,Bhagat, Pundlik Rambhau,Kumar, Sellappan Senthil,Muskawar, Prashant Narayan,Aswar, Sachin Arunrao
, p. 983 - 990 (2012)
1-Glycyl-3-methyl imidazolium chloride-iron (III) complex [[Gmim]Cl-Fe(III)] was found to be a heterogeneous catalyst for an efficient and greener solvent free synthesis of esters by the condensation of carboxylic acids and alcohols with excellent yield at ambient temperature. This operation formulates very interesting, ecological perspective due to simple reaction condition, isolation, and purification of products. In addition, this method features reusability of catalyst, reduced waste, thus making new protocol more environmentally suitable whilst no catalyst leaching was observed. Iranian Chemical Society 2012.
Lanthanoid-Catalyzed Aldehyde Dimerization and Its Application to Polyester Synthesis
Onozawa, Shun-ya,Sakakura, Toshiyasu,Tanaka, Masato
, p. 531 - 534 (1994)
A wide variety of aldehydes are efficiently dimerized to esters by a catalytic amount of (C5Me5)2LnCH(SiMe3)2 (Ln=Nd, La).The reaction is applicable to the synthesis of polyesters from dialdehydes.A stoichiometric reaction of the lanthanum complex with benzaldehyde indicates the intermediacy of alkoxo complexes in the catalysis.
Synthesis of Pyrazines and Quinoxalines via Acceptorless Dehydrogenative Coupling Routes Catalyzed by Manganese Pincer Complexes
Daw, Prosenjit,Kumar, Amit,Espinosa-Jalapa, Noel Angel,Diskin-Posner, Yael,Ben-David, Yehoshoa,Milstein, David
, p. 7734 - 7741 (2018)
Base-metal catalyzed dehydrogenative self-coupling of 2-amino alcohols to selectively form functionalized 2,5-substituted pyrazine derivatives is presented. Also, 2-substituted quinoxaline derivatives are synthesized by dehydrogenative coupling of 1,2-diaminobenzene and 1,2-diols. In both cases, water and hydrogen gas are formed as the sole byproducts. The reactions are catalyzed by acridine-based pincer complexes of earth-abundant manganese.
Ruthenium complexes featuring cooperative phosphine-pyridine-iminophosphorane (PNN) ligands: Synthesis, reactivity and catalytic activity
Cheisson, Thibault,Mazaud, Louis,Auffrant, Audrey
, p. 14521 - 14530 (2018)
The coordination to ruthenium(ii) centres of two phosphine-pyridine-iminophosphorane ligands LR (PPh2CH2(C6H3N)CH2NPR3, R = Ph or Cy) differing by the nature of the substituent of the PN phosphorus was explored. Coordination to [RuCl2(PPh3)3] afforded the complexes [RuLRCl2(PPh3)] that were successfully deprotonated at the acidic phosphinomethyl position. With LCy, coordination led to a mixture of two isomers. The complexes [RuLRHCl(PPh3)] were similarly obtained from [RuHCl(PPh3)3]. The stability of these complexes depends on the ligand substitution pattern; with LPh a CH activation process took place, while [RuLCyHCl(PPh3)] was thermally stable. Deprotonation of this latter complex was achieved and gave a catalytically competent species for the acceptorless dehydrogenative coupling of alcohols.
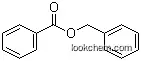
 Xn:Harmful;
Xn:Harmful;



